Abstract
Background:
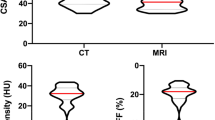
Muscle mass and density assessed from CT-images at the L3 level are prognostic for survival and predict toxicity in cancer patients. However, L3 is not always included on routine CT-scans. We aimed to investigate whether images at the Th4 level may be used instead.
Methods:
Patients from three chemotherapy trials in advanced NSCLC were eligible (n = 1305). Skeletal muscle area (cm2), skeletal muscle index (SMI, cm2/m2) and skeletal muscle density (SMD) at Th4 and L3 levels were assessed from baseline CT-scans. SMI and SMD at the Th4 and L3 level were transformed into z-scores and the agreement between scores was investigated by Bland–Altman plots and estimated by intra-class correlation analyses. Linear regression was used to test if Th4 SMI and SMD z-scores predicted L3 SMI and SMD z-scores.
Results:
CT-images from 401 patients were analysable at both levels. There was a moderate agreement between Th4 and L3 SMI z-scores with an intra-class correlation of 0.71 (95% CI 0.64–0.77) for men and 0.53 (95% CI 0.41–0.63) for women. Regression models predicting L3 SMI z-scores from Th4 SMI z-scores showed coefficients of 0.71 (95% CI 0.62–0.80) among men and 0.53 (95% CI 0.40–0.66) among women. R-squares were 0.51 and 0.28, respectively, indicating moderate agreement. A similar, moderate agreement between Th4 and L3 SMD z-scores was observed.
Conclusion:
There was only moderate agreement between muscle measures from Th4 and L3 levels, indicating that missing data from the L3 level cannot be replaced by analysing images at the Th4 level.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Muhlberg W, Sieber C. Sarcopenia and frailty in geriatric patients: implications for training and prevention. Z Gerontol Geriatr. 2004;37:2–8. https://doi.org/10.1007/s00391-004-0203-8.
Landi F, Cruz-Jentoft AJ, Liperoti R, Russo A, Giovannini S, Tosato M, et al. Sarcopenia and mortality risk in frail older persons aged 80 years and older: results from ilSIRENTE study. Age Ageing. 2013;42:203–9. https://doi.org/10.1093/ageing/afs194.
Baracos V, Kazemi-Bajestani SM. Clinical outcomes related to muscle mass in humans with cancer and catabolic illnesses. Int J Biochem Cell Biol. 2013;45:2302–8. https://doi.org/10.1016/j.biocel.2013.06.016.
Prado CM, Lieffers JR, McCargar LJ, Reiman T, Sawyer MB, Martin L, et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol. 2008;9:629–35. https://doi.org/10.1016/s1470-2045(08)70153-0.
Tan BH, Birdsell LA, Martin L, Baracos VE, Fearon KC. Sarcopenia in an overweight or obese patient is an adverse prognostic factor in pancreatic cancer. Clin Cancer Res. 2009;15:6973–9. https://doi.org/10.1158/1078-0432.ccr-09-1525.
van Vledder MG, Levolger S, Ayez N, Verhoef C, Tran TC, Ijzermans JN. Body composition and outcome in patients undergoing resection of colorectal liver metastases. Br J Surg. 2012;99:550–7. https://doi.org/10.1002/bjs.7823.
Martin L, Birdsell L, Macdonald N, Reiman T, Clandinin MT, McCargar LJ, et al. Cancer cachexia in the age of obesity: skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J Clin Oncol. 2013;31:1539–47. https://doi.org/10.1200/jco.2012.45.2722.
Prado CM, Baracos VE, McCargar LJ, Mourtzakis M, Mulder KE, Reiman T, et al. Body composition as an independent determinant of 5-fluorouracil-based chemotherapy toxicity. Clin Cancer Res. 2007;13:3264–8. https://doi.org/10.1158/1078-0432.ccr-06-3067.
Prado CM, Baracos VE, McCargar LJ, Reiman T, Mourtzakis M, Tonkin K, et al. Sarcopenia as a determinant of chemotherapy toxicity and time to tumor progression in metastatic breast cancer patients receiving capecitabine treatment. Clin Cancer Res. 2009;15:2920–6. https://doi.org/10.1158/1078-0432.ccr-08-2242.
Barret M, Antoun S, Dalban C, Malka D, Mansourbakht T, Zaanan A, et al. Sarcopenia is linked to treatment toxicity in patients with metastatic colorectal cancer. Nutr Cancer. 2014;66:583–9. https://doi.org/10.1080/01635581.2014.894103.
Cousin S, Hollebecque A, Koscielny S, Mir O, Varga A, Baracos VE, et al. Low skeletal muscle is associated with toxicity in patients included in phase I trials. Invest New Drugs. 2014;32:382–7. https://doi.org/10.1007/s10637-013-0053-6.
