Abstract
Ectopic pregnancy is the major cause of maternal morbidity and mortality in the first trimester of pregnancy. Tubal ectopic pregnancy (TEP) accounts for nearly 98% of all ectopic pregnancies. TEP is usually associated with salpingitis but the underlying mechanism in salpingitis leading to TEP remains unclear. Adrenomedullin (ADM) is a peptide hormone abundantly expressed in the fallopian tube with potent anti-inflammatory activities. Its expression peaks at the early luteal phase when the developing embryo is being transported through the fallopian tube. In the present study, we demonstrated reduced expression of ADM in fallopian tubes of patients with salpingitis and TEP. Using macrophages isolated from the fallopian tubes of these women, our data revealed that the salpingistis-associated ADM reduction contributed to aggravated pro-inflammatory responses of the tubal macrophages resulting in production of pro-inflammatory and pro-implantation cytokines IL-6 and IL-8. These cytokines activated the expression of implantation-associated molecules and Wnt signaling pathway predisposing the tubal epithelium to an adhesive and receptive state for embryo implantation. In conclusion, this study provided evidence for the role of ADM in the pathogenesis of TEP through regulating the functions of tubal macrophages.
Similar content being viewed by others
Introduction
Ectopic pregnancy (EP) is defined as implantation of an embryo outside the uterine cavity.1 It is a leading cause of maternal death in the first trimester with a prevalence of 1.3–2% of all pregnancies.2 Nearly 98% of EP are in the fallopian tube, known as tubal ectopic pregnancy (TEP).3 Tubal damage induced by pelvic inflammatory disease (PID), or more specifically salpingitis, is the leading epidemiologic risk factor for TEP.2 The exact mechanism leading to TEP remains unclear and there are no suitable animal models for TEP because the disease is rare in animals.4 Defective cilia beating or muscular contractions for embryo transport and pro-inflammatory environment in the fallopian tube caused by infection and/or smoking are suggested to be the pathogenic mechanisms for TEP.5
Adrenomedullin (ADM) is a member of the calcitonin/calcitonin-gene-related peptide (CGRP)/amylin peptide family.6,7 ADM exerts its biological activities by binding to its receptor, which is composed of two transmembrane components, namely the calcitonin-receptor-like receptor (CRLR) and the receptor-activity-modifying protein (RAMP).8 ADM is strongly expressed in the apical region of fallopian tube and its expression is modulated by steroids.9 The fallopian tubes of women having TEP expressed significantly less ADM.10 Since ADM stimulates ciliary beat frequency (CBF) and muscle contraction in the fallopian tube dose-dependently,9 ADM reduction in women having TEP has been suggested to impair embryo transport within the fallopian tube.9 As retention of blastocysts in mouse fallopian tube does not result in TEP,11 there may be other ADM insufficiency-induced concomitant alterations in the human fallopian tube causing TEP.
Extravasation of leukocytes together with enhanced expression of pro-inflammatory cytokines and chemokines occurs in the uterus during implantation.12 Similarly, women with TEP have high levels of pro-inflammatory mediators, such as interleukin (IL)-6, IL-8 and tumor necrosis factor (TNF-α), in serum and fallopian tube.13,14 The local inflammatory signals in the fallopian tube may create a permissive condition encouraging attachment of blastocyst onto the tubal epithelium15
While natural killer (NK) cells are the largest immune cell population in decidua of early eutopic pregnancy, macrophages are the predominant leukocyte population in the fallopian tube of TEP.16,17,18 A role of macrophages in the pathogenesis of TEP is supported by a doubling of the macrophage density at the tubal implantation site when compared to the distant part of the fallopian tube.19 Decidual macrophages may have a role in implantation.20 They produce implantation-associated pro-inflammatory cytokines IL-6 and IL-8.21,22,23 The roles of macrophages in tubal implantation remain largely unknown.
ADM is a well-known anti-inflammatory regulator in cultured cells and in inflammation-associated diseases.24,25 In the present study, we hypothesized that a reduced ADM expression in the fallopian tube triggers excessive macrophages-mediated inflammation inducing a tubal environment susceptible to embryo implantation.
Results
ADM expression in fallopian tubes
ADM expression was predominately localized to the tubal epithelium (Fig. 1a). A significant reduction of ADM protein (Fig. 1a and Supplementary Table 1) (p < 0.05) and mRNA (Fig. 1b) (p < 0.05) was observed in the TEP group. In fallopian tube with salpingitis, a well-known predisposing state of TEP,2 ADM expression was similarly reduced relative to the control group (Fig. 1a, b and Supplementary Table 1), consistent with the possibility that ADM insufficiency predisposed the fallopian tube to TEP.
ADM expression is decreased in fallopian tubes with salpingitis and TEP. a Immunohistochemical staining showed ADM expression and localization in fallopian tubes of control, salpingitis and TEP groups. Epi: tubal epithelium, LP: lumina propria. Scale bar = 100 μm; n = 5 for each group. Negative control: use isotype control antibody as the primary antibody. b qPCR was used to measure ADM mRNA levels in tubal mucosa collected from control (n = 13), salpingitis (n = 11) and TEP (n = 11); data are presented as dot-plots with mean ± SEM; t-test; *p < 0.05.
