Abstract
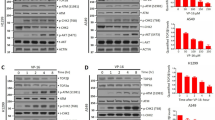
Cancer chemotherapeutic agents such as doxorubicin are DNA damage inducers that also kill normal cells, making them highly toxic to cancer patients. To improve the efficacy and safety of chemotherapy, it is important to develop new chemotherapeutic agents that selectively kill cancer cells. Here we demonstrate that artemisitene (ATT), a natural derivative of the antimalarial drug artemisinin, selectively induces DNA double-stranded breaks (DSBs) and apoptosis in various human cancer cells by suppressing the expression of topoisomerases in human cancer cells. ATT effectively kills human cancer cells without apparent cytotoxicity on normal human cells or mouse liver and kidney. We discovered that c-Myc induces the expression of topoisomerases to prevent accumulation of DNA damage in human cancer cells. ATT selectively destabilizes c-Myc in human cancer cells by promoting the ubiquitination of c-Myc through the specific induction of the c-Myc E3 ligase NEDD4. Therefore, ATT represents a promising new chemotherapeutic drug candidate that can eliminate human cancer cells with minimized cytotoxic effects on normal cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, et al. Cancer statistics in China, 2015. CA A Cancer J Clin. 2016;66:115–32.
Bhandari A, Woodhouse M, Gupta S. Colorectal cancer is a leading cause of cancer incidence and mortality among adults younger than 50 years in the USA: a SEER-based analysis with comparison to other young-onset cancers. J Invest Med. 2017;65:311–5.
Liu Q, Chen Z, Jiang G, Zhou Y, Yang X, Huang H, et al. Epigenetic down regulation of G protein-coupled estrogen receptor (GPER) functions as a tumor suppressor in colorectal cancer. Mol Cancer. 2017;16:87.
Singh A, Sweeney MF, Yu M, Burger A, Greninger P, Benes C, et al. TAK1 inhibition promotes apoptosis in KRAS-dependent colon cancers. Cell. 2012;148:639–50.
Nitiss JL. Targeting DNA topoisomerase II in cancer chemotherapy. Nat Rev Cancer. 2009;9:338–50.
Zhang P, Wang H, Rowe PSN, Hu B, Wang Y. MEPE/OF45 as a new target for sensitizing human tumour cells to DNA damage inducers. Br J Cancer. 2010;102:862–6.
Bollimpelli VS, Dholaniya PS, Kondapi AK. Topoisomerase IIbeta and its role in different biological contexts. Arch Biochem Biophys. 2017;633:78–84.
Cuya SM, Bjornsti MA, van Waardenburg R. DNA topoisomerase-targeting chemotherapeutics: what’s new? Cancer Chemother Pharmacol. 2017;80:1–14.
Rubin EH. DNA topoisomerase expression in tumors--a novel target for chemotherapy. Human Pathol. 2000;31:631–2.
Jadaun A, Subbarao N, Dixit A. Allosteric inhibition of topoisomerase I by pinostrobin: molecular docking, spectroscopic and topoisomerase I activity studies. J Photochem Photobiol B. 2017;167:299–308.
Cowell IG, Sondka Z, Smith K, Lee KC, Manville CM, Sidorczuk-Lesthuruge M, et al. Model for MLL translocations in therapy-related leukemia involving topoisomerase IIbeta-mediated DNA strand breaks and gene proximity. Proc Natl Acad Sci USA. 2012;109:8989–94.
Lyu YL, Kerrigan JE, Lin CP, Azarova AM, Tsai YC, Ban Y, et al. Topoisomerase IIbeta mediated DNA double-strand breaks: implications in doxorubicin cardiotoxicity and prevention by dexrazoxane. Cancer Res. 2007;67:8839–46.
Gilbert DC, Chalmers AJ, El-Khamisy SF. Topoisomerase I inhibition in colorectal cancer: biomarkers and therapeutic targets. Br J Cancer. 2012;106:18–24.
Hsiang YH, Hertzberg R, Hecht S, Liu LF. Camptothecin induces protein-linked DNA breaks via mammalian DNA topoisomerase I. J Biol Chem. 1985;260:14873–8.
