Abstract
Background:
Some allergic inflammation–associated mediators have been reported in acute stage of Henoch–Schönlein purpura (HSP). However, the association of children with allergic diseases and their subsequent risks of HSP and HSP nephritis remain unknown.
Methods:
In this study, we included 2,240 children with HSP diagnosed between 2000 and 2008 as well as 8,960 non-HSP controls matched for age, sex, and level of urbanization. The odds ratios (ORs) of HSP were calculated with respect to associations with pre-existing allergic diseases.
Results:
Children with allergic diseases had an increased subsequent risk of HSP; the lowest adjusted OR (aOR) was 1.33 for allergic conjunctivitis (95% confidence interval (CI): 1.17–1.52) and the highest was 1.68 for asthma (95% CI: 1.48–1.91). The aOR increased to 2.03 (95% CI: 1.80–2.31) in children with at least two allergic diseases. Children who visited medical institutes more often per year for associated allergic diseases had an increased risk of HSP. Of the 2,240 children with HSP, 249 (11%) had HSP nephritis and 45.8% of those with nephritis had history of any allergic disease.
Conclusion:
Atopic children had an increased subsequent risk of HSP but not an increased risk of HSP nephritis.
Similar content being viewed by others
Main
Henoch–Schönlein purpura (HSP) is a common systemic small vessel vasculitis that primarily affects children and is characterized by nonthrombocytopenic purpura, abdominal pain and bleeding, arthritis, and renal involvement. Although the clinical course of HSP is generally benign and self-limited (1), renal function impairment, bowel perforation, and central nerve system involvement are rare but cause severe morbidity and long-term complication which need more aggressive treatment (1,2,3). HSP with renal involvement, HSP nephritis, has been reported to occur in 20–50% of the patients with HSP, and the long-term outcome of HSP depends primarily on the extent of renal involvement (4,5,6,7). The manifestations of HSP nephritis vary from asymptomatic hematuria and/or proteinuria to nephritic syndrome, nephrotic syndrome, and rapidly progressive glomerulonephritis (4,5,6,7). To date, the exact etiology and pathogenesis of HSP are unknown. However, HSP is regarded as a specific immune-mediated inflammation induced by environmental factors, such as infections and seasonal variation (1,8). Some inflammatory mediators, including tumor necrosis factor (TNF)-α, interleukin-6, IL-8, transforming growth factor (TGF)-β, vascular endothelial growth factor (VEGF), and cysteinyl leukotrienes, have been reported to be higher in children with acute HSP than in healthy controls (9,10,11). Cysteinyl leukotrienes are potent inflammatory mediators which contribute to asthma (12). Elevated Th2-mediated biological markers such as elevated serum IgE, serum eosinophil cationic protein (ECP), and urinary leukotriene E4 levels were found in acute stage of HSP (10,11,12,13). The onset of HSP has been reported after exposure to drug or food allergens, vaccines, and insect bites (14,15,16). In our clinical experience, some children with HSP also have allergic diseases. To date, no large population study systemically analyzes the association between allergic diseases and risks of HSP and HSP nephritis. In this nationwide, population-based, case–control study, we aim to investigate temporal relationships between allergic diseases, including allergic conjunctivitis, allergic rhinitis (AR), asthma, and atopic dermatitis, and the subsequent risk of HSP and HSP nephritis in Taiwan children.
Results
The annual incidence rate of HSP in Taiwanese children was 8.10 (7.10–8.79) per 100,000 children between 2000 and 2008 ( Table 1 ). A total of 2,240 subjects with HSP were identified, including 1,174 male (52.4%) and 1,066 female subjects (47.6%). The mean (SD) age at HSP diagnosis was 7.46 (3.94) y. More than half of the subjects with HSP resided in urban areas (approximately 61%) and were older than 5 y (70.3%). The incidence rates of the following atopic diseases were significantly higher in the HSP group than in the non-HSP group as shown in Table 2 .