Macrophages in the fallopian tubes with TEP and salpingitis skew towards M1 phenotype and express ADM receptors
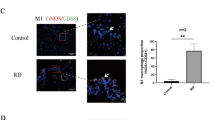
The proportion of CD68+ macrophages were significantly higher in the TEP group (p < 0.01) and the salpingitis group (p < 0.05) than the control group (Fig. 2a). The results were supported by immunohistochemical (IHC) staining using anti-CD68 antibody (Fig. 2b). The macrophages were widely spread in the mucosa and the lamina propria of the fallopian tube (Supplementary Fig. 1). Clusters of macrophages adjacent to blood vessels were observed (Supplementary Fig. 1), suggesting a blood origin of these macrophages.
a Percentages of CD68+ macrophage within mononuclear cells in control (n = 9), salpingitis (n = 7) and TEP (n = 7) groups were analyzed by flow cytometry. b Immunofluorescent staining of CD68 (green) and DAPI (blue) in fallopian tube frozen sections of the control, salpingitis and TEP groups. Upper right corner: quantification of the number of CD68+ macrophages in fallopian tubes of control (n = 5), salpingitis (n = 6) and TEP (n = 6). c Quantification of mRNA levels of iNOS, CD163 and CD206 in human monocytes derived M1, M2 macrophages, and fallopian tube macrophages from salpingitis and TEP patients (n = 4 in each group). #undetectable. Fallopian tube frozen sections were stained with immunofluorescence-labeled CD68 (green), DAPI (blue), and CRLR or RAMP2 (red). d Representative image showing CRLR-expressing CD68+ macrophages as indicated by the white arrows. e Representative image with RAMP2-expressing CD68+ macrophages. Scale bar = 20 µm. All values represent meanl ± SEM, t-test; *p < 0.05, **p < 0.01.
qPCR results showed that macrophages from the TEP and the salpingitis groups expressed iNOS (M1 macrophage marker) but not CD163/CD206 (M2 macrophage markers) (Fig. 2c). Similar observations were obtained using immunofluorescence staining of the fallopian tube macrophages (Supplementary Fig. 2A and B).
ADM exerts its biological activities by binding to CRLR/RAMP2 and CRLR/RAMP3 receptor complexes. CRLR and RAMP2 (Fig. 2d, e), but not RAMP3 (Supplementary Fig. 2C) immunoreactivities were identified in fallopian tube macrophages of the TEP and the salpingitis groups.
M1 macrophages increase the susceptibility of tubal epithelium to embryo implantation
E-cadherin, active β-catenin, leukemia inhibitory factor (LIF) and homeobox protein A10 (HoxA-10) are highly up-regulated in endometrium in the peri-implantation phase. They play key roles in establishing endometrial receptivity.26,27,28,29 Their dysregulation lead to implantation failure and infertility. The expressions of these molecules in the fallopian tube were significantly higher in the salpingitis and the TEP groups than the control (Fig. 3a and Supplementary Table 1). Incubation with the spent media from macrophages of the salpingitis and the TEP groups significantly enhanced the expression of these molecules in and attachment of trophoblastic spheroids onto the fallopian tube cells (OE-E6/E7, Fig. 3b), indicating that the tubal macrophages increased the susceptibility of tubal epithelium for embryo implantation.
a IHC images of implantation-related molecules (E-cadherin, active β-catenin, LIF and HoxA-10) in the fallopian tubes of the control, salpingitis and TEP groups. Negative control was set up by omitting the corresponding primary antibody. Scale bar=100 μm; n = 5 for each group. b OE-E6/E7 cells were stimulated with the spent medium derived from fallopian tube macrophages with or without ADM treatment. Upper panel: expression of implantation-related molecules in OE-E6/E7 cells were detected by Western blotting; n = 4. Lower panel: attachment rates of JEG-3 trophoblastic spheroids onto OE-E6/E7 monolayer were quantified; n = 8. c OE-E6/E7 cells were treated with the spent medium derived from M1 macrophages with or without LPS/ADM treatment. Upper panel: expression of implantation-relation-molecules in OE-E6/E7 cells; n = 4. Lower panel: Attachment rate of trophoblastic spheroids onto OE-E6/E7 monolayer, n = 6. All values are shown as mean ± SEM; t-test; *p < 0.05, **p < 0.01, ***p < 0.001.
Implantation involves a local inflammatory event in the endometrium.30 Consistently, pro-inflammatory M1 macrophages were dominant in the fallopian tube of TEP and salpingitis (see above). Spent medium of LPS-activated M1 macrophages had similar stimulatory effects on the expression of the above implantation-associated molecules (Fig. 3b) and the attachment of trophoblast spheroids (Fig. 3c), suggesting that M1 macrophages enhanced receptivity of the fallopian tube.
The stimulatory effects of spent media of fallopian tube (salpingitis and TEP) macrophages (Fig. 3b) and LPS-activated M1 macrophages (Fig. 3c) were significantly suppressed by treatment of the macrophages with ADM.
