Abstract
White matter microstructural changes can be detected with diffusion tensor imaging. It was hypothesized that diffusion parameters in the posterior limb of the internal capsule (PLIC) and corpus callosum (CC) bundles in preterm infants at term equivalent age (TEA) were associated with neurodevelopment at 2 y corrected age. In 67 preterm infants, fiber tracking was performed at TEA for the CC and both PLIC bundles. Volume, length, fractional anisotropy (FA), mean diffusivity, axial diffusivity, and radial diffusivity were determined for the three bundles. These parameters were assessed in relation to outcome on the Bayley Scales of Infant and Toddler Development III. In girls, volume and length of the CC bundle and right PLIC bundle volume were associated with cognition. In boys, volume, FA, mean and radial diffusivity, and length of the left PLIC were associated with fine motor scores. Correction for GA, birth weight, intraventricular hemorrhage, white matter injury, and maternal education did not change the results. Fiber tracking parameters in the PLIC and CC bundles in preterm infants at TEA revealed different associations with neurodevelopment between boys and girls. This study suggested that fiber tracking is a useful method to predict neurodevelopment in preterm infants.
Similar content being viewed by others
Main
Preterm infants are susceptible to brain injury, mainly intraventricular hemorrhages (IVH) and white matter (WM) injury (1). Abnormal MRI findings have been shown to be related to impaired neurodevelopment in preterm infants (2,3). Conventional MRI is commonly performed to assess WM injury. However, outcome has been noted to be abnormal in the absence of abnormalities on conventional MRI (4). Therefore, more objective analysis of (subtle) brain injury may be needed with more advanced methods to understand the underlying pathology and subsequent neurodevelopment in children born prematurely.
Diffusion tensor imaging (DTI) can be used to assess brain connectivity (5). DTI describes the diffusion of water molecules in tissues and is assumed to reflect the direction of the underlying microstructure (6,7). With fiber tracking, it is possible to reconstruct and visualize the underlying linear structure defined by the diffusion tensor (7). WM maturation is accompanied by an increase in fractional anisotropy (FA) and a decrease in apparent diffusion coefficient (ADC) (8). In preterm infants, lower FA and higher ADC values in WM have been described before abnormalities were seen on conventional MRI (9). In addition, the expected increase in FA and decrease in ADC values failed to appear in infants with WM injury (10).
It has been hypothesized that boys and girls show variations in brain development and men should have more numerous, smaller neuronal units; however, they display less neuronal processes compared with females (11). In addition, gender differences in diffusion parameters have been described previously (12,13). Information regarding the relationship between diffusion parameters around term equivalent age (TEA) in preterm infants and neurodevelopment is scarce. Lower FA values in the posterior limb of the internal capsule (PLIC) assessed between birth and TEA in preterm infants were related to an impaired neuromotor development at 18–24 mo (14–16). ADC values in the WM illustrated a negative relationship with the Griffiths' developmental quotient at 2 y (17). In most studies, diffusion parameters were assessed in manually drawn regions of interest (ROIs), and only few studies examined entire WM bundles (18–20).
It was investigated whether fiber tracking parameters, i.e. diffusion parameters and volume and length of WM bundles passing through the PLIC and the corpus callosum (CC), in preterm infants at TEA were causally associated with neurodevelopment at 2 y corrected age, which was assessed with the Bayley Scales of Infant and Toddler Development, Third Edition (BSITD-III).
METHODS
Newborns admitted to our level three NICUs with a GA < 31 wk and who reached TEA between January 2007 and July 2008 were recruited for a prospective preterm cohort study performed in the Wilhelmina Children's Hospital in Utrecht, The Netherlands. Neonates with either dysmorphic features or an infection of their CNS were excluded. Of the 175 consecutively admitted neonates, 22 died in the neonatal period, no parental consent was obtained for 14 neonates, and 15 neonates were examined on a 1.5-Tesla system. MRI of the brain was acquired around TEA. Written informed parental consent was obtained for all included infants. This study was approved by the Medical Ethics Committee of the University Medical Center Utrecht.
