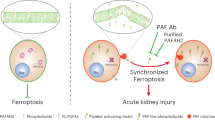
Abstract
Platelet-activating factor (PAF) is a proinflammatory phospholipid mediator implicated in necrotizing enterocolitis. Regulation of PAF-acetylhydrolase (AH), the enzyme degrading PAF, is poorly understood. In this study we found that administration of a dose of PAF (1.5 μg/kg, i.v.), which does not cause gross intestinal injury, increased plasma and intestinal PAF-AH in the rat. Cycloheximide (CHX, 5 mg/kg, i.v.) reduced the activity of plasma (but not intestinal tissue) AH in control, as well as in PAF-injected rats, and aggravated systemic inflammation and tissue injury in the latter. The intestinal necrosis induced by PAF and CHX was ameliorated by posttreatment with WEB2170 (a PAF antagonist), indicating a role of endogenous PAF in mediating injury. Both WEB2170 and anti-TNF antibody reduced PAF-induced AH activity in intestinal tissue, but not in the plasma. Allopurinol largely prevented the injury induced by PAF and CHX, but had no effect on the up-regulation of AH. We conclude: 1) de novo protein synthesis is required to maintain physiologic AH level in the plasma; 2) PAF up-regulates plasma and intestinal AH activity; 3) CHX enhances the injurious effect of PAF; 4) endogenous PAF and TNF also play a role in the up-regulation of intestinal AH; the former probably mediating the intestinal injury by PAF; and 5) reactive oxygen species may mediate the injurious effect of PAF plus CHX, but do not contribute to the regulation of AH by PAF.
Similar content being viewed by others
Main
PAF (PAF-acether) is a potent phospholipid mediator with a multitude of proinflammatory effects(1, 2) and is associated with various inflammatory diseases of the gastrointestinal tract, such as ulcerogenesis(3), inflammatory bowel disease(4), and NEC(5). The basal PAF level in the plasma and tissues of various organs in normal rats is low(6, 7). Agents or stimuli that induce intestinal injury such as PAF(8), lipopolysaccharide(9, 10), TNF(11, 12), and hypoxia(13), raise the level of PAF in serum and tissues; the injury is prevented by PAF antagonists(14), suggesting that endogenous PAF plays a central role in intestinal injury.
The plasma and tissue concentration of PAF is determined by a balance between its biosynthesis and degradation. The degradation of PAF is governed by a PAF-specific AH, or PAF 2-AH (EC 3.1.1.47), which removes the acetyl moiety from the sn-2 position of the glycerol backbone of the molecule. The importance of this enzyme on the regulation of the inflammatory response has recently been demonstrated by its prevention of vascular leakage in pleurisy and paw edema in animal models(15). Neonates and premature infants have a much lower plasma AH level, compared with infants more than 4 mo of age(16), and patients who had developed NEC were especially apt to have suppressed AH, compared with infants of the same gestational age(5). These observations may account for the enhanced susceptibility of these infants to NEC. The risk for NEC is reported to be lower in breast fed-babies, possibly due to the presence of AH in the maternal milk(17, 18). The physiologic regulation of PAF-AH is incompletely understood. Antiinflammatory steroids (such as dexamethasone), which block PAF-induced intestinal injury, are also reported to increase plasma PAF-AH activity(19). A recent report showed that PAF stimulated PAF-AH generation in hepatocytes(20, 21), implicating a potential clearance mechanism of PAF in vivo. However, the in vivo regulation of PAF-AH in physiologic and pathologic conditions is inadequately studied. The intestinal damage caused by PAF in rats is dose-dependent. PAF at high doses (2.5-3 μg/kg, i.v.) causes a systemic inflammatory response and severe intestinal necrosis, whereas at low doses (1.0-1.5 μg/kg, i.v.), it does not cause gross intestinal injury(14), but induces intestinal production of proinflammatory factors such as TNF(22) and type II phospholipase A2(23).
In the first part of this study, we injected low dose PAF into rats and examined 1) Whether PAF has a regulatory effect on PAF-AH activity in vivo; 2) whether a difference exists in the response of PAF-AH in plasma and in intestinal tissue, because the sources of PAF-AH in the plasma and gastrointestinal tissue are reportedly different(15, 24); and 3) whether CHX, a protein synthesis inhibitor, which blocks PAF-AH production by macrophages(25) and HL-60 cells(26), inhibits PAF-AH in vivo and aggravates PAF-induced tissue injury. Because we observed that low dose PAF and CHX synergized to cause intestinal damage, we investigated, in the second part of this study, whether the injury is related to a suppressed PAF-AH activity in tissue or plasma. Furthermore, we examined whether inflammatory mediators involved in the pathogenesis of PAF-induced intestinal damage, such as TNF, endogenous PAF, and reactive oxygen species generated from XO, participate in PAF-AH regulation.
