Abstract
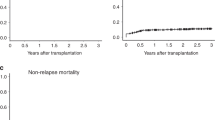
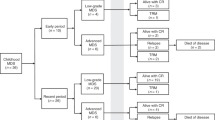
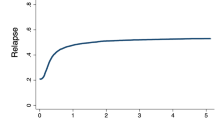
Allocating patients with acute myeloid leukemia and high-risk cytogenetic abnormalities (HR-AML) for allogeneic hematopoietic stem cell transplantation (allo-HSCT) is part of the standard treatment protocol; however, whether allo-HSCT truly improves the outcomes in these patients is debatable. Data on 169 children and adolescents with HR-AML who received their first allo-HSCT in first or second remission between 2000 and 2015 were extracted from a nationwide, Japanese HSCT registry. The 3-year disease-free survival (DFS) and overall survival (OS) rates were 55.2% (95% CI, 46.8–62.9%) and 69.6% (61.4–76.3%), respectively, for all the HR-AML patients. In univariate analysis, the cytogenetic subgroup had a significant impact on both the DFS (P = 0.011) and OS (P < 0.001) rates. In particular, 14 patients with t(16;21) showed an extremely poor outcome. Additionally, older age at allo-HSCT (10–19 years old, P = 0.025), myeloablative conditioning with total-body irradiation (P = 0.019), and grade II–IV acute graft-versus-host disease (GVHD, P = 0.049) were associated with inferior OS. The donor type and occurrence of chronic GVHD did not affect the outcome. Multivariate analysis revealed t(16;21) to be associated with increased overall mortality (hazard ratio = 4.416, P < 0.001). Because the outcome of patients with certain HR-AML subgroups, such as t(16;21)-positive cases, is extremely poor even with allo-HSCT in remission, a novel therapy is urgently required.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Woods WG, Neudorf S, Gold S, Sanders J, Buckley JD, Barnard DR, et al. A comparison of allogeneic bone marrow transplantation, autologous bone marrow transplantation, and aggressive chemotherapy in children with acute myeloid leukemia in remission. Blood. 2001;97:56–62.
Niewerth D, Creutzig U, Bierings MB, Kaspers GJL. A review on allogeneic stem cell transplantation for newly diagnosed pediatric acute myeloid leukemia. Blood. 2010;116:2205–14.
Tsukimoto I, Tawa A, Horibe K, Tabuchi K, Kigasawa H, Tsuchida M, et al. Risk-stratified therapy and the intensive use of cytarabine improves the outcome in childhood acute myeloid leukemia: the AML99 trial from the Japanese Childhood AML Cooperative Study Group. J Clin Oncol. 2009;27:4007–13.
Tomizawa D, Tawa A, Watanabe T, Saito AM, Kudo K, Taga T, et al. Appropriate dose reduction in induction therapy is essential for the treatment of infants with acute myeloid leukemia: a report from the Japanese Pediatric Leukemia/Lymphoma Study Group. Int J Hematol. 2013;98:578–88.
Rubnitz JE, Inaba H, Dahl G, Ribeiro RC, Bowman WP, Taub J, et al. Minimal residual disease-directed therapy for childhood acute myeloid leukaemia: results of the AML02 multicentre trial. Lancet Oncol. 2010;11:543–52.
Gamis AS, Alonzo TA, Meshinchi S, Sung L, Gerbing RB, Raimondi SC, et al. Gemtuzumab ozogamicin in children and adolescents with de novo acute myeloid leukemia improves event-free survival by reducing relapse risk: results from the randomized phase III Children’s Oncology Group trial AAML0531. J Clin Oncol. 2014;32:3021–32.
Creutzig U, Zimmermann M, Bourquin J-P, Dworzak MN, Fleischhack G, Graf N, et al. Randomized trial comparing liposomal daunorubicin with idarubicin as induction for pediatric acute myeloid leukemia: results from Study AML-BFM 2004. Blood. 2013;122:37–43.
Locatelli F, Masetti R, Rondelli R, Zecca M, Fagioli F, Rovelli A, et al. Outcome of children with high-risk acute myeloid leukemia given autologous or allogeneic hematopoietic cell transplantation in the aieop AML-2002/01 study. Bone Marrow Transplant. 2015;50:181–8.
Hasle H, Abrahamsson J, Forestier E, Ha S-Y, Heldrup J, Jahnukainen K, et al. Gemtuzumab ozogamicin as postconsolidation therapy does not prevent relapse in children with AML: results from NOPHO-AML 2004. Blood. 2012;120:978–84.
Gibson BES, Webb DKH, Howman AJ, de Graaf SSN, Harrison CJ, Wheatley K, et al. Results of a randomized trial in children with Acute Myeloid Leukaemia: medical research council AML12 trial. Br J Haematol. 2011;155:366–76.
Horan JT, Alonzo TA, Lyman GH, Gerbing RB, Lange BJ, Ravindranath Y, et al. Impact of disease risk on efficacy of matched related bone marrow transplantation for pediatric acute myeloid leukemia: the Children’s Oncology Group. J Clin Oncol. 2008;26:5797–801.