Kim EJ, Kim SY, Kim SM, Lee M. A novel topoisomerase 2a inhibitor, cryptotanshinone, suppresses the growth of PC3 cells without apparent cytotoxicity. Toxicol Appl Pharmacol. 2017;330:84–92.
Roca J, Ishida R, Berger JM, Andoh T, Wang JC. Antitumor bisdioxopiperazines inhibit yeast DNA topoisomerase II by trapping the enzyme in the form of a closed protein clamp. Proc Natl Acad Sci USA. 1994;91:1781–5.
Damiani RM, Moura DJ, Viau CM, Caceres RA, Henriques JAP, Saffi J. Pathways of cardiac toxicity: comparison between chemotherapeutic drugs doxorubicin and mitoxantrone. Arch Toxicol. 2016;90:2063–76.
Matsumoto Y, Tamiya T, Nagao S. Resistance to topoisomerase II inhibitors in human glioma cell lines overexpressing multidrug resistant associated protein (MRP) 2. J Med Investig. 2005;52:41–48.
Azarova AM, Lyu YL, Lin CP, Tsai YC, Lau JYN, Wang JC, et al. Roles of DNA topoisomerase II isozymes in chemotherapy and secondary malignancies. Proc Natl Acad Sci USA. 2007;104:11014–9.
Ezoe S. Secondary leukemia associated with the anti-cancer agent, etoposide, a topoisomerase II inhibitor. Int J Environ Res Public Health. 2012;9:2444–53.
Jiang J, Yang ES, Jiang G, Nowsheen S, Wang H, Wang T, et al. p53-dependent BRCA1 nuclear export controls cellular susceptibility to DNA damage. Cancer Res. 2011;71:5546–57.
Bandi S, Joseph B, Berishvili E, Singhania R, Wu YM, Cheng K, et al. Perturbations in ataxia telangiectasia mutant signaling pathways after drug-induced acute liver failure and their reversal during rescue of animals by cell therapy. Am J Pathol. 2011;178:161–74.
Polager S, Ginsberg D. p53 and E2f: partners in life and death. Nat Rev Cancer. 2009;9:738–48.
Singh S, Englander EW. Nuclear depletion of apurinic/apyrimidinic endonuclease 1 (Ape1/Ref-1) is an indicator of energy disruption in neurons. Free Radic Biol Med. 2012;53:1782–90.
Chumduri C, Gurumurthy RK, Zadora PK, Mi Y, Meyer TF. Chlamydia infection promotes host DNA damage and proliferation but impairs the DNA damage response. Cell Host Microbe. 2013;13:746–58.
Rogakou EP, Pilch DR, Orr AH, Ivanova VS, Bonner WM. DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J Biol Chem. 1998;273:5858–68.
Hsu E. The history of qing hao in the Chinese materia medica. Trans R Soc Trop Med Hyg. 2006;100:505–8.
Li T, Chen H, Wei N, Mei X, Zhang S, Liu DL, et al. Anti-inflammatory and immunomodulatory mechanisms of artemisinin on contact hypersensitivity. Int Immunopharmacol. 2012;12:144–50.
Choi WH. Novel Pharmacological activity of artesunate and artemisinin: their potential as anti-tubercular agents. J Clin Med. 2017;6:1–15.
Chong CM, Zheng W. Artemisinin protects human retinal pigment epithelial cells from hydrogen peroxide-induced oxidative damage through activation of ERK/CREB signaling. Redox Biol. 2016;9:50–56.
Slezakova S, Ruda-Kucerova J. Anticancer activity of artemisinin and its derivatives. Anticancer Res. 2017;37:5995–6003.
Posner GH, Northrop J, Paik IH, Borstnik K, Dolan P, Kensler TW, et al. New chemical and biological aspects of artemisinin-derived trioxane dimers. Bioorg Med Chem. 2002;10:227–32.
Efferth T, Herrmann F, Tahrani A, Wink M. Cytotoxic activity of secondary metabolites derived from Artemisia annua L. towards cancer cells in comparison to its designated active constituent artemisinin. Phytomedicine. 2011;18:959–69.