ADM suppresses IL-6 and IL-8 production from tubal/M1 macrophages via NF-κB signaling pathway
The cytokine/chemokine secretome of the tubal macrophages from the salpingitis and the TEP groups with or without ADM treatment were analyzed using membrane-based antibody array (Fig. 4a) and ELISA (Fig. 4b). ADM administration significantly suppressed the pro-inflammatory IL-6 and IL-8 production by the tubal macrophages of TEP and salpingitis (Fig. 4a, b). LPS induced IL-6 and IL-8 production from blood monocytes-derived M1 macrophages and THP-1 derived macrophages, and were attenuated by ADM dose-dependently (Fig. 4c). ADM receptor mediated the action of ADM as siRNA suppression of CRLR protein expression in the THP-1 derived macrophages abolished the inhibitory effect of ADM on LPS-induced IL-6 and IL-8 production (Fig. 4d).
a Upper panel: human cytokine array coordinate. Lower panel: cytokine profiles probed with the spent medium of fallopian tube macrophages of salpingitis and TEP with or without ADM treatment (n = 2), each blot representing immunoreactivity against the respective cytokine. b ELISA quantification of IL-6 and IL-8 concentration of the spent medium above; t-test; n = 7; *p < 0.05; **p < 0.01. c M1 macrophages (left panel) and THP-1 derived macrophages (right panel) were treated with LPS (100 ng/ml) with ADM in various concentrations, IL-8 and IL-6 expression in the spent medium were detected; n = 5. d Upper panel: THP-1 derived macrophages were transfected with CRLR siRNA and control siRNA, transfection efficiency was quantified; n = 3. Lower panel: siRNA (control vs CRLR) transfected THP-1 derived macrophages were challenged with LPS (100 ng/ml) with or without ADM (100 nM) for 12 h. IL-8 and IL-6 concentrations in the spent medium were detected; n = 4. e Western-blotting was applied to detect expression of key components of NF-κB signaling pathway in THP-1 derived macrophages with LPS and ADM treatments; n = 4. All values are shown as mean ± SEM; t-test; #p < 0.05, ##p < 0.01 vs control; *p < 0.05, **p < 0.01, ***p < 0.001 compared to LPS treatment group.
NF-κB signaling pathway is closely associated with macrophage-induced inflammation.31 ADM treatment dose-dependently reversed the LPS-induced up-regulation of p-IκB-α, p-p65 and p-p50 in the THP-1 derived macrophages (Fig. 4e), showing that ADM ablated LPS-induced NF-κB signaling in the human macrophages.
IL-6 and IL-8 stimulate tubal receptivity
The role of macrophages-derived IL-6 and IL-8 on induction of fallopian tube receptivity was studied by adding functional blocking antibodies against IL-6 or IL-8 to the spent medium of tubal macrophages from the salpingitis group. Such treatment significantly diminished the stimulatory effect of the media on expression of implantation-associated molecules and trophoblastic spheroids attachment (Fig. 5).
OE-E6/E7 cells were treated with spent medium of fallopian tube macrophages of salpingitis with isotype antibody, anti-IL-6 antibody, anti-IL-8 antibody and ADM. Expression of implantation-related molecules expression in OE-E6/E7 cells were detected (n = 4) and attachment rates of trophoblastic spheroids onto OE-E6/E7 monolayer were calculated (n = 5). All values are shown as mean ± SEM; t-test; *p < 0.05, **p < 0.01 and ***p < 0.001.
Discussion
In the present study, we demonstrated reduced expression of ADM in fallopian tubes of patients with salpingitis and TEP, and revealed that ADM reduction contributed to aggravated pro-inflammatory responses with release of cytokines IL-6 and IL-8, predisposing the tubal epithelium to a receptive state for embryo implantation. To our knowledge, this is the first functional study on the role of tubal macrophages in TEP.
Macrophages account for 20–30% of decidual leukocytes in intrauterine pregnancy. These macrophages display an immunosuppressive M2 phenotype.32 Macrophages are capable of creating a permissive microenvironment for embryo implantation, decidualization and placentation by secretion of cytokines, proteases and growth factors.33 Although macrophages are the predominant immune cells in the fallopian tube of TEP, little is known about their roles in pathogenesis of the disease.17 Our study filled in this knowledge gap. Compared with the control group, there were 4- to 8-fold increases in macrophages with iNOS-expressing M1 phenotype in the fallopian tubes of salpingitis and TEP, consistent with a pro-inflammatory tubal microenvironment in these conditions.
Cytokines play a fundamental role at the fetal-maternal interface in early pregnancy, and dysregulation in their production leads to pregnancy disorder. IL-6 and IL-8 contribute to establishment of a receptive endometrium and initiation of implantation.34 Uterine expression of IL-6 peaks at the mid-secretory phase,35 and is significantly reduced in women with recurrent miscarriages.36 IL-8 expression in human endometrium peaks in the late secretory phase and early pregnancy.21 A reduced IL-8 expression is associated with fetal loss in mice and humans.37 Both IL-6 and IL-8 are crucial in regulation of extravillous trophoblast invasion, decidualization, vascular angiogenesis and placenta development.38,39 The high plasma levels of IL-6 and IL-8 in patients have been proposed to be used as diagnostic biomarkers for PID and TEP.14 Activated peritoneal macrophages from TEP patients are potent producers of IL-6 and IL-8.40
ADM immunoreactivity was predominantly localized to the tubal epithelium, indicating that the tubal epithelial cells were the main source of ADM. ADM acted on tubal macrophages expressing ADM receptor components. Intriguingly, we found that a reduced tubal ADM expression was associated with an increased proportion of tubal M1 macrophages and an overexpression of IL-6 and IL-8, and that ADM inhibited LPS-induced IL-6 and IL-8 production from both monocyte-derived M1 macrophages and THP-1 derived macrophages, via suppression of NF-κB signaling. These results demonstrated that the high levels of IL-6 and IL-8 in salpingitis and TEP are at least partly resulting from the pro-inflammatory responses of the tubal macrophages induced by reduction in ADM expression. According to our study, the action of macrophages from salpingitis and TEP patients on enhancement of implantation is largely due to their increased production of IL-6 and IL-8, and suppression of the cytokine production by ADM administration abolished the macrophages’ action on tubal implantation.