Sjoblom B, Gronberg BH, Benth JS, Baracos VE, Flotten O, Hjermstad MJ, et al. Low muscle mass is associated with chemotherapy-induced haematological toxicity in advanced non-small cell lung cancer. Lung Cancer. 2015;90:85–91. https://doi.org/10.1016/j.lungcan.2015.07.001.
Lieffers JR, Bathe OF, Fassbender K, Winget M, Baracos VE. Sarcopenia is associated with postoperative infection and delayed recovery from colorectal cancer resection surgery. Br J Cancer. 2012;107:931–6. https://doi.org/10.1038/bjc.2012.350.
Romero-Corral A, Somers VK, Sierra-Johnson J, Thomas RJ, Collazo-Clavell ML, Korinek J. et al.Accuracy of body mass index in diagnosing obesity in the adult general population.Int. J. Obes.2008;32(6):959–966.https://doi.org/10.1038/ijo.2008.11.
Mourtzakis M, Prado CM, Lieffers JR, Reiman T, McCargar LJ, Baracos VE. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl Physiol Nutr Metab. 2008;33:997–1006. https://doi.org/10.1139/h08-075.
Shen W, Punyanitya M, Wang Z, Gallagher D, St-Onge MP, Albu J, et al. Total body skeletal muscle and adipose tissue volumes: estimation from a single abdominal cross-sectional image. J Appl Physiol. 2004;97:2333–8. https://doi.org/10.1152/japplphysiol.00744.2004.
Cesari M, Fielding RA, Pahor M, Goodpaster B, Hellerstein M, van Kan GA. et al. Biomarkers of sarcopenia in clinical trials-recommendations from the International Working Group on Sarcopenia. J Cachex Sarcopenia Muscle. 2012;3(3):181–190. https://doi.org/10.1007/s13539-012-0078-2.
Aubrey J, Esfandiari N, Baracos VE, Buteau FA, Frenette J, Putman CT, et al. Measurement of skeletal muscle radiation attenuation and basis of its biological variation. Acta Physiol. 2014;210:489–97. https://doi.org/10.1111/apha.12224.
Anderson DE, D’Agostino JM, Bruno AG, Demissie S, Kiel DP, Bouxsein ML. Variations of CT-based trunk muscle attenuation by age, sex, and specific muscle. J Gerontol A Biol Sci Med Sci. 2013;68:317–23. https://doi.org/10.1093/gerona/gls168.
Esfandiari N, Ghosh S, Prado CM, Martin L, Mazurak V, Baracos VEAge. Obesity, sarcopenia, and proximity to death explain reduced mean muscle attenuation in patients with advanced cancer. J Frailty Aging. 2014;3:3–8. https://doi.org/10.14283/jfa.2014.1.
Sjoblom B, Gronberg BH, Wentzel-Larsen T, Baracos VE, Hjermstad MJ, Aass N et al. Skeletal muscle radiodensity is prognostic for survival in patients with advanced non-small cell lung cancer. Clin Nutr. 2016; https://doi.org/10.1016/j.clnu.2016.03.010
Baracos VE, Reiman T, Mourtzakis M, Gioulbasanis I, Antoun S. Body composition in patients with non-small cell lung cancer: a contemporary view of cancer cachexia with the use of computed tomography image analysis. Am J Clin Nutr. 2010;91:1133s–7s. https://doi.org/10.3945/ajcn.2010.28608C.
Wieland BM, Stewart GD, Skipworth RJ, Sangster K, Fearon KC, Ross JA. et al. Is there a human homologue to the murine proteolysis-inducing factor?. Clin Cancer Res. 2007;13(17):4984–4992. 10.1158/1078-0432.ccr-07-0946.
Go SI, Park MJ, Song HN, Kang MH, Park HJ, Jeon KN, et al. Sarcopenia and inflammation are independent predictors of survival in male patients newly diagnosed with small cell lung cancer. Support Care Cancer. 2016;24:2075–84. https://doi.org/10.1007/s00520-015-2997-x.
Kim EY, Kim YS, Park I, Ahn HK, Cho EK, Jeong YM, et al. Evaluation of sarcopenia in small-cell lung cancer patients by routine chest CT. Support Care Cancer. 2016;24:4721–6. https://doi.org/10.1007/s00520-016-3321-0.
Helbekkmo N, Sundstrom SH, Aasebo U, Brunsvig PF, von Plessen C, Hjelde HH, et al. Vinorelbine/carboplatin vs gemcitabine/carboplatin in advanced NSCLC shows similar efficacy, but different impact of toxicity. Br J Cancer. 2007;97:283–9. https://doi.org/10.1038/sj.bjc.6603869.
Gronberg BH, Bremnes RM, Flotten O, Amundsen T, Brunsvig PF, Hjelde HH, et al. Phase III study by the Norwegian lung cancer study group: pemetrexed plus carboplatin compared with gemcitabine plus carboplatin as first-line chemotherapy in advanced non-small-cell lung cancer. J Clin Oncol. 2009;27:3217–24. https://doi.org/10.1200/jco.2008.20.9114.
