Abstract
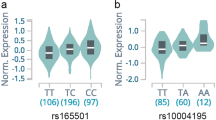
Emerging evidence revealed that thyroglobulin (TG) contributes to the development of autoimmune disease, and the relationship between TG and autoimmune thyroid disease (AITD) is still controversial. The aim of this study was to quantify the association between rs2076740, rs853326, rs180223, and rs2069550 TG polymorphisms and risk of AITD using a meta-analysis approach. We identified all studies that assessed the association between TG polymorphisms and AITD from PubMed, Embase, and Web of Science databases. A total of 3013 cases and 1812 controls from ten case–control studies were included. There was no significant associations found between rs2069550, rs180223, and rs853326 polymorphisms and AITD risk. The association between the rs2076740 polymorphism and AITD risk was significant in the codominant model (P = 0.005), suggesting the CC rs2076740 genotype might be a protective factor for AITD. Sensitivity analysis by removing one or two study changed the results in dominant rs2076740 and rs853326 and rs2069550 allele models (P = 0.016, 0.024, 0.027). Latitude and ethnicity significantly affected the association between rs2076740 and rs2069550 polymorphisms and AITD, indicating their protective effects in allele or dominant model (P = 0.012, 0.012, 0.012, 0.009, 0.009). The association between rs2076740, rs2069550, and rs853326 polymorphisms and AITD risk is significantly affected by study characteristics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wang B, Shao X, Song R, Xu D, Zhang JA. The emerging role of epigenetics in autoimmune thyroid diseases. Front Immunol. 2017;8:396.
Coppede F. Epigenetics and autoimmune thyroid diseases. Front Endocrinol (Lausanne). 2017;8:149.
Brown RS. Autoimmune thyroid disease: unlocking a complex puzzle. Curr Opin Pediatr. 2009;21:523–8.
Ferrari SM, Fallahi P, Antonelli A, Benvenga S. Environmental issues in thyroid diseases. Front Endocrinol (Lausanne). 2017;8:50.
Mizuma T, Watanabe M, Inoue N, Arakawa Y, Tomari Tomari, Hidaka Y. et al. Association of the polymorphisms in the gene encoding thyroglobulin with the development and prognosis of autoimmune thyroid disease. Autoimmunity. 2017;50:386–92.
Balazs C. The role of hereditary and environmental factors in autoimmune thyroid diseases. Orv Hetil. 2012;153:1013–22.
Wiersinga WM. Clinical relevance of environmental factors in the pathogenesis of autoimmune thyroid disease. Endocrinol Metab (Seoul). 2016;31:213–22.
Guarneri F, Benvenga S. Environmental factors and genetic background that interact to cause autoimmune thyroid disease. Curr Opin Endocrinol Diabetes Obes. 2007;14:398–409.
Kus A, Arlukowicz-Grabowska M, Szymanski K, Wunsch E, Milkiewicz M, Ploski R, et al. Genetic risk factors for autoimmune thyroid disease might affect the susceptibility to and modulate the progression of primary biliary cholangitis. J Gastrointestin Liver Dis. 2017;26:245–52.
Tomer Y. Genetic susceptibility to autoimmune thyroid disease: past, present, and future. Thyroid. 2010;20:715–25.
Ramgopal S, Rathika C, Padma MR, Murali V, Arun K, Kamaludeen MN, et al. Interaction of HLA-DRB1* alleles and CTLA4 (+49 AG) gene polymorphism in autoimmune thyroid disease. Gene. 2017;642:430–8.
Xiaoheng C, Yizhou M, Bei H, Huilong L, Xin W, Rui H, et al. General and specific genetic polymorphism of cytokines-related gene in AITD. Mediat Inflamm. 2017;2017:3916395.
Ting WH, Chien MN, Lo FS, Wang CH, Huang CY, Lin CL, et al. Association of cytotoxic T-lymphocyte-associated protein 4 (CTLA4) gene polymorphisms with autoimmune thyroid disease in children and adults: case–control study. PLoS ONE. 2016;11:e0154394.
Ban Y, Tozaki T, Taniyama M, Skrabanek L, Nakano Y, Ban Y, et al. Multiple SNPs in intron 41 of thyroglobulin gene are associated with autoimmune thyroid disease in the Japanese population. PLoS ONE. 2012;7:e37501.
Rivolta CM, Targovnik HM. Molecular advances in thyroglobulin disorders. Clin Chim Acta. 2006;374:8–24.
Kong YM, Brown NK, Morris GP, Flynn JC. The essential role of circulating thyroglobulin in maintaining dominance of natural regulatory T cell function to prevent autoimmune thyroiditis. Horm Metab Res. 2015;47:711–20.
Gough S. The thyroglobulin gene: the third locus for autoimmune thyroid disease or a false dawn? Trends Mol Med. 2004;10:302–5.
Collins JE, Heward JM, Howson JM, Foxall H, Carr-Smith J, Franklyn JA, et al. Common allelic variants of exons 10, 12, and 33 of the thyroglobulin gene are not associated with autoimmune thyroid disease in the United Kingdom. J Clin Endocrinol Metab. 2004;89:6336–9.