Human tubal epithelial cells and embryos express IL-6 receptors.13,41,42 IL-8 receptors CXC-chemokine receptors have been detected in tubal epithelium and trophoblasts.13,41 Therefore, a high concentration of IL-6 and IL-8 could predispose the fallopian tube to a receptive state on the one hand,13 and facilitate early trophectoderm development on the other.43 In addition, a high level of IL-6 reduces CBF contributing to embryo retention within the fallopian tube.44
A large number of paracrine signals are involved in intrauterine implantation.45 Implantation fails in LIF-deficient mice, due to unresponsiveness of endometrium to the implanting blastocysts.27,46 HoxA-10 is crucially involved in pinopods formation and endometrium receptivity. A decreased endometrial HoxA-10 expression is associated with reduced implantation in mice.26,47 E-cadherin is a receptivity marker distinctly expressed on the apical surface of endometrial epithelial cells in the peri-implantation period and in receptive endometrial cell lines.28 It mediates adhesion and attachment between the endometrial epithelium and the trophectoderm.48 The expression of Wnt/β-catenin signaling is upregulated in the epithelial cells at the implantation site.49
Intriguingly, we found elevation of the above implantation-associated molecules in the fallopian tubes of patients with salpingitis and TEP when compared to that of non-pregnant controls. Consistently, the expression of LIF, HoxA-10 and Wnt signaling are enhanced in the fallopian tubes of women with TEP and chronic inflammation.50,51,52 Thus, infection-induced tubal damages dysregulate these signals, inducing an adhesive and receptive tubal epithelium predisposing to embryo-tubal implantation.45
The tubal macrophages from patients with salpingitis and TEP enhanced expression of implantation-associated molecules and attachment of trophoblastic spheroids onto the tubal epithelial cells. ADM administration weakened these macrophage-induced effects. We confirmed that the inhibition of macrophages-derived IL-6 and IL-8 production by ADM reduced the expression of the implantation-associated molecules.
ADM expression peaks in the early luteal phase when the developing embryo resides in the fallopian tube.53 Low level of tubal ADM was associated with impaired CBF and smooth muscle contraction, leading to dysfunctional transport and retention of embryo within the tube.10 The present study further showed that inflammation-associated reduced ADM expression promoted a hospitable tubal microenvironment for embryo implantation. Interestingly, a decreased ADM expression occurs in various pathological inflammatory conditions.54,55,56 Our research included tubal samples from women with salpingitis, suggesting that ADM might have the potential to restore the pathological changes of the fallopian tube before embryo implantation.
The main limitation of the present study was the small sample size. Pathophysiological studies of TEP are difficult, because it is ethically impossible to obtain ideal fallopian tube samples from normal pregnancy at a gestational age comparable to that of the TEP patients. We used fallopian tube samples from early or mid-luteal phase of non-pregnant women as controls, because they have hormonal levels close to that during implantation. In order to exclude the influence of embryos and to better understand the pathological changes leading to TEP, fallopian tubes from women with salpingitis were used as a predisposing state of TEP. However, since both the control and salpingitis groups included patients with gynecological disorders, such as endometriosis and adenomyosis, we cannot exclude the possibility that these pathological conditions may affect the functions of the fallopian tube, and thereby our observations. For example, ADM levels have shown to be elevated in the eutopic endometrium and plasma of women with endometriosis.57 This possibility will be the subject of a future study. Another limitation of our study is that in some of our experiments, we used in vitro polarized macrophages, which could not completely simulate tubal macrophages. The physiology and pathophysiology of tubal macrophages requires further in-depth investigation.
Our findings are summarized in Fig. 6. Specifically, inflammation-induced reduction in tubal ADM stimulates tubal macrophages to produce high amount of IL-6 and IL-8, and consequently predisposing the tubal epithelium to an adhesive and receptive phenotype susceptible to embryo implantation. In conjunction with our previous study,10 this study focuses on the anti-inflammatory activities of ADM and confirms that ADM reduction contributes to the pathophysiology of TEP partly by triggering excessive inflammatory responses of the residing macrophages.
Reduced ADM secretion by tubal epithelial cells contributes to excessive IL-6 and IL-8 production in fallopian tube macrophages, resulting in the up-regulation of implantation-related molecules (E-cadherin, active β-catenin, LIF and HoxA-10) in the tubal epithelium. Reduction of ADM caused decreased ciliary beat frequency, leading to embryo retention within the fallopian tube.
Materials and methods
Study population and human fallopian tube samples collection
This study was approved by the Institutional Review Board of the University of Hong Kong/Hospital Authority Hong Kong West Cluster (Ref. no: UW 13–224; Date of approval: 15 Apr 2013) and the Ethics Committee of Zhongnan Hospital of Wuhan University (Ref. no: 2017030; Date of approval: 29 Mar 2017) before the study was started. Women in their early/mid-luteal phase underwent hysterectomy and salpingectomy for benign diseases (adenomyosis, myoma and CIN III) or TEP from April to November 2017 were recruited with signed consent before the surgery. The ampulla of the fallopian tubes were collected aseptically and transported to the laboratory for processing within 2 h of collection.