In the entire study population, an increased subsequent risk of HSP was observed in children with each allergic disease ( Table 3 ). The increased risks constantly existed in both genders when the association was evaluated according to sex ( Table 3 ). We further examined the joint effects of certain allergic diseases and their association with HSP and found that the adjusted odds ratios (aORs) increased to 2.03 (95% confidence interval (CI): 1.80–2.31) in children with two more allergic diseases ( Table 3 ). The aORs of HSP increased to 2.28 (95% CI: 1.79–2.92), 1.97 (95% CI: 1.56–2.49), and 1.97 (95% CI: 1.63–2.38) for the age groups of younger than 5, 5–7, and older than 7 y, respectively, among those with two or more allergic diseases (P value < 0.001; Table 4 ). We further examined the association between the annual frequency of medical visits due to allergic diseases and the risk of HSP. Compared with those without medical visits for allergic diseases, the aORs increased in children with more allergic disease–related medical visits per year ( Table 5 ). Of the 2,240 children with HSP, 249 children (11%) had renal involvement (girls 12%, boys 10.3%). Of those with renal involvement, 114 children (45.8 %) had history of at least one allergic disease, and AR was the most prevalent allergic disease ( Table 6 ).
Discussion
This is one of the few studies on association between allergy and HSP. HSP had been termed anaphylactoid purpura because HSP was reported to develop after exposure to allergens in drugs or food, vaccine, and insect bites (15,16). Most of these studies were anecdotal reports but failed to demonstrate a causative role of atopy in HSP. Elevated soluble IL-2 receptor levels and a shift toward increased Th1 lymphocyte production in children with HSP in some studies indicate that HSP is a Th1-polarized disease (10,17,18). On the contrary, elevated Th2-mediated biological markers such as elevated serum IgE, serum ECP, and urinary leukotriene E4 levels were also found during acute stage of HSP in other studies, indicating a shift toward Th2 immune response (10,11,12,13). The complexity of Th1/Th2 interaction of HSP raises a research interest whether Th2-mediated allergic diseases have any influence on the development of HSP and HSP nephritis. This population-based study suggests that children with allergic diseases, including allergic conjunctivitis, AR, asthma, and atopic dermatitis, were more likely to develop HSP regardless of sex, age, and season. The risks increased further with the cumulative effects of concurrent allergic diseases and allergic disease–related medical visits. However, allergic diseases did not associate with further increase in renal involvement among HSP children. This study has several strengths. First, it robustly demonstrates the relationship between atopic diathesis and risk of HSP using a large number of study subjects. Second, we analyzed the effect not only of a specific type of allergic disease but of a large number of common allergic diseases, the cumulative effect of comorbid allergic diseases, and the frequency of seeking medical care, which might reflect the severity and burden of clinically evident allergic disease. Third, the current study is a population-based study with physician-diagnosed allergic diseases which minimizes selection and recall bias. Fourth, we adjusted for possible confounding factors, including age, gender, urbanization level, and season. Season is an important confounder adjusted in the current study because HSP more commonly occurs during autumn and winter (8). In addition, the onset of HSP is often preceded by an upper respiratory tract infection, and many respiratory pathogens have characteristic seasonal patterns. Fifth, ethnic influence was not considered in this study because of homogenous ethnicity in Taiwan.
The incidence of HSP in literatures has been reported to be 6–24 per 100,000 children younger than 17 y of age, depending on the ethnic and geographic background (2,3,8). Our data revealed an annual incidence of 8.1 (7.1–9.2) per 100,000 Taiwanese children younger than 19 y of age in 2000–2008. The incidence of HSP is slightly higher in boys than in girls (2,3,8). During this study period, there were 1,174 males and 1,066 females, with a male to female ratio of 1:1.1. The mean age of HSP onset was 7.46 ± 3.94 y in this study, which was similar to that of previous researches (2,3,8).
