Abstract
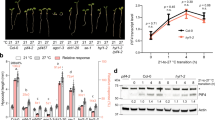
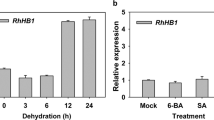
Plants have evolved sophisticated systems in response to environmental changes, and growth arrest is a common strategy used to enhance stress tolerance. Despite the growth–survival trade-off being essential to the shaping of plant productivity, the mechanisms balancing growth and survival remain largely unknown. Aquaporins play a crucial role in growth and stress responses by controlling water transport across membranes. Here, we show that RhPIP2;1, an aquaporin from rose (Rosa sp.), interacts with a membrane-tethered MYB protein, RhPTM. Water deficiency triggers nuclear translocation of the RhPTM C terminus. Silencing of RhPTM causes continuous growth under drought stress and a consequent decrease in survival rate. RNA sequencing (RNA-seq) indicated that RhPTM influences the expression of genes related to carbohydrate metabolism. Water deficiency induces phosphorylation of RhPIP2;1 at Ser 273, which is sufficient to promote nuclear translocation of the RhPTM C terminus. These results indicate that the RhPIP2;1-RhPTM module serves as a key player in orchestrating the trade-off between growth and stress survival in Rosa.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request. All primers used in this study are described in Supplementary Table 3. Information on the genes used in this study is given in Supplementary Table 4. RNA-Seq data that support the findings of this study have been deposited in the NCBI Bioproject database under the accession number PRJNA486271 (https://www.ncbi.nlm.nih.gov/bioproject/?term=PRJNA486271). The proteomic data of validation for anti-RhPTM antibody and RhPIP2;1 phosphorylation have been deposited in the PRIDE archive (Nos. PXD011942 and PXD011943, respectively; https://www.ebi.ac.uk/pride/archive).
References
Achard, P. et al. Integration of plant responses to environmentally activated phytohormonal signals. Science 311, 91–94 (2006).
Claeys, H. & Inze, D. The agony of choice: how plants balance growth and survival under water-limiting conditions. Plant Physiol. 162, 1768–1779 (2013).
Sharp, R. E. & LeNoble, M. E. ABA, ethylene and the control of shoot and root growth under water stress. J. Exp. Bot. 53, 33–37 (2002).
Xiong, L., Wang, R. G., Mao, G. & Koczan, J. M. Identification of drought tolerance determinants by genetic analysis of root response to drought stress and abscisic acid. Plant Physiol. 142, 1065–1074 (2006).
Chaumont, F. & Tyerman, S. D. Aquaporins: highly regulated channels controlling plant water relations. Plant Physiol. 164, 1600–1618 (2014).
Verdoucq, L., Rodrigues, O., Martinière, A., Luu, D. T. & Maurel, C. Plant aquaporins on the move: reversible phosphorylation, lateral motion and cycling. Curr. Opin. Plant Biol. 22, 101–107 (2014).
Tyerman, S. D., Niemietz, C. M. & Bramley, H. Plant aquaporins: multifunctional water and solute channels with expanding roles. Plant Cell Environ. 25, 173–194 (2002).
Martre, P. Plasma membrane aquaporins play a significant role during recovery from water deficit. Plant Physiol. 130, 2101–2110 (2002).
Bienert, G. P. & Chaumont, F. in Transporters and Pumps in Plant Signaling (eds Geisler, M. and Venema, K.) 3–36 (Springer, Berlin, 2011).
Prado, K. & Maurel, C. Regulation of leaf hydraulics: from molecular to whole plant levels. Front. Plant Sci. 4, 255 (2013).
Lian, H. L. et al. Upland rice and lowland rice exhibited different PIP expression under water deficit and ABA treatment. Cell Res. 16, 651–660 (2006).
Guo, L. et al. Expression and functional analysis of the rice plasma-membrane intrinsic protein gene family. Cell Res. 16, 277–286 (2006).
Maurel, C. et al. Aquaporins in plants. Physiol. Rev. 95, 1321–1358 (2015).
Boursiac, Y. et al. Stimulus-induced downregulation of root water transport involves reactive oxygen species-activated cell signalling and plasma membrane intrinsic protein internalization. Plant J. 56, 207–218 (2008).
Luu, D.-T., Martinière, A., Sorieul, M., Runions, J. & Maurel, C. Fluorescence recovery after photobleaching reveals high cycling dynamics of plasma membrane aquaporins in Arabidopsis roots under salt stress: root plasma membrane aquaporin trafficking in salt stress. Plant J. 69, 894–905 (2012).