All the recruited patients had regular 21–35 days menstrual cycles and did not use any hormonal nor antibiotic treatment within 3 months prior to the surgery. Patients complicated with severe comobidities or ongoing pharmacologic treatment were excluded. The phase of the menstrual cycle was determined by day of last menstruation and histology of endometrial biopsy.
The patients were divided into the control group with normal fallopian tubes (mean age ± SD = 46.5 ± 1.7; n = 13), and the salpingitis group (mean age = 47.1 ± 2.8; n = 15) based on intra-operative findings and histological diagnosis of PID or salpingitis. All the patients in control and salpingitis groups only underwent hysterectomy and bilateral salpingectomy. None of them referred to mono/bilateral oophorectomy. Table 1 shows their demographic information.
Women having salpingectomy for TEP and not receiving methotrexate with no history of in vitro fertilization treatment were assigned as the TEP group (mean age = 33.5 ± 4.0; n = 15). TEP was diagnosed by pelvic ultrasound for the presence of a conceptus in the fallopian tube without intrauterine implantation. Tubal tissues obtained were at least 0.5 cm away from the gestational mass. Table 2 shows the demographic information of the TEP patients.
Fallopian tube macrophages isolation and flow-cytometry
Fallopian tubes were digested in the Rosewell Park Memorial Institute (RPMI)-1640 medium containing collagenase V (10 mg/ml) and 0.01% DNase (Sigma-Aldrich, St. Louis, MO) for 2 h at 37 °C with gentle shaking. The resulting cell suspensions were successively filtered through 100 μm and 40 μm Millipore filters (Millipore, Billerica, MA) followed by centrifugation. The cell pellet was washed, resuspended in RPMI-1640 medium, laid on Ficoll-plaque (GE healthcare, Sweden), and centrifuged at 700g for 30 min. Leukocytes in the interface layer were collected and macrophages were purified by the negative immuno-magnetic beads isolation method using a commercial kit (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany). Purity of the isolated macrophages was determined by flow cytometry with CD68-FITC staining (Supplementary Fig. 3).
Monocyte isolation and macrophage polarization
Leukocytes were obtained from female buffy coat by Ficoll-Paque density gradient centrifugation, and monocytes were isolated by the CD14 monocyte isolation kit (Miltenyi Biotec). The isolated monocytes were cultured in the RMPI-1640 medium and challenged with 50 ng/ml granulocyte-macrophage colony-stimulating factor for 7 days to induce M1 polarization.58 M2 macrophage polarization was induced by 50 ng/ml macrophage colony-stimulating factor for 9 days. Purities of the M1 and M2 macrophages were determined by flow cytometry using antibodies against CD68, CD80 (M2 marker), CD206 (M2 marker) and CD163 (M2 marker) (Supplementary Fig. 4).
Cell lines
Human JEG-3 cells and human monocyte THP-1 cells were obtained from ATCC (American Type Culture Collection, Manassas, VA). JEG-3 is a choriocarcinoma cell line with properties of extravillous trophoblasts.59 The immortalized fallopian tube epithelial cell line, OE-E6/E7, were cultured as described.60,61 THP-1 derived macrophages were obtained by stimulating these cells with PMA (100 ng/ml) for 48 h as described.62
Confocal microscopy and IHC
For confocal microscopy, fresh fallopian tube tissues were snap frozen by liquid nitrogen. Cryosections of thickness 5 μm were fixed in acetone and permeabilized with 0.01% Triton. For IHC, fallopian tube tissues were fixed in 10% formalin for 24 h, dehydrated in ethanol, paraffin-embedded, and sliced into 5 μm sections. The sections were dewaxed, rehydrated and incubated in 3% H2O2 for 15 min to block endogenous tissue peroxidase. Afterwards, the slides were immersed in 0.1 M sodium citrate and antigen retrieval was done in a microwave oven at 750 W for 20 min, followed by washing and blocking with 10% donkey serum (Sigma) in PBS.
The sections were incubated with specific antibodies (Supplementary Table 2) overnight at 4 °C, washed with PBS and incubated with fluorescence (for confocal microscopy) or biotin-labeled (for IHC) secondary antibodies for 1 h. A positive brown signal in IHC was visualized with 3,3-diaminobenzidine (DakoCytomation) as substrate. The sections were examined under a Zeiss LSM 510 confocal microscope or light microscope.
Reverse transcription-quantitative PCR (RT-qPCR)
TRIZOL reagent (Invitrogen) were used to extract total RNA from the mucosal layer of fallopian tubes. The total mRNA was reverse transcribed with the TaqMan reverse transcription reagent kit (Applied Biosystems, Foster City, CA, USA), and the resulting cDNA was subjected to qPCR analysis using primers listed in Supplementary Table 3. Each sample was assayed in duplicate. Gene expression levels were determined using the threshold cycle (2-ΔΔCT) method with reference to endogenous 18 S.
Western blotting
Cells (1 × 106) were lysed in RIPA buffer supplemented with protease inhibitors (0.1% w/v), resolved in 10% polyacrylamide gel and transferred onto the polyvinylidene difluoride membrane for Western blot analysis using specific antibodies (Supplementary Table 4). Intensity of specific proteins was quantified by densitometry using the Image J software (National Institutes of Health, Bethesda, Maryland, US).