The cause of HSP is unclear, but most cases of HSP are often preceded by bacterial or viral infection (14,15,16). In addition, allergic reactions also may participate in the onset of HSP. HSP has been reported to follow drug or food allergy, insect bites, and immunization (14,15,16). Most of previous studies were anecdotal reports or studies with limited number of patients that found HSP occurs after allergic reaction or elevated Th2-mediated biological markers such as serum IgE, serum ECP, and urinary leukotriene E4 levels in acute stage of HSP but failed to demonstrate a temporal relationship of atopy in HSP and HSP nephritis (2,3,8,9,10,11,14,15,16,19). Our results prove that children with allergic diseases were more likely to develop HSP.
Although most of the children with HSP have a good outcome, long-term prognosis of HSP depends on the severity of renal involvement. Previous studies reported that severe abdominal symptoms, older age, and persistent purpura decreased factor XIII activity and increased the risk of renal involvement (4,5). However, no other study except ours investigated whether pre-existing allergic diseases increased risk of HSP with renal involvement. Our data show that allergic diseases do not increase subsequent risks of renal involvement in HSP children.
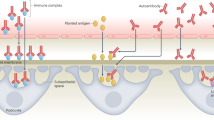
HSP is a systemic form of small-vessel vasculitis characterized by elevated serum IgA levels, vascular deposition of IgA-contained immune complexes, and increased proinflammatory cytokines during the acute stage (17). To date, the exact triggers of the HSP cascade response are not clear. HSP was originally considered a Th1-mediated systemic form of vasculitis with a cytokine cascade and small-vessel endothelial cell lesions in the acute stage (17,18,20). The serum levels of Th1-related cytokines such as IL-1, IL-6, and TNF as well as the soluble IL-2 receptor were found to be elevated during the acute phase of HSP (17,18,20). In contrast, increasing numbers of studies have revealed elevated levels of Th2-related biomarkers in children with HSP (10,11,12,13). Allergic diseases are caused by inappropriate Th2 cell–mediated immunological responses to allergens. Th2 cells secrete IL-4 and IL-5, which lead to type 1 hypersensitivity (21). HSP onset usually follows bacterial or viral infection, drug or food allergy, insect bites, and immunization, suggesting that allergic reactions might participate in disease initiation (15,16). In the presence of IL-4, most B cells undergo immunoglobulin class switching to produce vast amounts of IgE (11,20). Davin et al. (12) reported that the incidence of increased plasma IgE levels relative to age-matched normal values was significantly higher among HSP patients, indicating that IgE may play a pathogenic role in HSP. IL-5 induces B-cell immunoglobulin class switching to IgA and also activates eosinophils (22,23,24). HSP has previously been considered an IgA-mediated vasculitis (1). Namgoong et al. (13) reported that the levels of ECP and soluble IL-2 receptor were significantly higher in HSP patients than in a control group, indicating that T-cell and eosinophil activation are involved in HSP pathogenesis. Tsuji et al. (10) found significantly higher serum IgE and urinary leukotriene E4 levels at HSP onset and also found IgE deposits in Langerhans and mast cells in patients with HSP nephritis. These findings implied that the stimulation of IgE-sensitized mast cells by specific antigens in the presence of IgA circulating immune complexes might increase capillary permeability and perivascular deposition of IgA circulating immune complexes in the small vessels of the skin and renal glomeruli (10,22). Kawasaki et al. (14) found elevated serum IL-4 levels in patients with acute HSP without nephritis as well as IL-5 and ECP-activated eosinophils at the onset of HSP nephritis. These findings support the results from our present study that indicate that pre-existing allergic disease increases subsequent risk of HSP. More recently, Li et al. (20) reported significantly elevated Th2 and Th17 cell levels in acute HSP. The authors concluded that the functional imbalance in Th cell subsets and aberrant Th2 and Th17 activation might be associated with the aberrant antibody production and vasculitis development (20). Epidemiological data reveal that the prevalence of Th1-mediated autoimmune diseases has been rising in parallel with Th2-mediated allergic diseases worldwide over the past two decades (21,22,23). The hygiene hypothesis suggests that early-life infections and microbial exposures may protect against the