Abstract
Previous studies have reported that a high free fat mass (FFM) was associated with elevated blood pressure in children. However, the observed relationship could be confounded by body weight. Thus, we performed cross-sectional analyses to understand the differences between the percentage of free fat mass (FFM%) and FFM in relation to blood pressure in healthy Chinese children. A total number of 2671 (1264 girls; aged 6–14 years) healthy Chinese children was recruited in 2014 (baseline). We further prospectively examined whether these indices predicted hypertension risk during the 2 years of follow-up (2014–2016) among 2094 participants who were free of hypertension at the baseline. Blood pressure was repeatedly measured in 2014, 2015 and 2016. Hypertension was defined as either high systolic blood pressure and/or high diastolic blood pressure (≥age- and sex-specific 95th percentile for Chinese children). The baseline FFM was assessed by bio-impedance analysis, and FFM% was calculated as FFM divided by the body weight. A high FFM was associated with a high baseline blood pressure and high hypertension risk after adjustment for potential confounders (all p < 0.001). By contrast, a high FFM% was associated with a low baseline blood pressure and low hypertension risk in the fully adjusted model (all p < 0.001). Each one-percent increment of FFM% was associated with a 9% lower risk of developing hypertension (adjusted hazard ratio = 0.91; 95% confidence interval: 0.89, 0.94). A high FFM% was associated with a low blood pressure and low hypertension risk in healthy Chinese children.
Similar content being viewed by others
Introduction
Obesity is a well-documented risk factor for childhood hypertension [1, 2]. A high fat mass (FM) and high percentage of fat mass (FM%) were observed to be associated with a high risk of childhood hypertension [3,4,5]. Unexpectedly, previous studies have consistently showed that a high free fat mass (FFM), or lean body mass (a strongly correlated index with FFM), was associated with a high blood pressure or high hypertension risk in children regardless of sample size (range: 72–6863), body composition measurement, age, and ethnicities [6,7,8,9,10,11]. Studies based on the FFM index (FFM in kilograms divided by the squared height in meters) [12, 13], or lean body mass index (lean body mass in kilograms divided by the squared height in meters) [14, 15], or lean body mass index z score [16] generated similar results. This association was thought to be due to the decreased ability of FFM to metabolize glucose and lipids [16] or altered cardiac output [7]. Notably, these studies examined absolute FFM or the FFM index rather than the percentage of FFM (FFM%). The observed positive relationship between FFM and blood pressure could be confounded by high body weight and high FM. FFM is increased simultaneously with the increase in FM in children and adolescents [17, 18]. The situation was similar in previous studies focusing on obesity and cardiovascular function. The most important limitation in these studies was to use BMI, rather than body composition indices (e.g., FM% and FFM%), to classify obesity [19]. In this context, FFM%, which is calculated by FFM divided by body weight, is considered a better index, relative to FFM, to reflect cardiovascular function [19]. In a case-control study including 94 children aged 6 to 18 years with hypertension, children with hypertension had a larger lean body mass percentage, which was strongly correlated with FFM%, compared with healthy control children [20]. However, this study was limited by its small sample size, wide range in age, and retrospective study design.
Therefore, we aimed to examine the differences between FFM% and FFM in the association with blood pressure during two years of follow-up, after the adjustment of potential confounders, in a community-based cohort study including approximately 2,700 healthy Chinese children. In secondary analyses, the associations between FM and FM% and blood pressure were also examined.