Flotten O, Gronberg BH, Bremnes R, Amundsen T, Sundstrom S, Rolke H, et al. Vinorelbine and gemcitabine vs vinorelbine and carboplatin as first-line treatment of advanced NSCLC. A phase III randomised controlled trial by the Norwegian Lung Cancer Study Group. Br J Cancer. 2012;107:442–7. https://doi.org/10.1038/bjc.2012.284.
Shen W, Chen J, Gantz M, Velasquez G, Punyanitya M, Heymsfield SB. A single MRI slice does not accurately predict visceral and subcutaneous adipose tissue changes during weight loss. Obesity. 2012;20:2458–63.
Acknowledgements
We want to thank Rachel Murphy (PhD) and Nina Esfandiari (B.Sc) both at Department of Oncology, University of Alberta, Canada, for their participation in the body composition analyses. Thanks to Nina Helbekkmo and Ingrid Sandstad for handling the collection of CT scans from the RCT1. We also thank the Departments of Radiology at Haukeland University Hospital, Bergen, St Olav University Hospital, Trondheim and Hamar, Gjøvik and Kongsvinger Hospital, Innlandet Hospital HF, as well as the following and their respective Departments of Radiology for their participation in the collection of CT scans: Bjørn Wembstad, Hammerfest Hospital and Kåre Hansen, Kirkenes Hospital; Helse Finnmark HF. Nina Helbekkmo, University Hospital North Norway Tromsø and Nada Zafran Groh, University Hospital North Norway Harstad; University Hospital North Norway HF. Heinrich Backmann, Nordland Hospital Bodø and Finn Larsen Aas, Nordland Hospital Vesterålen; Nordland Hospital HF. Kristina Helander, Helgeland Hospital Mo i Rana, Hans Henrik Strøm, Helgeland Hospital Sandnessjøen, Reidar Berntsen Helgeland, Hospital Mosjøen; Helgeland Hospital HF. Randi Sudbø, Namsos Hospital and Thor Naustdal, Levanger Hospital; Nord Trøndelag HF. Bjørn Jakobsen, Molde Hospital, Finn Wammer, Ålesund Hospital, Ivar Blix, Kristiansund Hospital and Inge Eskeland, Volda Hospital; Helse Møre og Romsdal HF. Anita Spikkeland, Voss Hospital; Helse Bergen HF. Tesfaye Madebo and Oddveig Garpestad, Stavanger University Hospital HF, Sverre Fluge, Haugesund Hospital; Helse Fonna HF. Frode Ramslien, Telemark Hospital Skien and Oddvar Øygarden, Telemark Hospital Rjukan; Telemark Hospital HF. Heidi Rolke Sørlandet Hospital Kristiansand and Terje Torp, Sørlandet Hospital Arendal; Sørlandet Hospital HF. Karin Semb, Vestfold Hospital Tønsberg; Vestfold Hospital HF. Per Fredrik Ekholdt, Østfold Hospital Fredrikstad; Østfold Hospital HF. Ellinor Heitman, Ringerike Hospital, Leiv Rusten, Drammen Hospital, Martin Cornelius Ruppert, Bærum Hospital, and Peter Gottschalk, Kongsberg Hospital; Vestre Viken HF. Anders Fjeld, Oslo University Hospital (OUS) Aker, Odd Terje Brustugun and Paal Fr Brunsvig, OUS Radiumhospitalet, Kjersti Hornslien and Frøydis Stornes, OUS Ullevål; OUS HF. Svein Olav Saxrud, Akershus University Hospital HF. Carl Birger Alm, Lovisenberg Hospital Oslo.
Funding:
The study was funded by the South-Eastern Norway Regional Health Authority. The collection of CT scans was supported by unrestricted grants from Pierre Fabre, Norway. The Canadian participation in the body composition analyses was supported by the Canadian Institute of Health Research and Alberta Cancer Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Grønberg, B.H., Sjøblom, B., Wentzel-Larsen, T. et al. A comparison of CT based measures of skeletal muscle mass and density from the Th4 and L3 levels in patients with advanced non-small-cell lung cancer. Eur J Clin Nutr 73, 1069–1076 (2019). https://doi.org/10.1038/s41430-018-0325-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-018-0325-5
This article is cited by
-
Comparison of CT derived body composition at the thoracic T4 and T12 with lumbar L3 vertebral levels and their utility in patients with rectal cancer
BMC Cancer (2023)
-
Prediction of abdominal CT body composition parameters by thoracic measurements as a new approach to detect sarcopenia in a COVID-19 cohort
Scientific Reports (2022)
-
Prognostic value of myosteatosis in patients with lung cancer: a systematic review and meta-analysis
International Journal of Clinical Oncology (2022)