Suppression of ADM receptor expression in macrophages
THP-1 derived macrophages (1 × 106 cells) were transfected with 100 nM of siRNA of CRLR or control siRNA (ThermoFisher, USA) by electroporation with 2 pulses at 1400 mV for 20 ms. The transfected cells were cultured in RPMI-1640 medium without antibiotics for 24 h before determination of the expression of CRLR by Western blotting.
In vitro implantation assay
Implantation potential was assessed by attachment of JEG-3 trophoblast spheroids onto OE-E6/E7 cells. The spheroids (Supplementary Fig. 5A) with diameter of 60–100 μm were cocultured on an OE-E6/E7 monolayer (Supplementary Fig. 5B) for 45 min. The non-attached spheroids were removed by rotation at 140 rpm for 10 min. The attachment rate was calculated as a percentage of the number of attached spheroids divided by the total number of spheroids added (Supplementary Fig. 5A).
Scoring and statistical analysis
We assessed immunoreactivities in fallopian tube sections using the McCarty’s H-scoring system with an equation: H-score = Σ Pi(i), where i was staining intensity graded as 0, 1, 2 and 3 (highest intensity), while Pi was percentage of tissue stained at each intensity (0–100%), giving a total score between 0 to 300. The Image Pro Plus 6.0 software (Media Cybernetics, Silver Spring, MD, USA) was used to determine the Pi value.63
Data were presented as mean ± standard error of the mean (SEM) unless otherwise indicated. The Kolgomorov-Smirnov test was used for determining normality of data. For normally distributed data, the t-tests was used to determine differences between groups. For data not normally distributed, Mann-Whitney U-test was used to compare two variable groups. Statistical significance was assessed by the GraphPad Prism v.5. software (GraphPad, San Diego, CA). A p-value < 0.05 was considered to be significant.
References
Pisarska, M. D., Carson, S. A. & Buster, J. E. Ectopic pregnancy. Lancet 351, 1115–1120 (1998).
Coste, J., Bouyer, J. & Job-Spira, N. [Epidemiology of ectopic pregnancy: incidence and risk factors]. Contracept. Fertil. Sex. 24, 135–139 (1996).
Westrom, L., Joesoef, R., Reynolds, G., Hagdu, A. & Thompson, S. E. Pelvic inflammatory disease and fertility. A cohort study of 1,844 women with laparoscopically verified disease and 657 control women with normal laparoscopic results. Sex. Transm. Dis. 19, 185–192 (1992).
Corpa, J. M. Ectopic pregnancy in animals and humans. Reproduction 131, 631–640 (2006).
Ankum, W. M., Mol, B. W., Van der Veen, F. & Bossuyt, P. M. Risk factors for ectopic pregnancy: a meta-analysis. Fertil. Steril. 65, 1093–1099 (1996).
Kitamura, K. [Structure, gene and biosynthesis of adrenomedullin]. Nihon Rinsho 62(Suppl 9), 184–188 (2004).
Muff, R., Born, W. & Fischer, J. A. Calcitonin, calcitonin gene-related peptide, adrenomedullin and amylin: homologous peptides, separate receptors and overlapping biological actions. Eur. J. Endocrinol. 133, 17–20 (1995).
McLatchie, L. M. et al. RAMPs regulate the transport and ligand specificity of the calcitonin-receptor-like receptor. Nature 393, 333–339 (1998).
Li, H. W. et al. Expression of adrenomedullin in human oviduct, its regulation by the hormonal cycle and contact with spermatozoa, and its effect on ciliary beat frequency of the oviductal epithelium. J. Clin. Endocrinol. Metab. 95, E18–E25 (2010).
Liao, S. B. et al. Possible role of adrenomedullin in the pathogenesis of tubal ectopic pregnancy. J. Clin. Endocrinol. Metab. 97, 2105–2112 (2012).
Wang, H. et al. Aberrant cannabinoid signaling impairs oviductal transport of embryos. Nat. Med. 10, 1074–1080 (2004).
van Mourik, M. S., Macklon, N. S. & Heijnen, C. J. Embryonic implantation: cytokines, adhesion molecules, and immune cells in establishing an implantation environment. J. Leukoc. Biol. 85, 4–19 (2009).
Balasubramaniam, E. S., Van Noorden, S. & El-Bahrawy, M. The expression of interleukin (IL)-6, IL-8, and their receptors in fallopian tubes with ectopic tubal gestation. Fertil. Steril. 98, 898–904 (2012).
Soriano, D., Hugol, D., Quang, N. T. & Darai, E. Serum concentrations of interleukin-2R (IL-2R), IL-6, IL-8, and tumor necrosis factor alpha in patients with ectopic pregnancy. Fertil. Steril. 79, 975–980 (2003).
Jabbour, H. N., Sales, K. J., Catalano, R. D. & Norman, J. E. Inflammatory pathways in female reproductive health and disease. Reproduction 138, 903–919 (2009).
Vacca, P., Moretta, L., Moretta, A. & Mingari, M. C. Origin, phenotype and function of human natural killer cells in pregnancy. Trends Immunol. 32, 517–523 (2011).
Vassiliadou, N. & Bulmer, J. N. Characterization of tubal and decidual leukocyte populations in ectopic pregnancy: evidence that endometrial granulated lymphocytes are absent from the tubal implantation site. Fertil. Steril. 69, 760–767 (1998).