Subjects and Methods
Study population
The current study was conducted at five primary schools in Gaohang Town, Shanghai, China. Briefly, all the children (n = 3,781) in these schools were invited to participate in the survey, which included anthropometric examination, blood pressure, body composition, and a questionnaire. All the children were apparently healthy and did not use antihypertensive drugs. Body weight, height, and body composition were measured in 2014 (baseline), and blood pressure was repeatedly measured by trained medical staff in 2014, 2015 and 2016. After excluding those who declined to participate in the survey, those with missing data, and born preterm, 2671 children and adolescents (1264 girls) aged 6–14 years old was included in the cross-sectional analysis of the exposures (e.g., FFM%) and blood pressure (referred to as sample one). Participants who were excluded from the analysis were older, with a higher BMI, average systolic blood pressure, FFM, and FM, and lower FFM% than those remaining in the analysis (all p < 0.05), while sex proportion and average diastolic blood pressure were similar between the two groups. To examine the exposure and change in blood pressure during the follow-up, we excluded 226 children who were lost to follow-up; all the remaining 2445 children (1166 girls) in the analysis (referred to as sample two) were followed up for 2 years. To study the risk of incident hypertension, we further excluded 351 children with hypertension (either high systolic blood pressure and/or high diastolic blood pressure, according to the age- and sex-specific 95th blood pressure percentile for Chinese children) [21] at baseline, leaving 2094 children (1009 girls) for the analysis (referred to as sample three). The details of participant recruitment are shown in Supplementary Figure 1. Parents/caretakers signed informed consent forms. The study was approved by the Ethics Committee of Ren Ji Hospital, School of Medicine, Shanghai Jiao Tong University (PW2016D-05).
Anthropometrical data and body composition
Height (to the nearest 0.1 cm), weight (to the nearest 0.1 kg), and body composition (Bio-impedance analysis, BIA; TBF-410, Tokyo, Japan) were measured while the children were barefoot and in underwear. FM and FFM were recorded to the nearest 0.1 kg. FM% (FM% = fat mass/body weight*100), and FFM% (FFM% = free fat mass/body weight*100) was calculated and recorded to the nearest 0.1%.
Resting blood pressure
Resting blood pressure was measured by trained medical staff (e.g., doctors and nurses) in the school from 1 p.m. to 5 p.m. Resting blood pressure was measured twice on the children’s right arm after resting for at least 10 minutes using a mercury sphygmomanometer and a special cuff that fits 2/3 of the children’s arm. Systolic blood pressure is the point at which the onset of Korotkoff sounds and the fourth Korotkoff sound (K4) is used to define diastolic blood pressure [22]. The interval between the two BP measurements was at least 10 min and the average of two blood pressure measurements was recorded to the nearest 1 mm Hg. Hypertension was defined as either high systolic blood pressure and/or high diastolic blood pressure, according to the age- and sex-specific 95th blood pressure percentile for Chinese children [21].
Assessment of covariates
We collected information on age, sex, birth weight, infant feeding pattern (breast feeding, part breast feeding, bottle feeding), physical activity (<1 h or ≥1 h per day), carbonated beverages (≤4 or ≥5 times per week), western fast food (≤4 or ≥5 times per month), traditional Chinese fried food (≤3 or ≥4 times per week), processed meat (≤4 or ≥5 times per week), and time of sleep (<9 h or ≥9 h per day) by the questionnaire. We also collected parental height, body weight, and the highest education level. Parental BMI was also calculated. Parents completed the questionnaire.
Statistical analyses
We completed all statistical analyses by SAS version 9.4 (SAS Institute, Inc, Cary, NC). Formal hypothesis testing was two-sided with a significance level of 0.05.
We performed partial correlation analysis between the body weight, BMI, and FM, FM%, FFM, FFM%, FFM-to-FM ratio, and FFM index at baseline in sample one. We used the generalized linear model (GLM) to analyze the association between exposures (e.g., FFM%) and baseline blood pressure in sample one. We used “PROC GPLOT” to produce a two-dimensional graph to demonstrate the association between exposure (FFM%, FFM, FM%, FM) and blood pressure. To examine whether FFM% and FFM were associated with changes in blood pressure during follow-up, “PROC GENMOD” was used based on sample two. Finally, the Cox proportional hazards model was used to examine whether the exposures predicted the risk of incident hypertension in sample three. The person-time of follow-up for each child was determined from the finishing date of the baseline (01 June 2014) survey to either the date of HBP or the end of follow-up (30 June 2016), whichever came first. We adjusted for potential confounders in different models: Model 1, adjusted for sex and baseline age (y); Model 2, adjusted for sex, baseline height (cm), age (y), birth weight (g), infant feeding (breast feeding, part breast feeding, bottle feeding), consumption of western fast food (≤4 or ≥5 times per month), Chinese traditional fried food (≤3 or ≥4 times per week), carbonated beverages (≤4 or ≥5 times per week), and processed meat (≤4 or ≥5 times per week), physical activity (<1 h or ≥1 h per day), duration of sleep (<9 h or ≥9 h per day), paternal BMI and education (≤middle school or ≥high school), and maternal education (≤middle school or ≥high school). Furthermore, in a cross-sectional study, model 3 was adjusted for variables in model 2 and baseline body weight (kg); in prospective analyses, model 3 was adjusted for variables in model 2 and baseline systolic and diastolic blood pressure (mmHg); model 4 was adjusted for variables in model 3 and baseline body weight (kg).