Cranial ultrasound and MRI.
Cerebral ultrasound was performed daily in the first week after birth and then once a week until discharge. They were evaluated by two independent researchers, who were unaware of the MRI findings and neurodevelopment. Periventricular leukomalacia (PVL) and the presence of an IVH were scored according to de Vries et al. (21).
MRIs were performed on a 3.0-Tesla MR system (Achieva, Philips Medical Systems, Best, The Netherlands) using a eight-channel Sense head coil. The infants were sedated with 50–60 mg/kg oral chloralhydrate. Heart rate, transcutaneous oxygen saturation, and respiratory rate were monitored during scanning. For hearing protection, Minimuffs (Natus Medical Incorporated, San Carlos, CA) were used. A neonatologist was present throughout the examination.
The protocol contained conventional sagittal T1-weighted imaging (repetition time (TR) = 886 ms; echo time (TE) = 15 ms; slice thickness = 3.0 mm), axial 3D T1-weighted imaging (TR = 9.4 ms; TE = 4.6 ms; slice thickness = 2.0 mm), and axial T2-weighted imaging (TR = 6293 ms; TE = 120 ms; slice thickness = 2.0 mm). DTI was performed using a single-shot EPI sequence with diffusion gradients in 32 directions (TR = 7745 ms; TE = 48 ms; voxel size = 1.41 × 1.41 × 2 mm; number of slices = 50; FOV = 180 mm RL; scan matrix = 128; fold-over direction = AP; b value = 0 and 800 s/mm2).
MRIs were evaluated independently by two neonatologists with a special interest in neuroimaging, blinded to the results of the neurodevelopmental assessment. In case of disagreement, a third reader was consulted to achieve consensus. WM injury was scored as published previously [adjusted from Woodward et al. (22)]. The WM score varies between 5 (normal) and 15 (severely abnormal) and was applied in the analysis as an indicator for WM injury.
DTI.
In 118/124 (95.2%) neonates, DTI was performed at TEA. In the other six infants, DTI could not be performed due to time constraints. The quality of the DTI and the result of the tractography were assessed independently by two researchers and when needed, consensus was reached using the opinion of a third researcher. DTI data were analyzed using an in-home developed fiber tracking program (23). Twenty-one datasets were excluded because of large artifacts in the FA colormaps, suggestive of motion, and three datasets because of sense artifacts. In the remaining 94 neonates, tractography was performed. The tracts through the left and right PLIC (denoted as “PLIC bundle”) and the CC (denoted as “CC bundle”) were identified primarily by the color-coded FA map. The PLIC and the CC bundles were chosen a priori because those structures are two of the most developed regions in the newborn brain and known to be affected by premature birth (24,25). For both PLIC bundles, one ROI was placed on an axial slice at the level of the foramen of Monro and the second on the adjacent slice above this landmark. For the CC bundle, two ROIs were placed on sagittal slices around the midplane of the CC. All fibers passing through both ROIs were traced. Fiber tracking was based on a line propagation technique, starting in every voxel in the brain and stopping for the “case linear ” anisotropy index (Cl) < 0.12 [Cl = (λ1 − λ2)/(λ1 + λ2 + λ3)] and maximum angle α > 10°. Tracts were excluded from the analysis when it was visually estimated that more than 10% of the volume was artificially traced, e.g. due to corrupted data caused by motion. In 69/94 (73.4%) neonates, both PLIC and CC bundles were traced correctly and eligible for assessment. For these traced bundles, different fiber tracking parameters were extracted, i.e. mean FA, mean diffusivity (MD, in 10−3 mm2/s), axial diffusivity (AD, λ1; in 10−3 mm2/s), radial diffusivity [RD, (λ2 + λ3)/2; in 10−3 mm2/s], and volume (in mm3) and length (in mm) of the fiber bundles (25). The volume of the bundle was defined as the volume of all voxels through which one or more fibers passed, and the average length of the bundle was defined as the average length of all fibers included in the bundle.