Wang, X. et al. Alteration of the immune cell profiles in the pathophysiology of tubal ectopic pregnancy. Am. J. Reprod. Immunol. 81, e13093 (2019).
von Rango, U., Classen-Linke, I., Kertschanska, S., Kemp, B. & Beier, H. M. Effects of trophoblast invasion on the distribution of leukocytes in uterine and tubal implantation sites. Fertil. Steril. 76, 116–124 (2001).
Renaud, S. J. & Graham, C. H. The role of macrophages in utero-placental interactions during normal and pathological pregnancy. Immunol. Invest. 37, 535–564 (2008).
Arici, A. et al. Interleukin-8 in the human endometrium. J. Clin. Endocrinol. Metab. 83, 1783–1787 (1998).
Matsusaka, T. et al. Transcription factors NF-IL6 and NF-kappa B synergistically activate transcription of the inflammatory cytokines, interleukin 6 and interleukin 8. Proc. Natl. Acad. Sci. USA 90, 10193–10197 (1993).
Prins, J. R., Gomez-Lopez, N. & Robertson, S. A. Interleukin-6 in pregnancy and gestational disorders. J. Reprod. Immunol. 95, 1–14 (2012).
Saito, R. et al. Function of adrenomedullin in inflammatory response of liver against LPS-induced endotoxemia. APMIS 120, 706–711 (2012).
Yang, S., Zhou, M., Fowler, D. E. & Wang, P. Mechanisms of the beneficial effect of adrenomedullin and adrenomedullin-binding protein-1 in sepsis: down-regulation of proinflammatory cytokines. Crit. Care Med. 30, 2729–2735 (2002).
Bagot, C. N., Kliman, H. J. & Taylor, H. S. Maternal Hoxa10 is required for pinopod formation in the development of mouse uterine receptivity to embryo implantation. Dev. Dyn. 222, 538–544 (2001).
Bhatt, H., Brunet, L. J. & Stewart, C. L. Uterine expression of leukemia inhibitory factor coincides with the onset of blastocyst implantation. Proc. Natl. Acad. Sci. USA 88, 11408–11412 (1991).
Jha, R. K., Titus, S., Saxena, D., Kumar, P. G. & Laloraya, M. Profiling of E-cadherin, beta-catenin and Ca(2+) in embryo-uterine interactions at implantation. FEBS Lett. 580, 5653–5660 (2006).
Matsuzaki, S., Darcha, C., Maleysson, E., Canis, M. & Mage, G. Impaired down-regulation of E-cadherin and beta-catenin protein expression in endometrial epithelial cells in the mid-secretory endometrium of infertile patients with endometriosis. J. Clin. Endocrinol. Metab. 95, 3437–3445 (2010).
Dimitriadis, E., White, C. A., Jones, R. L. & Salamonsen, L. A. Cytokines, chemokines and growth factors in endometrium related to implantation. Hum. Reprod. Update 11, 613–630 (2005).
Kim, Y. S., Ahn, C. B. & Je, J. Y. Anti-inflammatory action of high molecular weight Mytilus edulis hydrolysates fraction in LPS-induced RAW264.7 macrophage via NF-kappaB and MAPK pathways. Food Chem. 202, 9–14 (2016).
Singh, U. et al. Immunological properties of human decidual macrophages-a possible role in intrauterine immunity. Reproduction 129, 631–637 (2005).
Hunt, J. S., Petroff, M. G. & Burnett, T. G. Uterine leukocytes: key players in pregnancy. Semin. Cell Dev. Biol. 11, 127–137 (2000).
Boomsma, C. M. et al. Cytokine profiling in endometrial secretions: a non-invasive window on endometrial receptivity. Reprod. Biomed. Online 18, 85–94 (2009).
Tabibzadeh, S., Kong, Q. F., Babaknia, A. & May, L. T. Progressive rise in the expression of interleukin-6 in human endometrium during menstrual cycle is initiated during the implantation window. Hum. Reprod. 10, 2793–2799 (1995).
Jasper, M. J., Tremellen, K. P. & Robertson, S. A. Reduced expression of IL-6 and IL-1alpha mRNAs in secretory phase endometrium of women with recurrent miscarriage. J. Reprod. Immunol. 73, 74–84 (2007).
Pitman, H., Innes, B. A., Robson, S. C., Bulmer, J. N. & Lash, G. E. Altered expression of interleukin-6, interleukin-8 and their receptors in decidua of women with sporadic miscarriage. Hum. Reprod. 28, 2075–2086 (2013).
De Oliveira, L. G. et al. Role of interleukin 8 in uterine natural killer cell regulation of extravillous trophoblast cell invasion. Placenta 31, 595–601 (2010).
Jovanovic, M. & Vicovac, L. Interleukin-6 stimulates cell migration, invasion and integrin expression in HTR-8/SVneo cell line. Placenta 30, 320–328 (2009).
Bergqvist, A., Bruse, C., Carlberg, M. & Carlstrom, K. Interleukin 1beta, interleukin-6, and tumor necrosis factor-alpha in endometriotic tissue and in endometrium. Fertil. Steril. 75, 489–495 (2001).
Dame, J. B. & Juul, S. E. The distribution of receptors for the pro-inflammatory cytokines interleukin (IL)-6 and IL-8 in the developing human fetus. Early Hum. Dev. 58, 25–39 (2000).
Wanggren, K. et al. Leukaemia inhibitory factor receptor and gp130 in the human Fallopian tube and endometrium before and after mifepristone treatment and in the human preimplantation embryo. Mol. Hum. Reprod. 13, 391–397 (2007).