We tested potential interactions among age, sex, and physical activity with the association of FFM% and baseline blood pressure. To test the robustness of the results obtained from the main analysis, we conducted three sensitivity analyses using the FFM-to-FM ratio and FFM index (calculated by FFM in kilogram divided by squared height in meter) as exposures and blood pressure at the end of follow-up as the outcome, respectively.
Results
Baseline characteristics
The baseline age was 9.2 ± 2.1 years. High FFM% was associated with a low proportional of girls, younger age, low height, body weight, BMI, systolic and diastolic blood pressure, birth weight, paternal and maternal BMI, and high physical activity (Table 1). By contrast, high FFM was associated with older age, high body weight, BMI, systolic and diastolic blood pressure, birth weight, parental BMI and education, and long sleep time, and low consumption of carbonated beverages, processed meat, western fast food, and traditional Chinese fried food (Supplementary Table 1).
Cross-sectional analyses
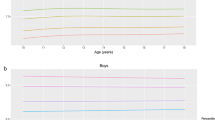
The baseline body weight and BMI were positively associated with FM, FM%, FFM, and the FFM index but was negatively associated with FFM% and the FFM-to-FM ratio (all p < 0.001; Supplementary Table 2). High FFM% was associated with low systolic [β = -0.42 mm Hg; 95% confident interval (CI): −0.49, −0.35 mm Hg] and diastolic (β = -0.29 mm Hg; 95% CI: −0.34, −0.23 mm Hg) blood pressure at the baseline (Fig. 1 and Table 2, model 2). By contrast, high FFM was associated with high systolic (β = 0.58 mm Hg, 95% CI: 0.46, 0.69 mm Hg) and diastolic (β = 0.33 mm Hg, 95% CI: 0.24, 0.42 mm Hg) blood pressure, after adjustment for potential confounders (Fig. 1 and Table 2) at the end of the follow-up (sample two). After further adjustment for body weight, the associations between FFM and systolic and diastolic blood pressure were largely attenuated and became insignificant for systolic blood pressure (Table 2, model 3).
Cross-sectional relationship between FFM and FFM% and systolic (a, c) and diastolic (b, d) blood pressure in 2671 Chinese children (sample one). a FFM and systolic blood pressure; b FFM and diastolic blood pressure; c FFM% and systolic blood pressure; d FFM% and diastolic blood pressure. FFM free fat mass, FFM% percentage of free fat mass = free fat mass (kg) / body weight (kg) × 100
Prospective analyses
High FFM% was also associated with a slow increase in blood pressure over time and low hypertension risk in a fully adjusted model (both p trend < 0.01) (Fig. 2). Each additional percent of FFM% at the baseline was associated with a 9% low likelihood to develop hypertension (HR = 0.91; 95% CI: 0.89, 0.94) (Table 3). By contrast, high FFM was associated with a fast increase in blood pressure and high hypertension risk (HR = 1.12; 95% CI: 1.08, 1.16) in the model (Fig. 2 and Table 3). High FM and FM% were consistently associated with a high baseline blood pressure and the risk of developing hypertension (Supplementary Figure 2 and Tables 2, 3). The inclusion of the baseline blood pressure in the Cox model did not change the pattern between the body composition and incidence of hypertension (Table 3, Supplementary Tables 3, 4).