development of allergy and autoimmune diseases, as a potential explanation for the increased occurrence of allergic and autoimmune diseases (24,25,26). In our previous population-based cohort studies, we found increased incidence and risk of systemic lupus erythematosus (SLE) in children with atopic dermatitis and asthma (27,28). The importance of elevated total serum IgE levels has also been previously highlighted in SLE patients and SLE disease activity (29,30,31). A recent study in a mouse model of SLE and human SLE subjects reported that the activation of the basophils by autoreactive IgE-containing immune complexes amplifies the production of autoantibodies and contributes to the pathogenesis of disease (32). In addition, genetic studies of candidate genes in HSP have been performed in human leukocyte antigen class II gene family, several other genes involved in the inflammatory system, and vasomotor regulation genes (33). A recent meta-analysis suggested that genetic variants in human leukocyte antigen (HLA)-DRB1, and angiotensin-converting enzyme insertion/deletion (ACE I/D) polymorphism, may confer susceptibility for HSP (33). However, HLA-DRB1 has also been reported be associated with allergic asthma (34), and the ACE I/D polymorphism may be a risk factor for AR and asthma (35,36). The abovementioned study corroborates our finding showing that the Th2 environment contributes to the development of autoimmune T1 diseases such as HSP, suggesting that the Th1/Th2 paradigm is oversimplified. Dysregulated immune responses to environmental stimuli and a genetic predisposition or gene interactions may contribute to both allergic and autoimmune diseases (37). Therefore, immunogenetic or early-life environmental factors may all contribute to the development of allergy and HSP.
This study has several limitations. This is a cross-sectional study, and causality could not be established between allergic diseases and HSP. In this study, we attempted to adjust for most potential confounders such as age, sex, urbanization level, season, and allergic diseases that might associate with HSP and atopic diathesis (8). However, there might have been some residual confounding factors such as detailed clinical information that was not collected in the National Health Insurance Research Database (NHIRD) (e.g., serum IgE levels, allergen test results, family history of allergic diseases, environmental exposures, infectious pathogens, and genetic factors). When interpreting these data, residual confounding may need to account for a biased estimate of the association between the risks of HSP and atopic diathesis. In addition, diagnostic accuracy should be considered. In this study, diagnoses of HSP and allergic diseases were made by physicians and identified by ICD-9 codes. Although diagnostic accuracy may vary at the individual clinician level, in Taiwan, a diagnosis of HSP is based on the criteria of American College of Rheumatology and is made by a pediatrician, rheumatologist, and dermatologist. According to the criteria of American College of Rheumatology, diagnostic biopsies, which have a sensitivity of 88% and specificity of 80%, are seldom performed in children (38). This study was conducted in the context of a Chinese population; therefore, our findings cannot be generalized to other populations.
This study adds to the evidence of the associations between allergic disease and its subsequent risk of HSP but not the risk of HSP nephritis. Further research is needed to clarify the pathogenesis of these associations.
Methods
Data Sources
This case–control study used research data from reimbursement claims of the National Health Insurance Program (NHIP) in Taiwan. Implemented in March 1995, the NHIP has covered more than 99% of 23.74 million Taiwan residents. The Taiwan National Health Research Institutes (NHRI) manages the insurance claims data that records all beneficiaries’ medical services, including outpatient and inpatient demographics, diagnoses, procedures, prescriptions, and medical expenditures, for administrative and research uses (39). We used a dataset comprising a randomly selected sample of half of all insured children in Taiwan (39,40). Because of personal electronic data privacy regulations, the insurants’ records are encrypted before being released to researchers; thus, informed consent was not required in this study. This study was approved by the Institutional Review Board at China Medical University Hospital (CMU-REC-101-012). The International Classification of Disease, Ninth Revision (ICD-9) was used to define the diagnostic disease codes.