Neurodevelopmental outcome.
At 2 y corrected age (mean 24.2 ± 0.6 mo), all children were assessed with the BSITD-III by a single developmental specialist who was blinded to the MRI findings (26). Only the cognitive and fine and gross motor subtests were used and not the language subtest due to the limited time the child was able to concentrate during one session. Both scaled scores of the three subtests as well as the cognitive and total motor composite scores were calculated corrected for prematurity (mean in a normative population: 10 ± 3 and 100 ± 15, respectively).
Data analysis and statistics.
SPSS version 15 was used for the analysis. Linear regression was used to assess the relationship between the fiber tracking parameters and neurodevelopmental outcome. GA, birth weight (BW) Z score, WM score, IVH, and maternal education were considered to be possible confounders. In the multivariable linear regression analyses, it was assessed whether the associations between the fiber tracking parameters and neurodevelopment remained statistical significant after correction for the neonatal and maternal confounders. Analyses were performed for the total study population and additionally for boys and girls separately. In the analysis, DTI parameters were corrected for postmenstrual age at time of the scan. A p-value < 0.05 was considered statistically significant.
RESULTS
Of the 69 infants with evaluable fiber tracts in both PLIC and CC bundles, two infants were lost to follow-up at the corrected age of 2 y. Consequently, 67/69 infants were evaluated in this study (Table 1). There were no differences in neonatal parameters between boys and girls and between the children included in this study and children who were excluded because 3.0-Tesla DTI data were not eligible for evaluation. However, the excluded children were scanned at an slightly older age than the children in this study (41.9 and 41.5 wk, respectively).
Cranial ultrasound and conventional MRI findings.
During the neonatal period, 35/67 (52.2%) neonates had PVL grade I on sequential cranial ultrasound examinations; however, cystic evolution was not seen in any of the infants. Eighteen infants were diagnosed to have a germinal matrix hemorrhage-IVH (Table 1). Three neonates developed posthemorrhagic ventricular dilatation requiring intervention. The median WM score based on the MRI was 8 [range: 5 (normal) to 12 (moderately abnormal)].
Fiber tracking parameters.
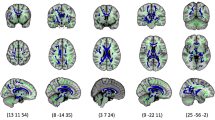
Figure 1 shows a representative example of fiber tracking in the whole neonatal brain. The results of the fiber tracking parameters are illustrated in Table 2.
Example of fiber tracking in the neonatal brain at TEA. View from front left. Fiber tracking was performed using stopping criteria Cl < 0.12 and α > 10°, starting in every voxel in the brain, displaying only fibers longer than 20 mm. The 3D fiber tracking image is superimposed on a 2D ADC map. Color coding: red are fibers from left to right (e.g. corpus callosum), blue from cranial to caudal (corticospinal tract), and green from anterior to posterior.
In boys, the average length was longer and the volume was larger for the CC bundle and left PLIC bundle compared with girls. However, only the differences in CC bundle length and PLIC bundle volume were statistically significant (p = 0.014 and p = 0.031, respectively).
Neurodevelopmental outcome.
Table 3 shows the results for the cognitive, fine motor, gross motor, and total motor scores on the BSITD-III. The infants scored significantly better on the fine motor subtests than on the gross motor subtests (p < 0.001). There were no children who developed CP or had other major motor deficits at 2 y corrected age. ANOVA showed a main effect of maternal education on cognitive scores. Subsequent post hoc analyses showed that both infants of a mother with a low education or with middle education scored significantly poorer than infants of a mother who attended high education (p = 0.019 and p = 0.023, respectively). Girls demonstrated better cognitive scores than boys (p = 0.045).
Fiber tracking parameters and neurodevelopment.
Assessing the total study cohort, no relationships could be demonstrated between the fiber tracking parameters at term and neurodevelopment after correction for GA, BW Z score, WM score, IVH, and maternal education. However, boys and girls showed different associations between the diffusion parameters and neurodevelopment after correction for the neonatal variables and maternal education, as detailed below (Table 4 and Fig. 2).
