Abstract
Background/objectives
Polymethoxyselenoflavone (PMSF) is a compound that substitutes the oxygen atom in a flavonoid with selenium. This study aimed to investigate the effects of PMSFs on lipid metabolism in adipocytes and their anti-obesity potential.
Subjects/methods
To test lipolytic and thermogenic effects of the compounds in vitro, adipocytes differentiated from immortalized pre-brown adipocyte progenitors and pre-white adipocyte cell lines were treated with 19 PMSFs. The expression levels of brown adipocyte markers and genes related to mitochondrial metabolism were analyzed by qPCR and western blot. In vivo anti-obesity effect was investigated using diet-induced obesity mouse models and adipocyte-specific ATGL knockout mice.
Results
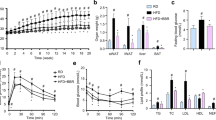
The qPCR analysis identified 2-(3,4-dimethoxyphenyl)-4H-selenochromen-4-one (DMPSC) as the most potent brown adipogenic candidate among the 19 compounds tested in this study. DMPSC treatment significantly increased the mitochondrial content and oxidative metabolism in adipocytes in vitro. Mechanistically, DMPSC treatment increased lipolysis through activation of PKA downstream signaling. Consistently, the in vivo treatment of DMPSC increased energy consumption, reduced body weight, and improved glucose tolerance in mice fed with high-fat diets. Moreover, DMPSC treatment increased brown adipocyte marker expression and mitochondrial content in adipose tissue of mice. The anti-obesity effects were absent in adipocyte-specific ATGL knockout mice, indicating that the DMPSC effect is mediated by cytosolic lipase-dependent mechanisms.
Conclusions
Collectively, our results indicated that DMPSC exerted anti-obesity effects partially through the PKA signaling-mediated activation of lipolysis and brown adipose tissue metabolism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rosen Evan D, Spiegelman Bruce M. What we talk about when we talk about fat. Cell. 2014;156:20–44.
Zwick RK, Guerrero-Juarez CF, Horsley V, Plikus MV. Anatomical, physiological, and functional diversity of adipose tissue. Cell Metab. 2018;27:68–83.
Lee Y-H, Mottillo EP, Granneman JG. Adipose tissue plasticity from WAT to BAT and in between. Biochim Biophys Acta. 2014;1842:358–69.
Yang W-R, Choi Y-S, Jeong J-H. Efficient synthesis of polymethoxyselenoflavones via regioselective direct C–H arylation of selenochromones. Organ Biomol Chem. 2017;15:3074–83.
Granneman JG. Renaissance of brown adipose tissue research: integrating the old and new. Int J Obes Suppl. 2015;5(Suppl 1):S7–S10.
Bartelt A, Heeren J. Adipose tissue browning and metabolic health. Nat Rev Endocrinol. 2014;10:24–36.
Contreras GA, Lee Y-H, Mottillo EP, Granneman JG. Inducible brown adipocytes in subcutaneous inguinal white fat: the role of continuous sympathetic stimulation. Am J Physiol-Endocrinol Metab. 2014;307:E793–E799.
Lee Y-H, Petkova AP, Konkar AA, Granneman JG. Cellular origins of cold-induced brown adipocytes in adult mice. FASEB J. 2015;29:286–99.
Pietta P-G. Flavonoids as antioxidants. J Nat Prod. 2000;63:1035–42.
Kumar S, Pandey A. Chemistry and biological activities of flavonoids: an overview. Sci World J. 2013; 2013: 16.
Nijveldt RJ, van Nood E, van Hoorn DE, Boelens PG, van Norren K, van Leeuwen PA. Flavonoids: a review of probable mechanisms of action and potential applications. Am J Clin Nutr. 2001;74:418–25.
Zhang X, Li X, Fang H, Guo F, Li F, Chen A, et al. Flavonoids as inducers of white adipose tissue browning and thermogenesis: signalling pathways and molecular triggers. Nutr Metab. 2019;16:47.
Davis C, Mudd J, Hawkins M. Neuroprotective effects of leptin in the context of obesity and metabolic disorders. Neurobiol Dis. 2014;72:61–71.
Choi Y-S, Kim D-M, Kim Y-J, Yang S, Lee K-T, Ryu JH, et al. Synthesis and evaluation of neuroprotective selenoflavanones. Int J Mol Sci. 2015;16:29574–82.
Kim S-N, Jung Y-S, Kwon H-J, Seong JK, Granneman JG, Lee Y-H. Sex differences in sympathetic innervation and browning of white adipose tissue of mice. Biol Sex Differ. 2016;7:67.
Lee YH, Petkova AP, Mottillo EP, Granneman JG. In vivo identification of bipotential adipocyte progenitors recruited by beta3-adrenoceptor activation and high-fat feeding. Cell Metab. 2012;15:480–91.
Kim S-N, Kwon H-J, Im S-W, Son Y-H, Akindehin S, Jung Y-S, et al. Connexin 43 is required for the maintenance of mitochondrial integrity in brown adipose tissue. Sci Rep. 2017;7:7159–7159.
Li P, Zhu Z, Lu Y, Granneman JG. Metabolic and cellular plasticity in white adipose tissue II: role of peroxisome proliferator-activated receptor-α. Am J Physiol-Endocrinol Metab. 2005;289:E617–E626.
Kim S-N, Kwon H-J, Akindehin S, Jeong HW, Lee Y-H. Effects of epigallocatechin-3-gallate on autophagic lipolysis in adipocytes. Nutrients. 2017;9:680.
Akindehin S, Jung YS, Kim SN, Son YH, Lee I, Seong JK. Myricetin exerts anti-obesity effects through upregulation of SIRT3 in adipose tissue. Nutrients. 2018;10:1962.