Törnroth-Horsefield, S. et al. Structural mechanism of plant aquaporin gating. Nature 439, 688–694 (2006).
Nyblom, M. et al. Structural and functional analysis of SoPIP2;1 mutants adds insight into plant aquaporin gating. J. Mol. Biol. 387, 653–668 (2009).
Bienert, G. P. & Chaumont, F. Aquaporin-facilitated transmembrane diffusion of hydrogen peroxide. Biochim. Bioph. Acta 1840, 1596–1604 (2014).
Thiagarajah, J. R., Chang, J., Goettel, J. A., Verkman, A. S. & Lencer, W. I. Aquaporin-3 mediates hydrogen peroxide-dependent responses to environmental stress in colonic epithelia. Proc. Natl Acad. Sci. USA 114, 568 (2017).
Bienert, G. P., Heinen, R. B., Berny, M. C. & Chaumont, F. Maize plasma membrane aquaporin ZmPIP2;5, but not ZmPIP1;2, facilitates transmembrane diffusion of hydrogen peroxide. Biochim. Bioph. Acta 1838, 216–222 (2014).
Bienert, G. P., Bienert, M. D., Jahn, T. P., Boutry, M. & Chaumont, F. Solanaceae XIPs are plasma membrane aquaporins that facilitate the transport of many uncharged substrates. Plant J. 66, 306–317 (2011).
Azad, A. K., Yoshikawa, N., Ishikawa, T., Sawa, Y. & Shibata, H. Substitution of a single amino acid residue in the aromatic/arginine selectivity filter alters the transport profiles of tonoplast aquaporin homologs. Biochim. Bioph. Acta 1818, 1–11 (2012).
Tian, S. et al. Plant aquaporin AtPIP1;4 links apoplastic H2O2 induction to disease immunity pathways. Plant Physiol. 171, 1635–1650 (2016).
Rodrigues, O. et al. Aquaporins facilitate hydrogen peroxide entry into guard cells to mediate ABA- and pathogen-triggered stomatal closure. Proc. Natl Acad. Sci. USA 114, 9200–9205 (2017).
Ma, N. et al. Rh-PIP2;1, a rose aquaporin gene, is involved in ethylene-regulated petal expansion. Plant Physiol. 148, 894 (2008).
Chen, W. et al. Involvement of rose aquaporin RhPIP1;1 in ethylene-regulated petal expansion through interaction with RhPIP2;1. Plant Mol. Biol. 83, 219–233 (2013).
Zelazny, E. et al. FRET imaging in living maize cells reveals that plasma membrane aquaporins interact to regulate their subcellular localization. Proc. Natl Acad. Sci. USA 104, 12359–12364 (2007).
Li, L. et al. Identification of AtSM34, a novel tonoplast intrinsic protein-interacting polypeptide expressed in response to osmotic stress in germinating seedlings. Chin. Sci. Bull. 56, 3518–3530 (2011).
Slabaugh, E., Held, M. & Brandizzi, F. Control of root hair development in Arabidopsis thaliana by an endoplasmic reticulum anchored member of the R2R3‐MYB transcription factor family. Plant J. 67, 395 (2011).
Chen, Y.-N., Slabaugh, E. & Brandizzi, F. Membrane-tethered transcription factors in Arabidopsis thaliana: novel regulators in stress response and development. Curr. Opin. Plant Biol. 11, 695–701 (2008).
Prado, K. et al. Regulation of Arabidopsis leaf hydraulics involves light-dependent phosphorylation of aquaporins in veins. Plant Cell 25, 1029–1039 (2013).
Grondin, A. et al. Aquaporins contribute to ABA-triggered stomatal closure through OST1-mediated phosphorylation. Plant Cell 27, 1945–1954 (2015).
Azad, T. et al. A LATS biosensor screen identifies VEGFR as a regulator of the Hippo pathway in angiogenesis. Nat. Commun. 9, 1061 (2018).
Fujikawa, Y. & Kato, N. Technical advance: split luciferase complementation assay to study protein–protein interactions in Arabidopsis protoplasts. Plant J. 52, 185–195 (2007).
Fetter, K. Interactions between plasma membrane aquaporins modulate their water channel activity. Plant Cell 16, 215–228 (2004).
Li, D. D. et al. Cotton plasma membrane intrinsic protein 2s (PIP2s) selectively interact to regulate their water channel activities and are required for fibre development. New Phytol. 199, 695–707 (2013).
Bellati, J. et al. Novel aquaporin regulatory mechanisms revealed by interactomics. Mol. Cell Proteomics 15, 3473–3487 (2016).