Corpus callosum bundle.
Better cognitive and fine motor scores were seen in girls with larger CC bundle volume or bundle length at TEA (cognition: p < 0.01; fine motor score: p < 0.05). In boys, an association was found between the MD, AD, and RD of the CC bundle and gross motor performance (p ≤ 0.05). After correction for GA, BW Z score, WM score, IVH, and maternal education, only the associations in girls between cognition and CC bundle volume and length remained statistically significant (Table 4). No other relationships could be demonstrated between fiber tracking parameters in the CC bundle and cognition, fine motor, and gross motor scores.
Left PLIC bundle.
Fiber tracking parameters in the left PLIC bundle were related to neurodevelopmental outcome only in boys. Better fine motor outcome was demonstrated in boys with a larger bundle volume, FA, and bundle length and lower MD, AD, and RD of the left PLIC bundle (p < 0.01). Gross motor performance was associated with MD, AD, RD, and length of the left PLIC bundle (p ≤ 0.05). After correction for GA, BW Z score, WM score, IVH, and maternal education, only associations between fine motor scores and fiber tracking parameters in the left PLIC bundle were demonstrated, showing a more significant relationship with volume, FA, and bundle length than with diffusivity (volume, FA, and length: p ≤ 0.055; MD, AD, and RD p = 0.07–0.14; Table 4). No other relationships could be demonstrated between fiber tracking parameters in the left PLIC bundle and cognition, fine motor, and gross motor scores.
Right PLIC bundle.
In girls, a positive association was found between right PLIC bundle volume and cognition (p = 0.052) and a negative relationship between the length of the right PLIC bundle and gross motor outcome (p = 0.019). In boys, a negative association was seen between the MD and RD of the right PLIC and fine motor outcome (p ≤ 0.05). However, only the association between right PLIC bundle volume and cognition in girls remained statistical significant after correction for GA, BW Z score, WM score, IVH, and maternal education (p < 0.001; Table 4). No other relationships could be demonstrated between fiber tracking parameters in the right PLIC bundle and cognition, fine motor, and gross motor scores.
DISCUSSION
In this study, fiber tracking parameters in the CC bundle and both PLIC bundles at TEA were assessed in relation to neurodevelopment at 2 y corrected age in preterm infants born before 31 wk gestation. It appeared that both size of the bundles (i.e. length and volume) and their microstructural differences were related with outcome at 2 y corrected age. In girls, DTI parameters in the CC bundle demonstrated more associations with both cognition and motor outcome compared with parameters in both PLIC bundles. Nevertheless, in boys, most relationships were seen between DTI parameters in the left PLIC bundle and motor outcome.
To the best of our knowledge, this was the first study assessing fiber tracking parameters in the CC and PLIC bundles in preterm infants at TEA in relation to neurodevelopmental outcome. Fiber tracking in newborns is a major challenge because the signal to noise ratio is relatively small due to the smaller voxel size needed as a consequence of the smaller anatomic structures. Moreover, the higher water content and the lower degree of myelination result in lower FA values compared with adults (1,27). Nevertheless, we were able to implement this technique in a large sample of preterm infants at TEA. For girls, we observed higher CC bundle volume and bundle length at TEA in relation to better cognitive outcome at 2 y corrected age, which is in agreement with the literature. Only in female adults born prematurely, larger callosal size was demonstrated to be associated with better cognitive performance (28). In addition, Kontis et al. (29) displayed that only in females, higher MD in the genu of the CC was associated with lower performance IQ in prematurely born young adults. It has been hypothesized that the greater interhemispheric connectivity in females facilitates cognitive performance while the processing is more bilateral. In contrast, males show a greater lateralization of cognitive functioning and therefore the interhemispheric connectivity may not affect their cognitive capacities (28).