Mean (95% confidence interval) annual changes in systolic and diastolic blood pressure during the follow-up based on the baseline status of FFM and FFM% (sample two). a FFM and systolic blood pressure; b FFM and diastolic blood pressure; c FFM% and systolic blood pressure; and d, FFM% and diastolic blood pressure. FFM free fat mass; FFM% percentage of free fat mass = free fat mass (kg) / body weight (kg) × 100. Models were adjusted for sex, baseline height (cm), age (y), birth weight (g), infant feeding (breast feeding, part breast feeding, bottle feeding), consumption of western fast food (≤4 or ≥5 times per month), Chinese traditional fried food (≤3 or ≥4 times per week), carbonated beverages (≤4 or ≥5 times per week), and processed meat (≤4 or ≥5 times per week), physical activity (<1 h or ≥1 h per day), duration of sleep (<9 h or ≥9 h per day), paternal BMI and education (≤middle school or ≥high school), and maternal education (≤middle school or ≥high school)
Interaction and sensitivity analyses
We did not observe interactions between age, sex, and daily physical activity with the association of FFM% and blood pressure (all p > 0.05). Sensitivity analyses using the FFM-to-FM ratio as the exposure showed that a high ratio was associated with a low baseline blood pressure and low hypertension risk after adjustment for potential confounders (Supplementary Table 3). We also observed that a high FFM index was associated with a high baseline blood pressure and high hypertension risk (Supplementary Table 4). Replacing baseline blood pressure with blood pressure at the end of follow-up as the outcome showed similar results (Supplementary Table 5).
Discussion
In this community-based study including 2,671 healthy Chinese children, we observed that high FFM% was associated with low blood pressure and a low risk of developing hypertension during 2 years of follow-up, after adjustment for potential confounders such as birth weight, infant feeding, diet, physical activity, and parental factors, which are possibly related with childhood hypertension. However, high FFM was associated with a high blood pressure and high risk of hypertension. The differences indicate that FFM%, and not the absolute value of FFM, is an optimal index to use for obesity and hypertension studies.
Although age- and sex-specific BMI is the most widely used index to assess childhood overweight and obesity, it failed to identify over one-quarter of children with excess body fat [23]. Some studies have reported that waist circumference is better than BMI in predicting childhood hypertension [24, 25], but it still cannot distinguish subcutaneous fat from visceral fat [26]. BIA provides a cheap, convenient and relatively precise alternative to estimate body fat in children [27, 28], and body fat has been suggested as a novel parameter for obesity and its related cardiovascular diseases in children [29]. The positive association between FFM and blood pressure was consistent with that in previous studies. Brion et al. [7] estimated the body composition by dual-energy X-ray absorptiometry (DXA) in 6,863 nine-year-old children and found that a high lean body mass was associated with high systolic blood pressure (β = 3.38 mmHg for each additional kg of lean body mass, 95% CI: 2.95, 3.81 mmHg) in a fully adjusted model. A high lean body mass index was also correlated with high blood pressure in a study including 733 Indian children aged 10-18 years (r = 0.23; p < 0.01) [14]. In another cross-sectional study including 3,004 adolescents (12–20 years) in the National Health and Nutrition Examination Survey, a high lean body mass index z-score was associated high odds of having hypertension (OR = 1.8; 95% CI: 1.1–2.9) [16]. However, the observed discrepancy relationship in these studies could be confounded by body weight [17, 18], which was strongly associated with both FFM and blood pressure. In the current study, further adjustment for body weight attenuated the association between FFM and blood pressure. This suggests that the reported positive FFM-hypertension relationship could be, at least in part, interpreted by the existence of residual confounders. Another possible explanation was that FFM increased simultaneously with the increase in FM [30], and the latter was shown to be closely associated with hypertension [29].
