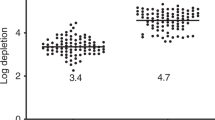
Abstract
Posttransplant relapsed B-cell precursor ALL can be cured by 2nd hematopoietic stem cell transplantation (HSCT) in 20% of patients. The major cause of death after second HSCT is leukemic relapse. One reliable predictor for survival after 2nd-HSCT are posttransplant MRD levels. Patients with detectable or increase of MRD are likely to relapse. Patients in complete molecular remission show the best leukemia-free survival and lowest cumulative incidence (CI) of relapse. As patients who undergo second or subsequent HSCT are high-risk patients, we evaluated the prophylactic use of the chimeric Fc-optimized CD19-4G7SDIE-mAb. Posttransplant relapsed CD19+ BCP-ALL patients, who underwent a second or subsequent haplo-HSCT from a T- and B-cell depleted graft received posttransplant prophylactic CD19-4G7SDIE-mAb treatment on compassionate use in complete molecular remission, to increase the antileukemic activity of the new reconstituting immune system by recruiting Fc-expressing effector cells. NK cells recovered early and robust. The 3 year overall survival in 15 evaluable patients was 56%, the 3 year event-free survival was 55% and the CI of relapse 38%. Compared to a historical control group, the CI of relapse was markedly lower and consecutively the EFS higher. Posttransplant-targeted therapy may overcome the need for unspecific GvL effect of undesired GvHD, that can cause severe morbidity and mortality. Due to a low adverse event profile the CD19-4G7SDIE-mAb may be suitable for broad administration to consolidate posttransplant MRD negativity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Kato M, Horikoshi Y, Okamoto Y, Takahashi Y, Hasegawa D, Koh K, et al. Second allogeneic hematopoietic SCT for relapsed ALL in children. Bone Marrow Transplant. 2012;47:1307–11.
Schlegel P, Lang P, Zugmaier G, Ebinger M, Kreyenberg H, Witte K-E, et al. Pediatric posttransplant relapsed/refractory B-precursor acute lymphoblastic leukemia shows durable remission by therapy with the T-cell engaging bispecific antibody blinatumomab. Haematologica. 2014;99:1212–9.
Bader P, Kreyenberg H, Stackelberg Av, Eckert C, Salzmann-Manrique E, Meisel R, et al. Monitoring of minimal residual disease after allogeneic stem-cell transplantation in relapsed childhood acute lymphoblastic leukemia allows for the identification of impending relapse: results of the ALL-BFM-SCT 2003 trial. J Clin Oncol. 2015;33:1275–84.
Lovisa F, Zecca M, Rossi B, Campeggio M, Magrin E, Giarin E, et al. Pre- and post-transplant minimal residual disease predicts relapse occurrence in children with acute lymphoblastic leukaemia. Br J Haematol. 2018;180:680–93.
Shaw BE. Related haploidentical donors are a better choice than matched unrelated donors: counterpoint. Blood Adv. 2017;1:401–6.
Locatelli F, Zecca M, Messina C, Rondelli R, Lanino E, Sacchi N, et al. Improvement over time in outcome for children with acute lymphoblastic leukemia in second remission given hematopoietic stem cell transplantation from unrelated donors. Leukemia. 2002;16:2228–37.
Zhang P, Chen BJ, Chao NJ. Prevention of GVHD without losing GVL effect: windows of opportunity. Immunol Res. 2011;49:49–55.
Dickinson AM, Norden J, Li S, Hromadnikova I, Schmid C, Schmetzer H, et al. Graft-versus-leukemia effect following hematopoietic stem cell transplantation for leukemia. Front Immunol. 2017;8:496.
Holtan SG, DeFor TE, Lazaryan A, Bejanyan N, Arora M, Brunstein CG, et al. Composite end point of graft-versus-host disease-free, relapse-free survival after allogeneic hematopoietic cell transplantation. Blood. 2015;125:1333–8.
Seidel UJE, Schlegel P, Grosse-Hovest L, Hofmann M, Aulwurm S, Pyz E, et al. Reduction of minimal residual disease in pediatric b-lineage acute lymphoblastic leukemia by an Fc-optimized CD19 antibody. Mol Ther: J Am Soc Gene Ther. 2016;24:1634–43.
Katz BZ, Herishanu Y. Therapeutic targeting of CD19 in hematological malignancies: past, present, future and beyond. Leuk Lymphoma. 2014;55:999–1006.
Seidel UJE, Schlegel P, Grosse-Hovest L, Hofmann M, Aulwurm S, Pyz E, et al. Reduction of minimal residual disease in pediatric b-lineage acute lymphoblastic leukemia by an Fc-optimized CD19 antibody. Mol Ther 2016;24:1634–43.
Bader P, Kreyenberg H, Henze GH, Eckert C, Reising M, Willasch A, et al. Prognostic value of minimal residual disease quantification before allogeneic stem-cell transplantation in relapsed childhood acute lymphoblastic leukemia: the ALL-REZ BFM Study Group. J Clin Oncol. 2009;27:377–84.
Lang P, Teltschik HM, Feuchtinger T, Muller I, Pfeiffer M, Schumm M, et al. Transplantation of CD3/CD19 depleted allografts from haploidentical family donors in paediatric leukaemia. Br J Haematol. 2014;165:688–98.
Kuhlen M, Willasch AM, Dalle JH, Wachowiak J, Yaniv I, Ifversen M, et al. Outcome of relapse after allogeneic HSCT in children with ALL enrolled in the ALL-SCT 2003/2007 trial. Br J Haematol. 2018;180:82–9.
Yaniv I, Krauss AC, Beohou E, Dalissier A, Corbacioglu S, Zecca M, et al. Second hematopoietic stem cell transplantation for post-transplantation relapsed acute leukemia in children: a retrospective EBMT-PDWP Study. Biol Blood Marrow Transplant: J Am Soc Blood Marrow Transplant. 2018;24:1629–42.
Oevermann L, Handgretinger R. New strategies for haploidentical transplantation. Pediatr Res. 2012;71(4 Pt 2):418–26.
Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. New Engl J Med. 2014;371:1507–17.
Acknowledgements
We acknowledge financial support by the Deutsche Forschungsgemeinschaft (DFG), CRC685 Immunotherapy, the Bundesministerium für Bildung und Forschung (BMBF iVac ALL, BMBF GO-Bio 0315096/0316070), the Reinhold Beitlich Stiftung, the Deutsche José Carreras Leukämie-Stiftung, the Stefan Morsch Stiftung, the Förderverein für krebskranke Kinder Tübingen, the Stiftung des Fördervereins für krebskranke Kinder e.V. Tübingen, the Elterninitiative Kinderkrebsklinik e.V.Düsseldorf, Hilfe für krebskranke Kinder Frankfurt e.V., the German Cancer Consortium (DKTK) and the German Cancer Research Center (DKFZ) Heidelberg, Germany. Publication of this supplement was sponsored by Gilead Sciences Europe Ltd, Cell Source, Inc., The Chorafas Institute for Scientific Exchange of the Weizmann Institute of Science, Kiadis Pharma, Miltenyi Biotec, Celgene, Centro Servizi Congressuali, Almog Diagnostic.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Schlegel, P., Jung, G., Lang, AM. et al. ADCC can improve graft vs leukemia effect after T- and B-cell depleted haploidentical stem cell transplantation in pediatric B-lineage ALL. Bone Marrow Transplant 54 (Suppl 2), 689–693 (2019). https://doi.org/10.1038/s41409-019-0606-1
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-019-0606-1