In the present study, fiber tracking parameters in the PLIC bundles were associated with motor outcome. These findings are in agreement with previous studies using DTI. Reduced FA values in the PLIC have been displayed in preterm infants at TEA compared with term born controls, and lower FA values assessed between childhood and (pre)adolescence were related to abnormal neuromotor outcome (14–16,30,31). In general, it is expected that a more mature brain, resulting in higher FA and Cl values due to more advanced myelination, is related to a better performance. With this fiber tracking method, higher Cl gives rise to longer length of the fiber bundles, as the anisotropy index Cl < 0.12 was a stopping criteria.
Boys demonstrated a larger volume and length of the CC and left PLIC bundle compared with girls, although only the differences in CC bundle length and left PLIC bundle volume were statistically significant. In preterm infants, larger gray and WM volumes at TEA were exhibited in preterm males compared with preterm females (32,33). Larger brain volumes seen in men could (partly) explain that we found larger volumes and length of the CC and PLIC in boys in the present study.
In this study, boys and girls displayed different associations between DTI parameters in the CC and PLIC bundles and neurodevelopment. It has been hypothesized that boys and girls demonstrate variations in brain development. Vasileiadis et al. revealed that girls had a larger cortical folding area compared with boys with similar brain volumes. They declared that the more “compact ” female brain provided evidence for an early sexually dimorphic brain development (32). The gender differences in brain morphogenesis considering the tension-based theory (34) could follow underlying microstructural variations detected, e.g. as tissue anisotropy. Variations in diffusion parameters reflect WM maturation, which depends on fiber organization, density, diameter, and myelination (5). Schmithorst et al. presented developmental differences in WM microstructure in healthy term born boys and girls at a mean age of 12 y. They hypothesized that a more constrained brain volume in females could be related to a lower fiber density. Therefore, girls may have a greater dependence on brain connectivity with more crossing fibers, resulting in lower FA and higher ADC values (12).
Several studies support the hypothesis that sexual dimorphism in brain development could be related to differences in neurodevelopment. In healthy adults, women demonstrated a relationship between intelligence and both WM and callosal volume, whereas cognition in males was more related to gray matter volume (28,35). In preterm infants, sex differences in cerebral gray and WM were displayed at 8 y of age; however, only prematurely born girls demonstrated a positive correlation between gray matter/total brain volume ratio and cognitive measurements (36). Male gender has been shown to be a relevant risk factor for an adverse neurodevelopmental outcome (37). This may be aggravated by a more serious neonatal course. Moreover, in the present study, boys appeared to be sicker than girls, showing significantly lower Apgar scores and a longer ventilation period. The male-female differences in the associations between neuroanatomical variables, i.e. fiber tracking parameters, and neurodevelopmental outcome found in the present study may be related to differences in brain development and subsequent outcome between prematurely born boys and girls. Our results may indicate that the sexual dimorphism in WM maturation is already detectable in newborns, which supports the suggestion that it is important to take gender into account in DTI developmental studies (12).
This study is subject to several limitations. Fiber tracking in the neonatal brain is difficult due to the low degree of myelination and the high water content compared with the adult brain. Using fiber tracking, we were only able to assess the most mature WM structures. A remark has to be made regarding the limited DTI data available for this study. We were able to analyze only the DTI data of 59% of the infants in our study cohort. DTI is inherently sensitive to artifacts due to small patient movements. Although the infants were sedated and noise reduction was administered, it could not be prevented that some infants woke up during the DTI sequence, which was at the end of our 30-min scanning protocol. Nevertheless, in our relatively large study cohort, consisting of 67 preterm infants, we were able to display different associations for boys and girls between fiber tracking parameters at TEA and neurodevelopmental outcome at 2 y corrected age. In this study, we used fiber tracking parameters as “readout ” for the neonatal period. Further research will be needed to determine the role of different potential risk factors for changes in the WM microstructure. Next, the BSITD-III reveals information concerning general cognitive, fine, and gross motor skills. More specific evaluation of tasks, which are processed by the CC and PLIC bundles, could have shown additional details to predict neurodevelopment in preterm infants. Finally, our results are limited to preterm infants due to the lack of a term born control group.
