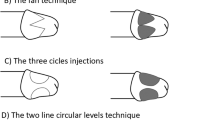
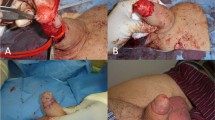
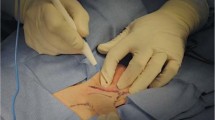
Abstract
Despite the controversy regarding penile augmentation (PA), glans penis augmentation (GPA) is used in penile reconstructive surgery in selected patients with small glans penis. Since 2003, injectable hyaluronic acid (HA) gel has been used for GPA. The attractiveness of HA gel fillers and interest in this technique have led to the use of other fillers for GPA, particularly irreversible fillers that improve the longevity of HA gels. Conversely, indirect glans augmentation via implantation of dermofat grafts or scaffolds between the corpus spongiosum and the distal tip of the corpus cavernosum is used to overcome poor longevity of the gel and glans surface undulation, which are the primary limitations of GPA using HA gel. Unfortunately, these implants form hard painful lumps over time in most patients and are difficult to remove. Blunt dissection is an invasive procedure that may cause vascular compromise and consequent glans necrosis in a few patients. We present our review regarding the current status of GPA for a more comprehensive understanding of this subject. Additionally, we have discussed a few complicated cases that were referred to us.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Moon DG, Yoo JW, Bae JH, Han CS, Kim YK, Kim JJ. Sexual function and psychological characteristics of penile paraffinoma. Asian J Androl. 2003;5:191–4.
Lindau ST, Schumm LP, Laumann EO, Levinson W, O’Muircheartaigh CA, Waite LJ. A study of sexuality and health among older adults in the United States. N Engl J Med. 2007;357:762–74.
Kim JJ, Kwak TI, Jeon BG, Cheon J, Moon DG. Human glans penis augmentation using injectable hyaluronic acid gel. Int J Impot Res. 2003;15:439–543.
Moon DG, Kwak TI, Cho HY, Bae JH, Park HS, Kim JJ. Augmentation of glans penis using injectable hyaluronic acid gel. Int J Impot Res. 2003;15:456–60.
Kwak TI, Jin MH, Kim JJ, Moon DG. Long-term effects of glans penis augmentation using injectable hyaluronic acid gel for premature ejaculation. Int J Impot Res. 2008;20:425–8.
Kwak TI, Oh M, Kim JJ, Moon, du G. The effects of penile girth enhancement using injectable hyaluronic acid gel, a filler. J Sex Med. 2011;8:3407–13.
Yang DY, Ko K, Lee SH, Moon DG, Kim JW, Lee WK. Efficacy and safety of newly developed cross-linked dextran gel injection for glans penis augmentation with a novel technique. Asian J Androl. 2018;20:80–4.
Abdallah H, Abdelnasser T, Hosny H, Selim O, Al-Ahwany A, Shamloul R. Treatment of premature ejaculation by glans penis augmentation using hyaluronic acid gel: a pilot study. Andrologia. 2012;44:650–3.
Kim JJ, Kwak TI, Jeon BG, Cheon J, Moon DG. Effects of glans penis augmentation using hyaluronic acid gel for premature ejaculation. Int J Impot Res. 2004;16:547–51.
Duffy DM. Complications of fillers: overview. Dermatol Surg. 2005;31:1626–33.
Alijotas-Reig J, Fernandez-Figueras MT, Puig L. Inflammatory, immune-mediated adverse reactions related to soft tissue dermal fillers. Semin Arthritis Rheum. 2013;43:241–58.
Moon du G, Kwak TI, Kim JJ. Glans penis augmentation using hyaluronic acid gel as an injectable filler. World J Mens Health. 2015;33:50–61.
Moon DG. General principles of glans penis augmentation. In: Park NC, Kim SW, Moon DG, editors. Penile augmentation. Berlin, Heidelberg: Springer; 2016. p. 169–73.
Park TH, Seo SW, Kim JK, Chang CH. Clinical experience with hyaluronic acid-filler complications. J Plast Reconstr Aesthet Surg. 2011;64:892–6.
Daines SM, Williams EF. Complications associated with injectable soft-tissue fillers: a 5-year retrospective review. JAMA Facial Plast Surg. 2013;15:226–31.
Vanaman M, Fabi SG, Carruthers J. Complications in the cosmetic dermatology patient: a review and our experience (part 1). dermatologic surgery: official publication for American Society for. Dermatol Surg [Et al]. 2016;42:1–11.
DeLorenzi C.Complications of injectable fillers, part 2: vascular complications.Aesthet Surg J. 2014;34:584–600.
DeLorenzi C. Complications of injectable fillers, part I. Aesthet Surg J. 2013;33:561–75.
Rohrich RJ, Ghavami A, Crosby MA. The role of hyaluronic acid fillers (Restylane) in facial cosmetic surgery: review and technical considerations. Plast Reconstr Surg. 2007;120:41s–54s.