Ooko E, Saeed ME, Kadioglu O, Sarvi S, Colak M, Elmasaoudi K, et al. Artemisinin derivatives induce iron-dependent cell death (ferroptosis) in tumor cells. Phytomedicine. 2015;22:1045–54.
Chen WM, Li SS, Li JW, Zhou W, Wu SH, Xu SM, et al. Artemisitene activates the Nrf2-dependent antioxidant response and protects against bleomycin-induced lung injury. FASEB J. 2016;30:2500–10.
Pommier Y, Leo E, Zhang HL, Marchand C. DNA topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem Biol. 2010;17:421–33.
Thomas LR, Tansey WP. Proteolytic control of the oncoprotein transcription factor Myc. Adv Cancer Res. 2011;110:77–106.
Farrell AS, Sears RC. MYC degradation. Cold Spring Harb Perspect Med. 2014;4:1–14.
King B, Trimarchi T, Reavie L, Xu L, Mullenders J, Ntziachristos P, et al. The ubiquitin ligase FBXW7 modulates leukemia-initiating cell activity by regulating MYC stability. Cell. 2013;153:1552–66.
Chen Y, Zhou C, Ji W, Mei Z, Hu B, Zhang W, et al. ELL targets c-Myc for proteasomal degradation and suppresses tumour growth. Nat Commun. 2016;7:11057.
Hoffman B, Liebermann DA. The proto-oncogene c-myc and apoptosis. Oncogene. 1998;17:3351–7.
Kim J, Xu S, Xiong L, Yu L, Fu X, Xu Y. SALL4 promotes glycolysis and chromatin remodeling via modulating HP1α-Glut1 pathway. Oncogene. 2017;36:6472.
Lake BB, Fink J, Klemetsaune L, Fu X, Jeffers JR, Zambetti GP, et al. Context-dependent enhancement of induced pluripotent stem cell reprogramming by silencing puma. Stem Cells. 2012;30:888–97.
Xu S, Chen W, Xie Q, Xu Y. Obacunone activates the Nrf2-dependent antioxidant responses. Protein Cell. 2016;7:684–8.
Acknowledgements
Wenjuan Li was supported by a fellowship from Postdoctoral Science Foundation of China (2015M582371). This study was supported by NSFC (nos. 81430032, U1601222, 81502595), Guangdong Provincial Key Laboratory of Cancer Immunotherapy, Guangzhou Key Laboratory of Tumor Immunology Research, Guangdong Provincial Key Special Science and Technology Project (2015B020225004), Guangdong NSF Major basic research developmental project, and grants from California Institute for Regenerative Medicine.
Author contributions
J.C., W.L., K.C., K.J., S.X. performed experiments. J.C. and Y.X. designed the study and analyzed the data. Y.X. provided the administrative support. J.C., W.L., K.C., and Y.X. wrote the first draft of the manuscript and all authors participated in the revision of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chen, J., Li, W., Cui, K. et al. Artemisitene suppresses tumorigenesis by inducing DNA damage through deregulating c-Myc-topoisomerase pathway. Oncogene 37, 5079–5087 (2018). https://doi.org/10.1038/s41388-018-0331-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-018-0331-z
This article is cited by
-
Dimeric oxyberberine CT4-1 targets LINC02331 to induce cytotoxicity and inhibit chemoresistance via suppressing Wnt/β-catenin signaling in hepatocellular carcinoma
Archives of Toxicology (2023)
-
Anti-malarial drug: the emerging role of artemisinin and its derivatives in liver disease treatment
Chinese Medicine (2021)
-
The role of ubiquitination and deubiquitination in cancer metabolism
Molecular Cancer (2020)
-
Berberine chloride suppresses non-small cell lung cancer by deregulating Sin3A/TOP2B pathway in vitro and in vivo
Cancer Chemotherapy and Pharmacology (2020)
-
CAS9 is a genome mutator by directly disrupting DNA-PK dependent DNA repair pathway
Protein & Cell (2020)