In conclusion, fiber tracking parameters at TEA in the CC and PLIC bundles were associated with neurodevelopmental outcome at 2 y corrected age. In female preterm infants, the CC bundle was the most important WM structure showing an association with cognitive, fine motor, and gross motor performance. In boys, fiber tracking parameters in the left PLIC bundle were related to fine and gross motor function.
Abbreviations
- AD:
-
axial diffusivity
- ADC:
-
apparent diffusion coefficient
- BSITD-III:
-
Bayley Scales of Infant and Toddler Development; Third Edition
- BW:
-
birth weight
- CC:
-
corpus callosum
- Cl:
-
“case linear” anisotropy index
- DTI:
-
diffusion tensor imaging
- FA:
-
fractional anisotropy
- IVH:
-
intraventricular hemorrhages
- MD:
-
mean diffusivity
- PLIC:
-
posterior limb of the internal capsule
- RD:
-
radial diffusivity
- ROI:
-
region of interest
- TE:
-
echo time
- TEA:
-
term equivalent age
- TR:
-
repetition time
- WM:
-
white matter.
References
Volpe JJ 2008 Neurology of the Newborn. Saunders/Elsevier, Philadelphia
Dyet LE, Kennea N, Counsell SJ, Maalouf EF, Ajayi-Obe M, Duggan PJ, Harrison M, Allsop JM, Hajnal J, Herlihy AH, Edwards B, Laroche S, Cowan FM, Rutherford MA, Edwards AD 2006 Natural history of brain lesions in extremely preterm infants studied with serial magnetic resonance imaging from birth and neurodevelopmental assessment. Pediatrics 118: 536–548
Spittle AJ, Boyd RN, Inder TE, Doyle LW 2009 Predicting motor development in very preterm infants at 12 months' corrected age: the role of qualitative magnetic resonance imaging and general movements assessments. Pediatrics 123: 512–517
Mirmiran M, Barnes PD, Keller K, Constantinou JC, Fleisher BE, Hintz SR, Ariagno RL 2004 Neonatal brain magnetic resonance imaging before discharge is better than serial cranial ultrasound in predicting cerebral palsy in very low birth weight preterm infants. Pediatrics 114: 992–998
Hüppi PS, Dubois J 2006 Diffusion tensor imaging of brain development. Semin Fetal Neonatal Med 11: 489–497
Basser PJ, Mattiello J, LeBihan D 1994 MR diffusion tensor spectroscopy and imaging. Biophys J 66: 259–267
Mori S, van Zijl PC 2002 Fiber tracking: principles and strategies—a technical review. NMR Biomed 15: 468–480
Ment LR, Hirtz D, Huppi PS 2009 Imaging biomarkers of outcome in the developing preterm brain. Lancet Neurol 8: 1042–1055
Mathur AM, Neil JJ, Inder TE 2010 Understanding brain injury and neurodevelopmental disabilities in the preterm infant: the evolving role of advanced magnetic resonance imaging. Semin Perinatol 34: 57–66
Miller SP, Vigneron DB, Henry RG, Bohland MA, Ceppi-Cozzio C, Hoffman C, Newton N, Partridge JC, Ferriero DM, Barkovich AJ 2002 Serial quantitative diffusion tensor MRI of the premature brain: development in newborns with and without injury. J Magn Reson Imaging 16: 621–632
Rabinowicz T, Petetot JM, Gartside PS, Sheyn D, Sheyn T, de CM 2002 Structure of the cerebral cortex in men and women. J Neuropathol Exp Neurol 61: 46–57
Schmithorst VJ, Holland SK, Dardzinski BJ 2008 Developmental differences in white matter architecture between boys and girls. Hum Brain Mapp 29: 696–710
Wilde EA, McCauley SR, Chu Z, Hunter JV, Bigler ED, Yallampalli R, Wang ZJ, Hanten G, Li X, Ramos MA, Sabir SH, Vasquez AC, Menefee D, Levin HS 2009 Diffusion tensor imaging of hemispheric asymmetries in the developing brain. J Clin Exp Neuropsychol 31: 205–218