We observed that high FFM% was associated with a low blood pressure and low risk of developing hypertension. This was consistent with the notion that FFM%, not absolute FFM, was a better predictor for hypertension and other cardiovascular diseases [19]. A small observational study, including 94 children aged 6–18 years with hypertension found a markedly lower lean body mass percentage z-score (an index strongly correlated with FFM%) in participants associated with hypertension, compared with 562 non-hypertension controls [20]. In this study, lean body mass z-scores tended to be slightly higher in boys and girls than in controls, but this observation was significant in boys (p < 0.0001) [20]. This observed gender difference should be interpreted with caution because of the difference in sample size between boys (n = 73) and girls (n = 21) in this study. Interestingly, another case-control study found that the absolute lean body mass was higher (20.8 vs. 17.8 kg, p = 0.002) while the percentage of lean body mass (76.8 vs. 79.9%, p = 0.024) was lower in girls with premature adrenarche compared with healthy controls [31]. Similarly, in a study including 18,687 Korean adults (aged 20-79 years), a lower FFM% and higher FFM at the baseline were significantly associated with the risk of developing diabetes during 4.3 years of follow-up [32]. These results, together with our study, suggest that FFM% or the ratio between FFM and FM, rather than absolute FFM, could better reflect the obesity status and predict health status.
Our study could be limited by residual confounding, although we adjusted for many potential confounders. For example, we did not collect information on salt intake, one of the major dietary determinants of hypertension risk. However, it was difficult to estimate salt intake using dietary records while 24 h urinary sodium excretion as a biomarker of salt intake was not applicable in the population study [33]. Similarly, we did not collect Tanner development of children and adolescents, and puberty was marked by rapid growth and increases in both FFM and FM [34]. However, we conducted a subgroup analysis stratified by age (6–9 years vs. 10-14 years) and did not observe interaction of age with the association between FFM% and hypertension risk. Furthermore, we did not collect information on secondary smoking and parental history of hypertension, which were reported as potential risk factors for pediatric hypertension [35, 36]. However, parental BMI, which was closely associated with the pediatric blood pressure, was adjusted in the final model. Inclusion of parental BMI in the analysis attenuated the association between FFM% and SBP, DBP, and the risk of developing hypertension slightly; however, they remained significant (data not shown). Another limitation was the short follow-up period. Thus, we cannot exclude the possibility that the observed association between FFM% and hypertension risk could be due to reverse causation. Possible selection bias should be considered because there were significant differences in some baseline characteristics between children who were excluded from the study and those that remained in the study. Finally, it was worth noting that BIA had some measurement errors. Previous studies reported BIA could overestimate FFM compared with DXA, and it was not suitable in severely obese adolescents [37, 38]. However, excluding 10 participants with morbid obesity [39] did not materially change the results (data not shown). Furthermore, a good correlation was confirmed in FFM derived by foot-to-foot BIA and DXA (r = 0.95 in boys and 0.96 in girls) in Asian children [40].
Conclusion
High FFM% was associated with a low baseline blood pressure, slow increase in blood pressure over time, and low hypertension risk in this community-based cohort of Chinese children and adolescents. Although we observed a positive association between high FFM and high blood pressure, the association could be largely explained by body weight. Further study with a longer follow-up duration and detailed assessment of dietary data are warranted to replicate our findings.
References
Kumar S, Kelly AS. Review of childhood obesity: from epidemiology, etiology, and comorbidities to clinical assessment and treatment. Mayo Clin Proc. 2017;92:251–65.
Rao G. Diagnosis, epidemiology, and management of hypertension in children. Pediatrics. 2016;138:e20153616.
Xu T, Liu J, Liu J, Zhu G, Han S. Relation between metabolic syndrome and body compositions among Chinese adolescents and adults from a large-scale population survey. BMC Public Health. 2017;17:337.
Brady TM. Obesity-related hypertension in children. Front Pediatr. 2017;5:197.
Gontarev S, Kalac R, Zivkovic V, Velickovska LA, Telai B. The association between high blood pressure, physical fitness and fatness in adolescents. Nutr Hosp. 2017;34:35–40.
Vaziri Y, Bulduk S, Shadman Z, Bulduk EO, Hedayati M, Koc H, et al. Lean Body mass as a predictive value of hypertension in young adults, in Ankara, Turkey. Iran J Public Health. 2015;44:1643–54.
Brion MA, Ness AR, Davey Smith G, Leary SD. Association between body composition and blood pressure in a contemporary cohort of 9-year-old children. J Hum Hypertens. 2007;21:283–90.