Qing, D. et al. Quantitative and functional phosphoproteomic analysis reveals that ethylene regulates water transport via the C-terminal phosphorylation of aquaporin PIP2;1 in Arabidopsis. Mol. Plant 9, 158–174 (2016).
Prak, S. et al. Multiple phosphorylations in the C-terminal tail of plant plasma membrane aquaporins role in subcellular trafficking of AtPIP2; 1 in response to salt stress. Mol. Cell Proteomics 7, 1019–1030 (2008).
Niittylä, T. et al. Temporal analysis of sucrose-induced phosphorylation changes in plasma membrane proteins of Arabidopsis. Mol. Cell. Proteomics 6, 1711–1726 (2007).
Di Pietro, M. et al. Coordinated post-translational responses of aquaporins to abiotic and nutritional stimuli in Arabidopsis roots. Mol. Cell. Proteomics M113, 028241 (2013).
Johansson, I. et al. Water transport activity of the plasma membrane aquaporin PM28A is regulated by phosphorylation. Plant Cell 10, 451–459 (1998).
Chinnusamy, V. & Zhu, J.-K. Epigenetic regulation of stress responses in plants. Curr. Opin. Plant Biol. 12, 133–139 (2009).
Nolan, T. M. et al. Selective autophagy of BES1 mediated by DSK2 balances plant growth and survival. Dev. Cell 41, 33–46 (2017).
Wang, P. et al. Reciprocal regulation of the TOR kinase and ABA receptor balances plant growth and stress response. Mol. Cell 69, 100–112 (2018).
Tian, J. et al. TRV-GFP: a modified tobacco rattle virus vector for efficient and visualizable analysis of gene function. J. Exp. Bot. 65, 311–322 (2014).
Wang, W., Vignani, R., Scali, M. & Cresti, M. A universal and rapid protocol for protein extraction from recalcitrant plant tissues for proteomic analysis. Electrophoresis 27, 2782–2786 (2006).
Wiśniewski, J. R., Zougman, A., Nagaraj, N. & Mann, M. Universal sample preparation method for proteome analysis. Nat. Methods 6, 359–362 (2009).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367 (2008).
Acknowledgements
We thank Z. Li (Biological Mass Spectrometry Laboratory of the Functional Genomic Technology Centre of China Agricultural University) for LC–MS experiments. We thank J. Zhu (Jingjie PTM Biolabs, Inc.) for analysis of LC–MS data. We thank Z. Gong (China Agricultural University) for providing the pSuper1300 vector. We thank D. Zhang (Tsinghua University) for his generous help with MYTH screening. We thank PlantScribe (www.plantscribe.com) for careful editing of this article. This work was supported by the National Natural Science Foundation of China (grant Nos. 31520103913, 31522049, 31730079 and 31401914) and the 111 Project of the Ministry of Education (No. B17043).
Author information
Authors and Affiliations
Contributions
S.Z., M.F., W.C., J.L., Y.W. and Y.L. performed the experiments. N.M. and J.G. designed the research. X.Z., C.J., S.G. and N.M. provided technical support, conceptual advice and data analysis. S.Z., N.M. and J.G. wrote the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–10, Supplementary Methods, Supplementary References, legends for Supplementary Tables and Datasets, and Supplementary Tables 1–3.
Supplementary Dataset 1
The entire database search results for validation of anti-RhPTM antibody by LC–MS.
Supplementary Dataset 2
Differentially expressed genes in RhPTM-silenced rose plants.
Supplementary Dataset 3
The entire database search results for phosphorylation of RhPIP2;1 by LC–MS.
Rights and permissions
About this article
Cite this article
Zhang, S., Feng, M., Chen, W. et al. In rose, transcription factor PTM balances growth and drought survival via PIP2;1 aquaporin. Nat. Plants 5, 290–299 (2019). https://doi.org/10.1038/s41477-019-0376-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-019-0376-1
This article is cited by
-
Petal size is controlled by the MYB73/TPL/HDA19-miR159-CKX6 module regulating cytokinin catabolism in Rosa hybrida
Nature Communications (2023)
-
Responsive Transcriptome Analysis of Senecio vulgaris L. Under Different Drought Stresses
Journal of Plant Growth Regulation (2023)
-
Transcription factors RhPIF4/8 and RhHY5 regulate autophagy-mediated petal senescence in rose (Rosa hybrida)
Horticulture Advances (2023)
-
LiMYB108 is involved in floral monoterpene biosynthesis induced by light intensity in Lilium ‘Siberia’
Plant Cell Reports (2023)
-
Disruption of transcription factor RhMYB123 causes the transformation of stamen to malformed petal in rose (Rosa hybrida)
Plant Cell Reports (2022)