Arzoumanian Y, Mirmiran M, Barnes PD, Woolley K, Ariagno RL, Moseley ME, Fleisher BE, Atlas SW 2003 Diffusion tensor brain imaging findings at term-equivalent age may predict neurologic abnormalities in low birth weight preterm infants. AJNR Am J Neuroradiol 24: 1646–1653
Drobyshevsky A, Bregman J, Storey P, Meyer J, Prasad PV, Derrick M, MacKendrick W, Tan S 2007 Serial diffusion tensor imaging detects white matter changes that correlate with motor outcome in premature infants. Dev Neurosci 29: 289–301
Rose J, Butler EE, Lamont LE, Barnes PD, Atlas SW, Stevenson DK 2009 Neonatal brain structure on MRI and diffusion tensor imaging, sex, and neurodevelopment in very-low-birthweight preterm children. Dev Med Child Neurol 51: 526–535
Krishnan ML, Dyet LE, Boardman JP, Kapellou O, Allsop JM, Cowan F, Edwards AD, Rutherford MA, Counsell SJ 2007 Relationship between white matter apparent diffusion coefficients in preterm infants at term-equivalent age and developmental outcome at 2 years. Pediatrics 120: e604–e609
Yoo SS, Park HJ, Soul JS, Mamata H, Park H, Westin CF, Bassan H, du Plessis AJ, Robertson RL Jr, Maier SE, Ringer SA, Volpe JJ, Zientara GP 2005 In vivo visualization of white matter fiber tracts of preterm- and term-infant brains with diffusion tensor magnetic resonance imaging. Invest Radiol 40: 110–115
Zhai G, Lin W, Wilber KP, Gerig G, Gilmore JH 2003 Comparisons of regional white matter diffusion in healthy neonates and adults performed with a 3.0-T head-only MR imaging unit. Radiology 229: 673–681
Berman JI, Mukherjee P, Partridge SC, Miller SP, Ferriero DM, Barkovich AJ, Vigneron DB, Henry RG 2005 Quantitative diffusion tensor MRI fiber tractography of sensorimotor white matter development in premature infants. Neuroimage 27: 862–871
de Vries LS, van Haastert IC, Rademaker KJ, Koopman C, Groenendaal F 2004 Ultrasound abnormalities preceding cerebral palsy in high-risk preterm infants. J Pediatr 144: 815–820
Woodward LJ, Anderson PJ, Austin NC, Howard K, Inder TE 2006 Neonatal MRI to predict neurodevelopmental outcomes in preterm infants. N Engl J Med 355: 685–694
Vilanova A, Berenschot G, van Pul C 2004 DTI visualization with streamsurfaces and evenly-spaced volume seeding. In: Deussen O, Hansen C, Keim DA, Saupte D (eds) Joint EUROGRAPHICS—IEEE TCVG Symposium on Visualization, pp 173–182. Eurographics Association, Konstanz, Germany
Gilmore JH, Lin W, Corouge I, Vetsa YS, Smith JK, Kang C, Gu H, Hamer RM, Lieberman JA, Gerig G 2007 Early postnatal development of corpus callosum and corticospinal white matter assessed with quantitative tractography. AJNR Am J Neuroradiol 28: 1789–1795
van Pul C, Buijs J, Vilanova A, Roos FG, Wijn PF 2006 Infants with perinatal hypoxic ischemia: feasibility of fiber tracking at birth and 3 months. Radiology 240: 203–214
Bayley N 2006 Bayley Scales of Infant and Toddler Development, 3rd ed. Pearson Education, San Antonio
Hüppi PS, Warfield S, Kikinis R, Barnes PD, Zientara GP, Jolesz FA, Tsuji MK, Volpe JJ 1998 Quantitative magnetic resonance imaging of brain development in premature and mature newborns. Ann Neurol 43: 224–235
Davatzikos C, Resnick SM 1998 Sex differences in anatomic measures of interhemispheric connectivity: correlations with cognition in women but not men. Cereb Cortex 8: 635–640
Kontis D, Catani M, Cuddy M, Walshe M, Nosarti C, Jones D, Wyatt J, Rifkin L, Murray R, Allin M 2009 Diffusion tensor MRI of the corpus callosum and cognitive function in adults born preterm. Neuroreport 20: 424–428