Study Design and Subjects
The claims data were collected from January 2000 through December 2008 and contained demographic information about insured children (including sex, birth date, and residential area) and medical care received during ambulatory and inpatient visits. Diagnoses of HSP were based on clinical manifestations, including nonthrombocytopenic purpura mostly located on dependent areas such as the lower extremities and buttocks, and at least one of the following findings: arthralgia or arthritis, abdominal pain, or nephritis, according to the 1990 criteria of the American College of Rheumatology. In this study, diagnoses of HSP and allergic diseases were made by physicians and were defined as at least one inpatient claim record or two ambulatory claims in any diagnosis field with respective ICD-9-CM codes. We identified a total of 2,240 patients aged 1–18 y with newly diagnosed HSP (ICD-9 Clinical Modification (ICD-9-CM) code 287.0) from 2000 to 2008; these patients comprised the HSP group. For each HSP patient, four controls with no history of HSP were matched by age (within 1-y intervals), sex, and urbanization level ( Figure 1 ). Urbanization level was defined according to a NHRI report (29). City districts and townships where the subjects were registered for insurance purposes were grouped into seven urbanization levels based on population density (people/km2). Levels 1 and 7 referred to the most and least urbanized areas, respectively. However, as there were few patients in levels 5, 6, and 7, these three levels were combined with level 4. We identified subjects who were diagnosed with allergic conjunctivitis (ICD-9 codes 372.05, 372.10, and 372.14), AR (ICD-9 code 477), asthma (ICD-9 codes 493 and 494), and atopic dermatitis (ICD-9 code: 691.8) before the diagnosis of HSP. HSP nephritis was defined as occurrence of any one of following condition: hematuria, proteinuria, nephritis, nephrosis, and acute or chronic renal failure (ICD-9 codes 599.7, 791.0, and 580–585), within 1 y after HSP diagnosis. The cumulative effect of disease severity was determined based on the number of allergic comorbidities and frequency of health-care utilization.
Flow diagram of participant selection.
Statistical Analysis
We used chi-square and t tests to compare demographic data between the HSP and non-HSP control groups and multivariate logistic regression models to calculate the ORs and 95% CIs for the association between allergic diseases and HSP after adjusting for age, sex, urbanization of residential area, and comorbidities. Additionally, the incidence rates were calculated for each cohort. Hazard ratios and 95% CIs were calculated using multivariable Cox proportional hazard regression models. All data analyses were performed using SAS 9.1 (SAS Institute Inc., Cary, NC), and a P value <0.05 was considered statistically significant.
Statement of Financial Support
The study was supported in part by Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence, Taiwan (MOHW105-TDU-B-212-133019); China Medical University Hospital, Academia Sinica Taiwan Biobank, Stroke Biosignature Project, Taichung, Taiwan (BM104010092); NRPB Stroke Clinical Trial Consortium, Taichung, Taiwan (MOST 103-2325-B-039-006); Tseng-Lien Lin Foundation, Taichung, Taiwan; Taiwan Brain Disease Foundation, Taipei, Taiwan; and Katsuzo and Kiyo Aoshima Memorial Funds, Japan.
Disclosure
The authors have indicated that they have no financial relationships relevant to this article.
References
Saulsbury FT. Clinical update: Henoch–Schonlein purpura. Lancet 2007;369: 976–8.
Gardner-Medwin JMM, Dolezalova P, Cummins C, Southwood TR. Incidence of Henoch–Schönlein, Kawasaki disease and rare vasculitides in children of different ethnic origins. Lancet 2002;360:1197–202.
Stewart M, Savage JM, Bell B, McCord B. Long term renal prognosis of Henoch–Schönlein purpura in an unselected childhood population. Eur J Pediatr 1988;147:113–5.
Kaku Y, Nohara K, Honda S. Renal involvement in Henoch–Schönlein purpura: a multivariate analysis of prognostic factors. Kidney Int 1998;53:1755–9.