Carpentier AC, Blondin DP, Virtanen KA, Richard D, Haman F, Turcotte ÉE. Brown adipose tissue energy metabolism in humans. Front Endocrinol. 2018;9:447–447.
Granneman JG, Moore H-PH. Location, location: protein trafficking and lipolysis in adipocytes. Trends Endocrinol Metab. 2008;19:3–9.
Mottillo EP, Balasubramanian P, Lee YH, Weng C, Kershaw EE, Granneman JG. Coupling of lipolysis and de novo lipogenesis in brown, beige, and white adipose tissues during chronic beta3-adrenergic receptor activation. J Lipid Res. 2014;55:2276–86.
Verma AK, Pratap R. The biological potential of flavones. Nat Product Rep. 2010;27:1571–93.
HORÁKOVÁ Ľ. Flavonoids in prevention of diseases with respect to modulation of Ca-pump function. Interdiscip Toxicol. 2011;4:114–24.
Prasain JK, Carlson SH, Wyss JM. Flavonoids and age-related disease: risk, benefits and critical windows. Maturitas. 2010;66:163–71.
Walle T. Methoxylated flavones, a superior cancer chemopreventive flavonoid subclass? Semin Cancer Biol. 2007;17:354–62.
Sawamoto A, Nakanishi M, Okuyama S, Furukawa Y, Nakajima M. Heptamethoxyflavone inhibits adipogenesis via enhancing PKA signaling. Eur J Pharmacol. 2019;865:172758–172758.
Li RW, Theriault AG, Au K, Douglas TD, Casaschi A, Kurowska EM, et al. Citrus polymethoxylated flavones improve lipid and glucose homeostasis and modulate adipocytokines in fructose-induced insulin resistant hamsters. Life Sci. 2006;79:365–73.
Song Y, Kim M-B, Kim C, Kim J, Hwang J-K. 5,7-dimethoxyflavone attenuates obesity by inhibiting adipogenesis in 3T3-L1 adipocytes and high-fat diet-induced obese C57BL/6J mice. J Med Food. 2016;19:1111–9.
Xie Y, Zhang Y, Su X. Antidiabetic and hypolipidemic effects of 5,7-dimethoxyflavone in streptozotocin-induced diabetic rats. Med Sci Monit. 2019;25:9893–901.
Saito T, Abe D, Sekiya K. Nobiletin enhances differentiation and lipolysis of 3T3-L1 adipocytes. Biochem Biophys Res Commun. 2007;357:371–6.
Yuan X, Wei G, You Y, Huang Y, Lee HJ, Dong M, et al. Rutin ameliorates obesity through brown fat activation. FASEB J. 2017;31:333–45.
Lee SG, Parks JS, Kang HW. Quercetin, a functional compound of onion peel, remodels white adipocytes to brown-like adipocytes. J Nutr Biochem. 2017;42:62–71.
Arias N, Picó C, Teresa Macarulla M, Oliver P, Miranda J, Palou A, et al. A combination of resveratrol and quercetin induces browning in white adipose tissue of rats fed an obesogenic diet. Obesity. 2017;25:111–21.
Cong H, Zhong W, Wang Y, Ikuyama S, Fan B, Gu J. Pycnogenol® induces browning of white adipose tissue through the PKA signaling pathway in apolipoprotein E-deficient mice. J Diabetes Res. 2018;2018:9713259–9713259.
Imran KM, Yoon D, Lee T-J, Kim Y-S. Medicarpin induces lipolysis via activation of protein kinase A in brown adipocytes. BMB Rep. 2018;51:249–54.
Betz MJ, Enerbäck S. Targeting thermogenesis in brown fat and muscle to treat obesity and metabolic disease. Nat Rev Endocrinol. 2018;14:77–87.
Peng I-W, Kuo S-M. Flavonoid structure affects the inhibition of lipid peroxidation in Caco-2 intestinal cells at physiological concentrations. J Nutr. 2003;133:2184–7.
Huang J-T, Cheng Y-Y, Lin L-C, Tsai T-H. Structural pharmacokinetics of polymethoxylated flavones in rat plasma using HPLC-MS/MS. J Agric Food Chem. 2017;65:2406–13.
Turner PV, Brabb T, Pekow C, Vasbinder MA. Administration of substances to laboratory animals: routes of administration and factors to consider. J Am Assoc Lab Anim Sci. 2011;50:600–13.
Xiao Z, Morris-Natschke SL, Lee K-H. Strategies for the optimization of natural leads to anticancer drugs or drug candidates. Med Res Rev. 2016;36:32–91.
Acknowledgements
This work was supported by Research Resettlement Fund for the new faculty of Seoul National University. This research was supported by Basic Science Research Program [NRF-2018R1A5A2024425, NRF-2019R1C1C1002014] of Ministry of Science of ICT and by Korea Mouse Phenotyping Project [NRF-2013M3A9D5072550] of Ministry of Science of ICT and Future Planning through National Research Foundation of Korea (NRF).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Kwon, HJ., Saha, A., Ahn, SY. et al. Polymethoxyselenoflavones exert anti-obesity effects through activation of lipolysis and brown adipocyte metabolism. Int J Obes 45, 122–129 (2021). https://doi.org/10.1038/s41366-020-0606-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-020-0606-7
This article is cited by
-
Anti-obesity effects of the dual-active adenosine A2A/A3 receptor-ligand LJ-4378
International Journal of Obesity (2022)
-
Development of CIDEA reporter mouse model and its application for screening thermogenic drugs
Scientific Reports (2021)