Kelly MJ, Horan JT, Alonzo TA, Eapen M, Gerbing RB, He W, et al. Comparable survival for pediatric acute myeloid leukemia with poor-risk cytogenetics following chemotherapy, matched related donor, or unrelated donor transplantation. Pediatr Blood Cancer. 2014;61:269–75.
Alloin A-L, Leverger G, Dalle J-H, Galambrun C, Bertrand Y, Baruchel A, et al. Cytogenetics and outcome of allogeneic transplantation in first remission of acute myeloid leukemia: the French pediatric experience. Bone Marrow Transplant. 2017;52:516–21.
Creutzig U, van den Heuvel-Eibrink MM, Gibson B, Dworzak MN, Adachi S, de Bont E, et al. Diagnosis and management of acute myeloid leukemia in children and adolescents: recommendations from an international expert panel. Blood. 2012;120:3187–205.
Taga T, Tomizawa D, Takahashi H, Adachi S. Acute myeloid leukemia in children: current status and future directions. Pediatr Int. 2016;58:71–80.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant. 2009;15:1628–33.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Stevens RF, Hann IM, Wheatley K, Gray RG. Marked improvements in outcome with chemotherapy alone in paediatric acute myeloid leukemia: results of the United Kingdom Medical Research Council’s 10th AML trial. MRC Childhood Leukaemia Working Party. Br J Haematol. 1998;101:130–40.
Kong XT, Ida K, Ichikawa H, Shimizu K, Ohki M, Maseki N, et al. Consistent detection of TLS/FUS-ERG chimeric transcripts in acute myeloid leukemia with t(16;21)(p11; q22) and identification of a novel transcript. Blood. 1997;90:1192–9.
Kawashima N, Shimada A, Taketani T, Hayashi Y, Yoshida N, Matsumoto K, et al. Childhood acute myeloid leukemia with bone marrow eosinophilia caused by t(16;21)(q24; q22). Int J Hematol. 2012;95:577–80.
Noort S, Zimmermann M, Reinhardt D, Cuccuini W, Martina P, Alonzo TA, et al. Distinct outcome based on fusion transcript in t(16;21) rearranged pediatric AML, a collaborative retrospective analysis by the I-BFM SG. Blood. 2017;130:184a.
Hollink IHIM, van den Heuvel-Eibrink MM, Zimmermann M, Balgobind BV, Arentsen-Peters STCJM, Alders M, et al. Clinical relevance of Wilms tumor 1 gene mutations in childhood acute myeloid leukemia. Blood. 2009;113:5951–60.
Meshinchi S, Alonzo TA, Stirewalt DL, Zwaan M, Zimmerman M, Reinhardt D, et al. Clinical implications of FLT3 mutations in pediatric AML. Blood. 2006;108:3654–61.
Leung W, Pui C-H, Coustan-Smith E, Yang J, Pei D, Gan K, et al. Detectable minimal residual disease before hematopoietic cell transplantation is prognostic but does not preclude cure for children with very-high-risk leukemia. Blood. 2012;120:468–72.
Karol SE, Coustan-Smith E, Cao X, Shurtleff SA, Raimondi SC, Choi JK, et al. Prognostic factors in children with acute myeloid leukaemia and excellent response to remission induction therapy. Br J Haematol. 2015;168:94–101.
Acknowledgements
The authors thank all the members of the hospitals and centers who contributed valuable data to the JSHCT registry. The authors also thank Mr. JR Valera of the Department of Education for Clinical Research, National Center for Child Health and Development, for his assistance in editing this manuscript. This work was supported by a grant for Practical Research for Innovative Cancer Control from the Japan Agency for Medical Research and Development (AMED) and a grant from the National Center for Child Health and Development (30-1).
Author contributions
DT, MY, TK, TM, TT, SA, and HI participated actively in the study conception and design; DT conducted the data analysis and interpretation and was the main author of the study; KK, MN, HK, KW, YC, TF, MK, NS, HG, JH, and MI provided patient data; MI, YH, YA, and HI contributed to the administrative support of the study; and all the authors contributed to carrying out the study, were involved in the review of the results, and gave their final approval of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Tomizawa, D., Yoshida, M., Kondo, T. et al. Allogeneic hematopoietic stem cell transplantation for children and adolescents with high-risk cytogenetic AML: distinctly poor outcomes of FUS-ERG-positive cases. Bone Marrow Transplant 54, 393–401 (2019). https://doi.org/10.1038/s41409-018-0273-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-018-0273-7
This article is cited by
-
FUS-ERG induces late-onset azacitidine resistance in acute myeloid leukaemia cells
Scientific Reports (2023)
-
TLS/FUS-ERG fusion gene in acute leukemia and myelodysplastic syndrome evolved to acute leukemia: report of six cases and a literature review
Annals of Hematology (2022)
-
Alternative donor peripheral blood stem cell transplantation for the treatment of high-risk refractory and/or relapsed childhood acute leukemia: a randomized trial
Experimental Hematology & Oncology (2020)