Sano H, Izumida M, Shimizu H, Ogawa Y. Risk factors of renal involvement and significant proteinuria in Henoch–Schönlein purpura. Eur J Pediatr 2002;161:196–201.
Rigante D, Candelli M, Federico G, Bartolozzi F, Porri MG, Stabile A. Predictive factors of renal involvement or relapsing disease in children with Henoch–Schönlein purpura. Rheumatol Int 2005;25:45–8.
Chang WL, Yang YH, Wang LC, Lin YT, Chiang BL. Renal manifestations in Henoch–Schönlein purpura: a 10-year clinical study. Pediatr Nephrol 2005;20:1269–72.
Yang YH, Hung CF, Hsu CR, et al. A nationwide survey on epidemiological characteristics of childhood Henoch–Schönlein purpura in Taiwan. Rheumatology (Oxford) 2005;44:618–22.
Yang YH, Chuang YH, Wang LC, Huang HY, Gershwin ME, Chiang BL. The immunobiology of Henoch–Schönlein purpura. Autoimmun Rev 2008;7:179–84.
Tsuji Y, Abe Y, Hisano M, Sakai T. Urinary leukotriene E4 in Henoch–Schonlein purpura. Clin Exp Allergy 2004;34:1259–61.
Kawasaki Y, Hosoya M, Suzuki H. Possible pathologenic role of interleukin-5 and eosino cationic protein in Henoch–Schönlein purpura nephritis. Pediatr Int 2005;47:512–7.
Davin JC, Pierard G, Dechenne C, et al. Possible pathogenic role of IgE in Henoch–Schönlein purpura. Pediatr Nephrol 1994;8:169–71.
Namgoong MK, Lim BK, Kim JS. Eosinophil cationic protein in Henoch–Schönlein purpura and in IgA nephropathy. Pediatr Nephrol 1997;11:703–6.
Sams WM Jr . Human hypersensitivity angiitis, an immune complex disease. J Invest Dermatol 1985;85:1 Suppl:S144–8.
Ackroyd JF. Allergic purpura, including purpura due to foods, drugs and infections. Am J Med 1953;14:605–32.
Burke DM, Jellinek HL. Nearly fatal case of Schoenlein–Henoch syndrome following insect bite. AMA Am J Dis Child 1954;88:772–4.
Besbas N, Saatci U, Ruacan S, et al. The role of cytokines in Henoch Schonlein purpura. Scand J Rheumatol 1997;26:456–60.
Del Vecchio GC, Penza R, Altomare M, et al. Cytokine pattern and endothelium damage markers in Henoch–Schonlein purpura. Immunopharmacol Immunotoxicol 2008;30:623–9.
Capra V, Thompson MD, Sala A, Cole DE, Folco G, Rovati GE. Cysteinyl-leukotrienes and their receptors in asthma and other inflammatory diseases: critical update and emerging trends. Med Res Rev 2007;27:469–527.
Li YY, Li CR, Wang GB, Yang J, Zu Y. Investigation of the change in CD4+ T cell subset in children with Henoch–Schonlein purpura. Rheumatol Int 2012;32:3785–92.
He X, Yu C, Zhao P, et al. The genetics of Henoch–Schönlein purpura: a systematic review and meta-analysis. Rheumatol Int 2013;33:1387–95.
Kontakioti E, Domvri K, Papakosta D, Daniilidis M. HLA and asthma phenotypes/endotypes: a review. Hum Immunol 2014;75:930–9.
Lin H, Lin D, Zheng CQ. Angiotensin-converting enzyme insertion/deletion polymorphism associated with allergic rhinitis susceptibility: evidence from 1410 subjects. J Renin Angiotensin Aldosterone Syst 2014;15:593–600.
Zhang YG, Li XB, Zhang J, et al. The I/D polymorphism of angiotensin-converting enzyme gene and asthma risk: a meta-analysis. Allergy 2011;66:197–205.
The International Study of Asthma and Allergies in Childhood (ISAAC) Steering Committee. Worldwide variation in prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and atopic eczema: ISAAC. Lancet. 1998;351:1225–32.
