Abstract
Background
MRI-US fusion prostate biopsies are becoming a common procedure to diagnose prostate cancer. There is a paucity of information regarding the learning curve for fusion biopsies. We aim to study the amount of experience needed to be both accurate and time-efficient in this procedure.
Methods
We prospectively collected data on all MRI-US fusion biopsies performed from April 2014 to August 2017. We used two parameters to define the learning curve. Process Measurement (efficiency) was measured by time from the beginning of anesthesia to end of procedure. Outcome Measurement (accuracy) was measured by cancer detection rate for PI-RAD 3 lesions. The end of the learning curve was defined graphically and mathematically. We performed a separate analysis for transrectal and transperineal biopsies.
Results
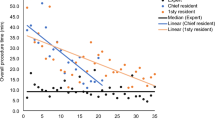
We completed 779 fusion biopsies (523 transrectal, 256 transperineal). Patients median age was 66 years (IQR 61–70) and median PSA 6.95 ng/ml (IQR 4.2–10.6). Prostate cancer was diagnosed in 385 (49%). Process Measurement—Procedure time decreased from 45 min in the first transrectal fusion biopsy to 15 min after 109 biopsies and remained stable (p < 0.0001). Time decreased from 55 min in the first transperineal biopsy to 18 min after 124 biopsies (p < 0.0001). Outcome Measurement—In transrectal fusion-biopsies detection rate for PI-RADS 3 lesions increased from 35 to 50% after 104 biopsies. In transperineal fusion-biopsies, detection rate increased from 40 to 55% after 119 cases for PI-RADS 3 lesions.
Conclusions
We measured the learning curve of fusion biopsies graphically and mathematically. We demonstrated that proficiency occurs after 110 transrectal and 125 transperineal fusion-biopsies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Siddiqui MM, Rais-Bahrami S, Turkbey B, George AK, Rothwax J, Shakir N. et al. Comparison of MR/Ultrasound Fusion–guided biopsy with ultrasound-guided biopsy for the diagnosis of prostate cancer. J Am Med Assoc. 2015;313:390
Porpiglia F, Manfredi M, Mele F, Cossu M, Bollito E, Veltri A, et al. Diagnostic pathway with multiparametric magnetic resonance imaging versus standard pathway: results from a randomized prospective study in biopsy-naïve patients with suspected prostate cancer. Eur Urol. 2017;72:282–8.
Schoots IG, Roobol MJ, Nieboer D, Bangma CH, Steyerberg EW, Hunink MGM. Magnetic resonance imaging-targeted biopsy may enhance the diagnostic accuracy of significant prostate cancer detection compared to standard transrectal ultrasound-guided biopsy: a systematic review and meta-analysis. Eur Urol. 2015;68:438–50.
Baco E, Rud E, Eri LM, Moen G, Vlatkovic L, Svindland A, et al. A randomized controlled trial to assess and compare the outcomes of two-core prostate biopsy guided by fused magnetic resonance and transrectal ultrasound images and traditional 12-core systematic biopsy. Eur Urol. 2016;69:149–56.
Ahmed HU, El-Shater Bosaily A, Brown LC, Gabe R, Kaplan R, Parmar MK, et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet. 2017;389:815–22.
Kasivisvanathan V, Rannikko AS, Borghi M, Panebianco V, Mynderse LA, Vaarala MH, et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med. 2018;378:1767–77.
Barentsz JO, Richenberg J, Clements R, Choyke P, Verma S, Villeirs G, et al. ESUR prostate MR guidelines 2012. Eur Radiol. 2012;22:746–57.
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, et al. PI-RADS prostate imaging—reporting and data system: 2015, Version 2. Eur Urol. 2016;69:16–40.
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA, et al. The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. Am J Surg Pathol. 2015;40:1.
Muller BG, Shih JH, Sankineni S, Marko J, Rais-Bahrami S, George AK, et al. Prostate cancer: interobserver agreement and accuracy with the revised prostate imaging reporting and data system at multiparametric MR imaging. Radiology. 2015;277:741–50.
Mertan FV, Greer MD, Shih JH, George AK, Kongnyuy M, Muthigi A, et al. Prospective Evaluation of the prostate imaging reporting and data system version 2 for prostate cancer detection. J Urol. 2016;196:690–6.
Gaziev G, Wadhwa K, Barrett T, Koo BC, Gallagher FA, Serrao E, et al. Defining the learning curve for multiparametric magnetic resonance imaging (MRI) of the prostate using MRI-transrectal ultrasonography (TRUS) fusion-guided transperineal prostate biopsies as a validation tool. BJU Int. 2016;117:80–6.
Meng X, Rosenkrantz AB, Huang R, Deng FM, Wysock JS, Bjurlin M et al. The institutional learning curve for MRI-US Fusion-Targeted Prostate Biopsy: temporal improvements in cancer detection over four years. J Urol. 2018. https://doi.org/10.1016/j.juro.2018.06.012.
Calio B, Sidana A, Sugano D, Gaur S, Jain A, Maruf M, et al. Changes in prostate cancer detection rate of MRI-TRUS fusion vs systematic biopsy over time: evidence of a learning curve. Prostate Cancer Prostatic Dis. 2017;20:436–41.
Khan N, Abboudi H, Khan MS, Dasgupta P, Ahmed K. Measuring the surgical ‘learning curve’: methods, variables and competency. BJU Int. 2014;113:504–8.
Subramonian K, Muir G. The ‘learning curve’ in surgery: what is it, how do we measure it and can we influence it? BJU Int. 2004;93:1173–4.
Akin O, Riedl CC, Ishill NM, Moskowitz CS, Zhang J, Hricak H. Interactive dedicated training curriculum improves accuracy in the interpretation of MR imaging of prostate cancer. Eur Radiol. 2010;20:995–1002.
Rosenkrantz AB, Ayoola A, Hoffman D, Khasgiwala A, Prabhu V, Smereka P, et al. The Learning curve in prostate MRI interpretation: self-directed learning versus continual reader feedback. AJR Am J Roentgenol. 2017;208:W92–W100.
Loeb S, Carter HB, Berndt SI, Ricker W, Schaeffer EM. Complications after prostate biopsy: data from SEER-medicare. J Urol. 2011;186:1830–4.
Rodríguez LV, Terris MK. Risks and complications of transrectal ultrasound guided prostate needle biopsy: a prospective study and review of the literature. J Urol. 1998;160:2115–20.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Halstuch, D., Baniel, J., Lifshitz, D. et al. Characterizing the learning curve of MRI-US fusion prostate biopsies. Prostate Cancer Prostatic Dis 22, 546–551 (2019). https://doi.org/10.1038/s41391-019-0137-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-019-0137-2
This article is cited by
-
The learning curve for robotic-assisted transperineal MRI/US fusion-guided prostate biopsy
Scientific Reports (2024)
-
Prostate cancer detection and complications of MRI-targeted prostate biopsy using cognitive registration, software-assisted image fusion or in-bore guidance: a systematic review and meta-analysis of comparative studies
Prostate Cancer and Prostatic Diseases (2024)
-
Expect the unexpected: investigating discordant prostate MRI and biopsy results
European Radiology (2024)
-
The design and validation of a low-cost trans perineal (TP) prostate biopsy simulator for training: improving trainees’ confidence and cognitive targeting skills
World Journal of Urology (2023)
-
Impact of operator expertise on transperineal free-hand mpMRI-fusion-targeted biopsies under local anaesthesia for prostate cancer diagnosis: a multicenter prospective learning curve
World Journal of Urology (2023)