Sidoti A, Nigrelli S, Rosati A, Bigazzi R, Caprioli R, Fanelli R, et al. Body mass index, fat free mass, uric acid, and renal function as blood pressure levels determinants in young adults. Nephrol (Carlton). 2017;22:279–85.
Wilks RJ, McFarlane-Anderson N, Bennett FI, Reid M, Forrester TE. Blood pressure in Jamaican children: relationship to body size and composition. West Indian Med J. 1999;48:61–68.
Janz KF, Burns TL, Mahoney LT. Predictors of left ventricular mass and resting blood pressure in children: the Muscatine Study. Med Sci Sports Exerc. 1995;27:818–25.
Latka M, Wojtowicz K, Drozdz T, Dąbrowska E, Kwinta P, Pietrzyk JA, et al. Relationship between water compartments, body composition assessed by bioelectrical impedance analysis and blood pressure in school children. Przegl Lek. 2016;73:1–5.
Zhang YX, Wang SR. [Relationship between body composition index and blood pressure among children aged 7-12]. Zhonghua Liu Xing Bing Xue Za Zhi. 2010;31:1135–8.
Gaskin PS, Hall RV, Chami P, St John MA, Gaskin DA, Molaodi OR, et al. Associations of blood pressure with body composition among Afro-Caribbean children in Barbados. PLoS ONE. 2015;10:e0121107.
Verma V, Sinah SK. Influence of lean body-mass index versus that of fat mass index on blood pressure of gujarati school going adolescents. Indian J Physiol Pharmacol. 2014;58:39–44.
Duncan MJ, James L, Griffiths L. The relationship between resting blood pressure, body mass index and lean body mass index in British children. Ann Hum Biol. 2011;38:324–9.
Weber DR, Leonard MB, Shults J, Zemel BS. A comparison of fat and lean body mass index to BMI for the identification of metabolic syndrome in children and adolescents. J Clin Endocrinol Metab. 2014;99:3208–16.
Wells JC, Fewtrell MS, Williams JE, Haroun D, Lawson MS, Cole TJ. Body composition in normal weight, overweight and obese children: matched case-control analyses of total and regional tissue masses, and body composition trends in relation to relative weight. Int J Obes (Lond). 2006;30:1506–13.
Al-Shorman A, Al-Domi H, Al-Atoum M. The associations of body composition and anthropometric measures with carotid intima-media thickness in obese and non-obese schoolchildren: A possible predictor for cardiovascular diseases. Vascular. 2017;26:285–90.
Carbone S, Lavie CJ, Arena R. Obesity and heart failure: focus on the obesity paradox. Mayo Clin Proc. 2017;92:266–79.
Pludowski P, Litwin M, Sladowska J, Antoniewicz J, Niemirska A, Wierzbicka A, et al. Bone mass and body composition in children and adolescents with primary hypertension: preliminary data. Hypertension. 2008;51:77–83.
Mi J, Wang TY, Meng LH, Zhu GJ, Han SM, Zhong Y, et al. Development of blood pressure reference standards for Chinese children and adolescents. Chin J Evid Based Pediatr. 2010;5:4–14.
Update on the 1987 Task Force Report on High Blood Pressure in Children and Adolescents: a working group report from the National High Blood Pressure Education Program. National High Blood Pressure Education Program Working Group on Hypertension Control in Children and Adolescents. Pediatrics. 1996;98(4 Pt 1):649–58.
Javed A, Jumean M, Murad MH, Okorodudu D, Kumar S, Somers VK, et al. Diagnostic performance of body mass index to identify obesity as defined by body adiposity in children and adolescents: a systematic review and meta-analysis. Pediatr Obes. 2015;10:234–44.
Tebar WR, Ritti-Dias RM, Farah BQ, Zanuto EF, Vanderlei LCM, Christofaro DGD. High blood pressure and its relationship to adiposity in a school-aged population: body mass index vs waist circumference. Hypertens Res. 2018;41:135–40.
Krzesinski P, Stanczyk A, Piotrowicz K, Gielerak G, Uzieblo-Zyczkowska B, Skrobowski A. Abdominal obesity and hypertension: a double burden to the heart. Hypertens Res. 2016;39:349–55.