Klein AW. Soft tissue augmentation 2006: filler fantasy. Dermatol Ther. 2006;19:129–33.
Funt D, Pavicic T. Dermal fillers in aesthetics: an overview of adverse events and treatment approaches. Clin Cosmet Investig Dermatol. 2013;6:295–316.
Surgery ASfAP. 2016 cosmetic surgery National Data Bank Statistics 2016. https://www.surgery.org/sites/default/files/ASAPS-Stats2016.pdf. Accessed May 10, 2018.
Elson ML. Soft tissue augmentation. A review. Dermatol Surg. 1995;21:491–500.
Dover JS. The filler revolution has just begun. Plast Reconstr Surg. 2006;117:38S–40S.
Watson W, Kaye RL, Klein A, Stegman S. Injectable collagen: a clinical overview. Cutis. 1983;31:543–6.
Sclafani AP, Romo T 3rd. Injectable fillers for facial soft tissue enhancement. Facial Plast Surg. 2000;16:29–34.
Smith KC. Reversible vs. nonreversible fillers in facial aesthetics: concerns and considerations. Dermatol Online J. 2008;14:3.
Monheit GD, Narins RS, Mariwalla K. NASHA family. In: Carruthers J, Carruthers A, Dover JS, Alam M, editors. Soft tissue augmentation: procedures in cosmetic dermatology series. New York, NY: Saunders Elsevier; 2012. p. 10–22.
Matarasso SL, Carruthers JD, Jewell ML. Consensus recommendations for soft-tissue augmentation with nonanimal stabilized hyaluronic acid (Restylane). Plast Reconstr Surg. 2006;117:3S–34S.
Turlier V, Delalleau A, Casas C, Rouquier A, Bianchi P, Alvarez S, et al. Association between collagen production and mechanical stretching in dermal extracellular matrix: in vivo effect of cross-linked hyaluronic acid filler. A randomised, placebo-controlled study. J Dermatol Sci. 2013;69:187–94.
Quan T, Wang F, Shao Y, Rittie L, Xia W, Orringer JS, et al. Enhancing structural support of the dermal microenvironment activates fibroblasts, endothelial cells, and keratinocytes in aged human skin in vivo. J Invest Dermatol. 2013;133:658–67.
Monheit GD, Coleman KM. Hyaluronic acid fillers. Dermatol Ther. 2006;19:141–50.
Wollina U, Goldman A. Hyaluronic acid dermal fillers: safety and efficacy for the treatment of wrinkles, aging skin, body sculpturing and medical conditions. Clin Med Rev Ther. 2011;2011:107–21.
Tezel A, Fredrickson GH. The science of hyaluronic acid dermal fillers. J Cosmet Laser Ther. 2008;10:35–42.
Gold M. The science and art of hyaluronic acid dermal filler use in esthetic applications. J Cosmet Dermatol. 2009;8:301–7.
Tran C, Carraux P, Micheels P, Kaya G, Salomon D. In vivo bio-integration of three hyaluronic acid fillers in human skin: a histological study. Dermatology. 2014;228:47–54.
Gladstone HB, Cohen JL. Adverse effects when injecting facial fillers. Semin Cutan Med Surg. 2007;26:34–9.
Requena L, Requena C, Christensen L, Zimmermann US, Kutzner H, Cerroni L. Adverse reactions to injectable soft tissue fillers. J Am Acad Dermatol. 2011;64:1–34.
Artzi O, Loizides C, Verner I, Landau M. Resistant and recurrent late reaction to hyaluronic acid-based gel. Dermatol Surg. 2016;42:31–7.
Perovic S, Radojicic ZI, Djordjevic M, Vukadinovic VV. Enlargement and sculpturing of a small and deformed glans. J Urol. 2003;170:1686–90.
Shaeer O. Supersizing the penis following penile prosthesis implantation. J Sex Med. 2010;7:2608–16.
Okubo M, Hyakusoku H, Kanno K, Fumiiri M. Complications after injection mammaplasty. Aesthetic Plast Surg. 1992;16:181–7.
Christensen LH, Breiting VB, Aasted A, Jorgensen A, Kebuladze I. Long-term effects of polyacrylamide hydrogel on human breast tissue. Plast Reconstr Surg. 2003;111:1883–90.
Cheng NX, Wang YL, Wang JH, Zhang XM, Zhong H. Complications of breast augmentation with injected hydrophilic polyacrylamide gel. Aesthetic Plast Surg. 2002;26:375–82.
Chen B, Song H. Management of breast deformity after removal of injectable polyacrylamide hydrogel: Retrospective study of 200 cases for 7 years. Aesthetic Plast Surg. 2016;40:482–91.
de Bree R, Middelweerd MJ, van der Waal I. Severe granulomatous inflammatory response induced by injection of polyacrylamide gel into the facial tissue. Arch Facial Plast Surg. 2004;6:204–6.