Nagy Z, Westerberg H, Skare S, Andersson JL, Lilja A, Flodmark O, Fernell E, Holmberg K, Bohm B, Forssberg H, Lagercrantz H, Klingberg T 2003 Preterm children have disturbances of white matter at 11 years of age as shown by diffusion tensor imaging. Pediatr Res 54: 672–679
Skranes J, Vangberg TR, Kulseng S, Indredavik MS, Evensen KA, Martinussen M, Dale AM, Haraldseth O, Brubakk AM 2007 Clinical findings and white matter abnormalities seen on diffusion tensor imaging in adolescents with very low birth weight. Brain 130: 654–666
Vasileiadis GT, Thompson RT, Han VK, Gelman N 2009 Females follow a more “compact” early human brain development model than males. A case-control study of preterm neonates. Pediatr Res 66: 551–555
Gilmore JH, Lin W, Prastawa MW, Looney CB, Vetsa YS, Knickmeyer RC, Evans DD, Smith JK, Hamer RM, Lieberman JA, Gerig G 2007 Regional gray matter growth, sexual dimorphism, and cerebral asymmetry in the neonatal brain. J Neurosci 27: 1255–1260
Van Essen DC 1997 A tension-based theory of morphogenesis and compact wiring in the central nervous system. Nature 385: 313–318
Haier RJ, Jung RE, Yeo RA, Head K, Alkire MT 2005 The neuroanatomy of general intelligence: sex matters. Neuroimage 25: 320–327
Reiss AL, Kesler SR, Vohr B, Duncan CC, Katz KH, Pajot S, Schneider KC, Makuch RW, Ment LR 2004 Sex differences in cerebral volumes of 8-year-olds born preterm. J Pediatr 145: 242–249
Wood NS, Costeloe K, Gibson AT, Hennessy EM, Marlow N, Wilkinson AR 2005 The EPICure study: associations and antecedents of neurological and developmental disability at 30 months of age following extremely preterm birth. Arch Dis Child Fetal Neonatal Ed 90: F134–F140
Acknowledgements
We thank Dr. Cuno S.P.M. Uiterwaal for his support in the statistical analysis; the families who took part in the study; Niels Blanken and the other MR technicians of the MR institute for their expert and enthusiastic help; and our colleagues of the NICU and Medium care of the Wilhelmina Children's Hospital in Utrecht, The Netherlands.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by The Netherlands Organization for Health Research and Development, project 94527022.
The authors report no conflicts of interest.
Rights and permissions
About this article
Cite this article
van Kooij, B., van Pul, C., Benders, M. et al. Fiber Tracking at Term Displays Gender Differences Regarding Cognitive and Motor Outcome at 2 Years of Age in Preterm Infants. Pediatr Res 70, 626–632 (2011). https://doi.org/10.1203/PDR.0b013e318232a963
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/PDR.0b013e318232a963
This article is cited by
-
Diffusion Tensor Tractography of the Cerebellar Peduncles in Prematurely Born 7-Year-Old Children
The Cerebellum (2017)
-
Neonatal brain microstructure correlates of neurodevelopment and gait in preterm children 18–22 mo of age: an MRI and DTI study
Pediatric Research (2015)
-
Data quality in diffusion tensor imaging studies of the preterm brain: a systematic review
Pediatric Radiology (2015)
-
A crucial role for white matter alterations in interference control problems of very preterm children
Pediatric Research (2014)
-
Isolated mild white matter signal changes in preterm infants: a regional approach for comparison of cranial ultrasound and MRI findings
Journal of Perinatology (2014)