Wang LQ, Wang TY, Sun QL, Qie YQ. Correlation between thyroglobulin gene polymorphisms and autoimmune thyroid disease. Mol Med Rep. 2015;12:4469–75.
Ueda H, Howson JM, Esposito L, Heward J, Snook H, Chamberlain G, et al. Association of the T-cell regulatory gene CTLA4 with susceptibility to autoimmune disease. Nature. 2003;423:506–11.
Jacobson EM, Concepcion E, Ho K, Kopp P, Vono Toniolo J, Tomer Y. cDNA immunization of mice with human thyroglobulin generates both humoral and T cell responses: a novel model of thyroid autoimmunity. PLoS ONE. 2011;6:e19200.
Kolypetri P, Noel NA, Carayanniotis KA, Carayanniotis G. Iodine content of thyroglobulin in Nod.H2h4 mice developing iodine-accelerated autoimmune thyroiditis. Hormones (Athens). 2010;9:151–60.
Belguith-Maalej S, Hadj Kacem H, Rebai A, Mnif M, Abid M, Ayadi H. Thyroglobulin polymorphisms in Tunisian patients with autoimmune thyroid diseases (AITD). Immunobiology. 2008;213:577–83.
Patel H, Mansuri MS, Singh M, Begum R, Shastri M, Misra A. Association of cytotoxic T-lymphocyte antigen 4 (CTLA4) and thyroglobulin (TG) genetic variants with autoimmune hypothyroidism. PLoS ONE. 2016;11:e0149441.
Kotnik P, Debeljak M, Avbelj M, Hovnik T, Ursic Bratina N, Krzisnik C, et al. Lack of association of common allelic variants in the thyroglobulin gene with Hashimoto’s thyroiditis in young subjects with type 1 diabetes. Horm Res Paediatr. 2010;73:244–7.
Gu LQ, Zhu W, Zhao SX, Zhao L, Zhang MJ, Cui B, et al. Clinical associations of the genetic variants of CTLA4, Tg, TSHR, PTPN22, PTPN12 and FCRL3 in patients with Graves’ disease. Clin Endocrinol (Oxf). 2010;72:248–55.
Maierhaba M, Zhang JA, Yu ZY, Wang Y, Xiao WX, Quan Y, et al. Association of the thyroglobulin gene polymorphism with autoimmune thyroid disease in Chinese population. Endocrine. 2008;33:294–9.
Hsiao JY, Tien KJ, Hsiao CT, Hsieh MC. Exon 33 T/T genotype of the thyroglobulin gene is a susceptibility gene for Graves’ disease in Taiwanese and exon 12 C/C genotype protects against it. Clin Exp Med. 2008;8:17–21.
Ban Y, et al. Association of a thyroglobulin gene polymorphism with Hashimoto’s thyroiditis in the Japanese population. Clin Endocrinol (Oxf). 2004;61:263–8.
Tomer Y, Greenberg D. The thyroglobulin gene as the first thyroid-specific susceptibility gene for autoimmune thyroid disease. Trends Mol Med. 2004;10:306–8.
Ban Y, Tozaki T, Taniyama M, Tomita M, Ban Y. Amino acid substitutions in the thyroglobulin gene are associated with susceptibility to human and murine autoimmune thyroid disease. Proc Natl Acad Sci USA. 2003;100:15119–24.
Stefan M, Jacobson EM, Huber AK, Greenberg DA, Li CW, Skrabanek L, et al. Novel variant of thyroglobulin promoter triggers thyroid autoimmunity through an epigenetic interferon alpha-modulated mechanism. J Biol Chem. 2011;286:31168–79.
Brent GA. Environmental exposures and autoimmune thyroid disease. Thyroid. 2010;20:755–61.
Martinez FD. Gene-environment interaction in complex diseases: asthma as an illustrative case. Novartis Found Symp. 2008;293:184–92. discussion192–7
Ioannidis JP, Ntzani EE, Trikalinos TA, Contopoulos-Ioannidis DG. Replication validity of genetic association studies. Nat Genet. 2001;29:306–9.
Manji N, Carr-Smith JD, Boelaert K, Allahabadia A, Armitage M, Chatterjee VK, et al. Influences of age, gender, smoking, and family history on autoimmune thyroid disease phenotype. J Clin Endocrinol Metab. 2006;91:4873–80.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097.
Thakkinstian A, McElduff P, D’Este C, Duffy D, Attia J. A method for meta-analysis of molecular association studies. Stat Med. 2005;24:1291–306.
Tizaoui K, Kaabachi W, Hamzaoui A, Hamzaoui K. Association between vitamin D receptor polymorphisms and multiple sclerosis: systematic review and meta-analysis of case–control studies. Cell Mol Immunol. 2015;12:243–52.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (no. 31570357).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, ML., Zhang, Dm., Wang, CE. et al. Association between thyroglobulin polymorphisms and autoimmune thyroid disease: a systematic review and meta-analysis of case–control studies. Genes Immun 20, 484–492 (2019). https://doi.org/10.1038/s41435-018-0042-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41435-018-0042-z
This article is cited by
-
Newborn screening and single nucleotide variation profiling of TSHR, TPO, TG and DUOX2 candidate genes for congenital hypothyroidism
Molecular Biology Reports (2020)