Champion, H., Innes, B. A., Robson, S. C., Lash, G. E. & Bulmer, J. N. Effects of interleukin-6 on extravillous trophoblast invasion in early human pregnancy. Mol. Hum. Reprod. 18, 391–400 (2012).
Papathanasiou, A., Djahanbakhch, O., Saridogan, E. & Lyons, R. A. The effect of interleukin-6 on ciliary beat frequency in the human fallopian tube. Fertil. Steril. 90, 391–394 (2008).
Giudice, L. C. Potential biochemical markers of uterine receptivity. Hum. Reprod. 14(Suppl 2), 3–16 (1999).
Stewart, C. L. et al. Blastocyst implantation depends on maternal expression of leukaemia inhibitory factor. Nature 359, 76–79 (1992).
Bagot, C. N., Troy, P. J. & Taylor, H. S. Alteration of maternal Hoxa10 expression by in vivo gene transfection affects implantation. Gene Ther. 7, 1378–1384 (2000).
Rahnama, F. et al. Epigenetic regulation of E-cadherin controls endometrial receptivity. Endocrinology 150, 1466–1472 (2009).
Tulac, S. et al. Identification, characterization, and regulation of the canonical Wnt signaling pathway in human endometrium. J. Clin. Endocrinol. Metab. 88, 3860–3866 (2003).
Krishnan, T. et al. The role of leukemia inhibitory factor in tubal ectopic pregnancy. Placenta 34, 1014–1019 (2013).
Li, P. et al. Enhanced beta-catenin expression and inflammation are associated with human ectopic tubal pregnancy. Hum. Reprod. 28, 2363–2371 (2013).
Salih, S. M. & Taylor, H. S. HOXA10 gene expression in human fallopian tube and ectopic pregnancy. Am. J. Obstet. Gynecol. 190, 1404–1406 (2004).
Chiu, P. C. et al. Adrenomedullin regulates sperm motility and oviductal ciliary beat via cyclic adenosine 5′-monophosphate/protein kinase A and nitric oxide. Endocrinology 151, 3336–3347 (2010).
Fujikura, T. & Okubo, K. Adrenomedullin level in the nasal discharge from allergic rhinitis cohort. Peptides 32, 368–373 (2011).
Hagner, S. et al. Suppression of adrenomedullin contributes to vascular leakage and altered epithelial repair during asthma. Allergy 67, 998–1006 (2012).
Zhang, S., Patel, A., Moorthy, B. & Shivanna, B. Adrenomedullin deficiency potentiates hyperoxic injury in fetal human pulmonary microvascular endothelial cells. Biochem. Biophys. Res. Commun. 464, 1048–1053 (2015).
Matson, B. C., Quinn, K. E., Lessey, B. A., Young, S. L. & Caron, K. M. Elevated levels of adrenomedullin in eutopic endometrium and plasma from women with endometriosis. Fertil. Steril. 109, 1072–1078 (2018).
Fleetwood, A. J., Lawrence, T., Hamilton, J. A. & Cook, A. D. Granulocyte-macrophage colony-stimulating factor (CSF) and macrophage CSF-dependent macrophage phenotypes display differences in cytokine profiles and transcription factor activities: implications for CSF blockade in inflammation. J. Immunol. 178, 5245–5252 (2007).
Apps, R. et al. Human leucocyte antigen (HLA) expression of primary trophoblast cells and placental cell lines, determined using single antigen beads to characterize allotype specificities of anti-HLA antibodies. Immunology 127, 26–39 (2009).
Lee, Y. L. et al. Establishment and characterization of an immortalized human oviductal cell line. Mol. Reprod. Dev. 59, 400–409 (2001).
Ling, L. et al. Expression of human oviductin in an immortalized human oviductal cell line. Fertil. Steril. 84(Suppl 2), 1095–1103 (2005).
Lee, C. L. et al. Glycodelin-A stimulates interleukin-6 secretion by human monocytes and macrophages through L-selectin and the extracellular signal-regulated kinase pathway. J. Biol. Chem. 287, 36999–37009 (2012).
Yung, S. S. F. et al. The effect of 7 days of letrozole pretreatment combined with misoprostol on the expression of progesterone receptor and apoptotic factors of placental and decidual tissues from first-trimester abortion: a randomized controlled trial. Contraception 93, 323–330 (2016).
Acknowledgements
The authors thank the Hong Kong Red Cross Blood Transfusion Service for their help in obtaining the buffy coat samples, and staffs of the Faculty Core Facility, Faculty of Medicine, The University of Hong Kong for their technical supports. This work was supported in part by the Hong Kong Research Grant Council Fund (17118917 and 17113314), and the Health and Medical Research Fund, Hong Kong (02131786).
Author information
Authors and Affiliations
Contributions
X.W. conceived and carried out the experiments, acquired and analyzed data, and drafted the manuscript; P.C.N.C., C.L.L. and R.H.W.L. designed the study, interpreted the data and prepared the manuscript; X.W.W. and Y.Z.Z. collected the sample and acquired data; M.V., W.S.B.Y., E.H.Y.N. and W.S.O. critically revised the manuscript. All authors read and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, X., Lee, CL., Vijayan, M. et al. Adrenomedullin insufficiency alters macrophage activities in fallopian tube: a pathophysiologic explanation of tubal ectopic pregnancy. Mucosal Immunol 13, 743–752 (2020). https://doi.org/10.1038/s41385-020-0278-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41385-020-0278-6