Masoli M, Fabian D, Holt S, Beasley R ; Global Initiative for Asthma (GINA) Program. The global burden of asthma: executive summary of the GINA Dissemination Committee report. Allergy 2004;59:469–78.
Harjutsalo V, Sjöberg L, Tuomilehto J. Time trends in the incidence of type 1 diabetes in Finnish children: a cohort study. Lancet 2008;371:1777–82.
Bach JF. The effect of infections on susceptibility to autoimmune and allergic diseases. N Engl J Med 2002;347:911–20.
Okada H, Kuhn C, Feillet H, Bach JF. The ‘hygiene hypothesis’ for autoimmune and allergic diseases: an update. Clin Exp Immunol 2010;160:1–9.
Romagnani S. The increased prevalence of allergy and the hygiene hypothesis: missing immune deviation, reduced immune suppression, or both? Immunology 2004;112:352–63.
Wei CC, Lin CL, Tsai JD, Shen TC, Sung FC. Increased Incidence of juvenile onset systemic lupus erythematosus in children with atopic dermatitis. Lupus 2014;23:1494–9.
Wei CC, Lin CL, Shen TC, Tsai JD, Chung CJ, Li TC. Increased incidence of juvenile-onset systemic lupus erythematosus among children with asthma. Pediatr Allergy Immunol 2014;25:374–9.
Laurent J, Lagrue G, Sobel A. Increased serum IgE levels in patients with lupus nephritis. Am J Nephrol 1986;6:413–4.
Elkayam O, Tamir R, Pick AI, Wysenbeek A. Serum IgE concentrations, disease activity, and atopic disorders in systemic lupus erythematosus. Allergy 1995;50:94–6.
Charles N, Hardwick D, Daugas E, Illei GG, Rivera J. Basophils and the T helper 2 environment can promote the development of lupus nephritis. Nat Med 2010;16:701–7.
Charles N, Rivera J. Basophils and autoreactive IgE in the pathogenesis of systemic lupus erythematosus. Curr Allergy Asthma Rep 2011;11:378–87.
Rigante D, Castellazzi L, Bosco A, Esposito S. Is there a crossroad between infections, genetics, and Henoch–Schönlein purpura? Autoimmun Rev 2013;12:1016–21.
Mills JA, Michel BA, Bloch DA, et al. The American College of Rheumatology 1990 criteria for the classification of Henoch–Schönlein purpura. Arthritis Rheum 1990;33:1114–21.
Wei CC, Lin CL, Shen TC, Sung FC. Occurrence of common allergic diseases in children with idiopathic nephrotic syndrome. J Epidemiol 2015;25:370–7.
Wei CC, Chang CH, Lin CL, Chang SN, Li TC, Kao CH. Neonatal jaundice and increased risk of attention-deficit hyperactivity disorder: a population-based cohort study. J Child Psychol Psychiatry 2015;56:460–7.
Author information
Authors and Affiliations
Corresponding author
PowerPoint slides
Rights and permissions
About this article
Cite this article
Chen, AC., Lin, CL., Shen, TC. et al. Association between allergic diseases and risks of HSP and HSP nephritis: a population-based study. Pediatr Res 79, 559–564 (2016). https://doi.org/10.1038/pr.2015.271
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2015.271
This article is cited by
-
Changes of inflammatory mediators and oxidative stress indicators in children with Henoch-Schönlein purpura and clinical effects of hemoperfusion in the treatment of severe Henoch-Schönlein purpura with gastrointestinal involvement in children
BMC Pediatrics (2019)
-
Incidence and risk factors for recurrent Henoch-Schönlein purpura in children from a 16-year nationwide database
Pediatric Rheumatology (2018)
-
Significance of histological crescent formation in patients with IgA vasculitis (Henoch-Schönlein purpura)-related nephritis: a cohort in the adult Chinese population
BMC Nephrology (2018)