Liu Y, Cen Y, Xu XW, Duan WQ. [Analysis of complications induced by polyacrylamide hydrogel injection for augmentation mammaplasty]. Zhonghua Zheng Xing Wai Ke Za Zhi. 2005;21:464–6.
Bergeret-Galley C, Latouche X, Illouz YG. The value of a new filler material in corrective and cosmetic surgery: DermaLive and DermaDeep. Aesthetic Plast Surg. 2001;25:249–55.
Sidwell RU, Dhillon AP, Butler PE, Rustin MH. Localized granulomatous reaction to a semi-permanent hyaluronic acid and acrylic hydrogel cosmetic filler. Clin Exp Dermatol. 2004;29:630–2.
Nicolau PJ. Long-lasting and permanent fillers: biomaterial influence over host tissue response. Plast Reconstr Surg. 2007;119:2271–86.
Rohrich RJ, Monheit G, Nguyen AT, Brown SA, Fagien S. Soft-tissue filler complications: the important role of biofilms. Plast Reconstr Surg. 2010;125:1250–6.
Christensen LH. Host tissue interaction, fate, and risks of degradable and nondegradable gel fillers. Dermatol Surg. 2009;35:1612–9.
Bentkover SH. The biology of facial fillers. Facial Plast Surg. 2009;25:73–85.
Parada MB, Michalany NS, Hassun KM, Bagatin E, Talarico S. A histologic study of adverse effects of different cosmetic skin fillers. Skinmed. 2005;4:345–9.
Zyczkowski M, Prokopowicz G, Zajecki W, Paradysz A. Complications following endoscopic treatment of vesicoureteric reflux with Deflux((R)) - two case studies. Cent Eur J Urol. 2012;65:230–1.
Brody HJ. Use of hyaluronidase in the treatment of granulomatous hyaluronic acid reactions or unwanted hyaluronic acid misplacement. Dermatol Surg. 2005;31:893–7.
Hirsch RJ, Brody HJ, Carruthers JD. Hyaluronidase in the office: a necessity for every dermasurgeon that injects hyaluronic acid. J Cosmet Laser Ther. 2007;9:182–5.
Perovic SV, Scepanovic DR, Vukadinovic VM, Djakovic NG, Djordjevic ML. [Penile disassembly technique: a new approach in the surgical reconstruction of hypospadias]. Prog Urol. 1999;9:371–9.
Shaeer O. Shaeer’s glans augmentation technique: a pilot study. J Sex Med. 2012;9:3264–9.
Lowe NJ, Maxwell CA, Patnaik R. Adverse reactions to dermal fillers: review. Dermatol Surg. 2005;31:1616–25.
Lenzer J. Watching over the medical device industry. BMJ. 2009;338:b2321.
Lohman ME, Ghobadi CW, Xu S. Device safety implications of the clinical data leading to US Food and Drug Administration approval of soft-tissue fillers: A systematic review. JAMA Facial Plast Surg. 2017;19:421–9.
Yang DY, Lee WK, Kim SC. Tolerability and efficacy of newly developed penile injection of cross-linked dextran and polymethylmethacrylate mixture on penile enhancement: 6 months follow-up. Int J Impot Res. 2013;25:99–103.
Kim MT, Ko K, Lee WK, Kim SC, Yang DY. Long-term safety and longevity of a mixture of polymethyl methacrylate and cross-linked dextran (Lipen-10(R)) after penile augmentation: Extension study from six to 18 months of follow-up. World J Mens Health. 2015;33:202–8.
Park TH, Seo SW, Kim JK, Chang CH. Clinical outcome in a series of 173 cases of foreign body granuloma: improved outcomes with a novel surgical technique. J Plast Reconstr Aesthet Surg. 2012;65:29–34.
Park TH, Seo SW, Kim JK, Chang CH. Clinical experience with polymethylmethacrylate microsphere filler complications. Aesthetic Plast Surg. 2012;36:421–6.
Lemperle G, Rullan PP, Gauthier-Hazan N. Avoiding and treating dermal filler complications. Plast Reconstr Surg. 2006;118:92S–107S.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ahn, S.T., Il Kwak, T., Park, K.S. et al. Complications of glans penis augmentation. Int J Impot Res 31, 245–255 (2019). https://doi.org/10.1038/s41443-018-0097-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41443-018-0097-4
This article is cited by
-
Complications and outcomes following injection of foreign material into the male external genitalia for augmentation: a single-centre experience and systematic review
International Journal of Impotence Research (2023)
-
Surgical treatment of premature ejaculation: a narrative review
International Journal of Impotence Research (2023)
-
Glans penis augmentation: when, how, and why?
International Journal of Impotence Research (2022)
-
New trend in medicine: aesthetic genital surgery
International Journal of Impotence Research (2022)
-
Current and emerging treatment options for premature ejaculation
Nature Reviews Urology (2022)