Bertoli S, Leone A, Vignati L, Spadafranca A, Bedogni G, Vanzulli A, et al. Metabolic correlates of subcutaneous and visceral abdominal fat measured by ultrasonography: a comparison with waist circumference. Nutr J. 2016;15:2.
McCarthy HD, Cole TJ, Fry T, Jebb SA, Prentice AM. Body fat reference curves for children. Int J Obes. 2006;30:598–602.
Kasvis P, Cohen TR, Loiselle SE, Kim N, Hazell TJ, Vanstone CA, et al. Foot-to-foot bioelectrical impedance accurately tracks direction of adiposity change in overweight and obese 7- to 13-year-old children. Nutr Res. 2015;35:206–13.
Dias IB, Panazzolo DG, Marques MF, Paredes BD, Souza MG, Manhanini DP, et al. Relationships between emerging cardiovascular risk factors, z-BMI, waist circumference and body adiposity index (BAI) on adolescents. Clin Endocrinol. 2013;79:667–74.
Zuo JLZQ, Liu AL, Zhao J, Hu XQ. The analysis of body composition in children and adolescents in Beijing. Chin Health Educ. 2011;27:415–8.
Cebeci AN, Tas A. Higher body fat and lower fat-free mass in girls with premature adrenarche. J Clin Res Pediatr Endocrinol. 2015;7:45–48.
Kim CH, Kim HK, Kim EH, Bae SJ, Park JY. Association between changes in body composition and risk of developing Type 2 diabetes in Koreans. Diabet Med. 2014;31:1393–8.
Shi L, Krupp D, Remer T. Salt, fruit and vegetable consumption and blood pressure development: a longitudinal investigation in healthy children. Br J Nutr. 2014;111:662–71.
Cheng HL, Amatoury M, Steinbeck K. Energy expenditure and intake during puberty in healthy nonobese adolescents: a systematic review. Am J Clin Nutr. 2016;104:1061–74.
Bloetzer C, Paccaud F, Burnier M, Bovet P, Chiolero A. Performance of parental history for the targeted screening of hypertension in children. J Hypertens. 2015;33:1167–73.
Hunt KJ, Hansis-Diarte A, Shipman K, Korte JE, Fowler SP, Stern MP. Impact of parental smoking on diabetes, hypertension and the metabolic syndrome in adult men and women in the San Antonio Heart Study. Diabetologia. 2006;49:2291–8.
Wan CS, Ward LC, Halim J, Gow ML, Ho M, Briody JN, et al. Bioelectrical impedance analysis to estimate body composition, and change in adiposity, in overweight and obese adolescents: comparison with dual-energy x-ray absorptiometry. BMC Pediatr. 2014;14:249.
Verney J, Metz L, Chaplais E, Cardenoux C, Pereira B, Thivel D. Bioelectrical impedance is an accurate method to assess body composition in obese but not severely obese adolescents. Nutr Res. 2016;36:663–70.
Bervoets L, Massa G. Defining morbid obesity in children based on BMI 40 at age 18 using the extended international (IOTF) cut-offs. Pediatr Obes. 2014;9:e94–98.
Sim PY, Su TT, Abd Majid H, Nahar AM, Jalaludin MY. A comparison study of portable foot-to-foot bioelectrical impedance scale to measure body fat percentage in Asian adults and children. Biomed Res Int. 2014;2014:475659.
Acknowledgements
The study was supported by grants from Pu Dong Medical Bureau (PW2016D-05).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Xu, R., Zhang, X., Zhou, Y. et al. Percentage of free fat mass is associated with elevated blood pressure in healthy Chinese children. Hypertens Res 42, 95–104 (2019). https://doi.org/10.1038/s41440-018-0129-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-018-0129-y
Key words
This article is cited by
-
Body composition predicts hypertension using machine learning methods: a cohort study
Scientific Reports (2023)
-
Association between body composition and blood pressure in normal-weight Chinese children and adolescents
BMC Pediatrics (2022)
-
A novel indicator, childhood lipid accumulation product, is associated with hypertension in Chinese children and adolescents
Hypertension Research (2020)