METHODS
Materials. PAF (1-O-hexadecyl-2-acetyl-sn-glycero-3-phosphocholine). CHX, allopurinol, and human leukocyte MPO were purchased from Sigma Chemical Co. (St. Louis, MO). PAF was prepared fresh in saline containing 5 mg/mL BSA. 1-O-[3H]Alkyl-2-acetyl-3-phosphorylcholine was obtained from Amersham Corp. (Arlington Height, IL). WEB2170, a PAF antagonist, was a gift from Dr. H. Heuer (Boerhinger Ingelheim, Mainz, Germany). Rabbit anti-mouse TNF antibody, which cross-reacts with rat's TNF, was generated in Dr. D. G. Remick's laboratory. The antibody was diluted 1:1 with saline before injection.
Animal experiment. Young male Sprague-Dawley rats (60-100 g) were anesthetized with Nembutal (65 mg/kg, i.p.) and placed a under warm light. The carotid artery and jugular vein were catheterized for continuous blood pressure recording, drug injection, and blood sampling. The first part of the study includes four groups: 1) sham operation, vehicle (saline) only, 1 mL/kg, i.v.; 2) CHX (5 mg/kg, i.v.); 3) PAF (1.5 μg/kg, i.v.); and 4) CHX, 30 min before PAF. The second part of the study includes three additional groups: 5) CHX, 30 min before PAF, and rabbit anti-mouse TNF antibody (10 mL/kg, i.p.), 20 min before PAF; 6) CHX 30 min before PAF, and allopurinol, 20 min (10 mg/kg, i.p.) after PAF; 7) CHX, 30 min before PAF, and WEB2170 (1 mg/kg, i.v.), 20 min after PAF. WEB2170 was injected after PAF to inhibit endogenously produced PAF. The time point (20 min) for WEB2170 injection was established based on preliminary experiments (n = 4), which determined the half-life of PAF.
In all experiments, PAF was injected at time 0, and blood samples were collected at -30, 0, 15, 30, 60, and 90 min for white blood cell count and Hct. The animals were killed at 90 min after PAF injection, and the small intestine was removed and rinsed with ice-cold saline to remove the intestinal content. The intestine was then examined for gross injury. The severity of gross injury was scored as follows(27): 1, mild (slight reddish discoloration); 2, moderate (red discoloration often with hemorrhage); and 3, severe (grossly necrotic, blackish red, and friable). Representative sections were taken from the abnormal areas, fixed in 10% buffered formalin, and processed for histologic examination. If no injury is found grossly, multiple random sections were taken for histologic confirmation. The histologic changes were scored in a blinded fashion as follows: 0, no injury with intact surface epithelium; 0.5, minimal injury with separation of the surface epithelium from the lamina propria; 1, mild mucosal injury involving the top one-half of the villi only; 2, moderate injury involving more than one-half of the length of the villi; 3, severe injury extending beyond the mucosal layer. The remaining intestinal tissue was frozen in liquid nitrogen. Blood samples were collected before and at the end of the experiment for assay of plasma PAF-AH and for differential white blood cell counts. The animal protocol was approved by the Institutional Animal Care and Use Committee.
Plasma and tissue PAF-AH assay. PAF-AH activity was assayed as previously described(13) with minor modification. Fresh plasma samples were diluted 1:4 with HEPES buffer (0.1 M, pH 7.2). The intestinal tissue was homogenized in 0.1 M HEPES buffer (pH 7.2, 10 mL/g tissue) on ice for 1 min and centrifuged at 15 000 × g for 15 min, and the supernatant was diluted 1:4 with HEPES buffer (pH 7.2). The PAF-AH activity was assayed as follows: 50 μL of reaction mixture containing 2 × 105 dpm of 1-O-[3H]alkyl-2-acetyl-3-phosphorylcholine, 80 μM PAF, 0.1 M HEPES buffer (pH 7.2), and 10 μL of sample (diluted plasma or supernatant of tissue homogenate) was incubated at 37 °C for 30 min. (Preliminary experiments showed the reaction to be linear within 30 min.) The reaction was stopped by addition of 50 μL of glacial acetic acid. Some samples were done in duplicates with one set of the reaction system containing 0.2% diisopropylfluorophosphate (PAF-AH inhibitor) or 1 mM mepacrine (phospholipase A2 inhibitor) to confirm that the activity was due to PAF-AH. The lipids were extracted with chloroform/methanol/water (2:1:0.8), plated on thin layer chromatography plates coated with Silica Gel G and developed in chloroform/methanol/water (65:35:6). The zones comigrating with PAF and lyso-PAF standards were scraped separately. PAF and lyso-PAF were extracted again from the silica gel powder with Bligh and Dyer method and counted in a scintillation counter. AH activity was expressed as nanomoles of lyso-PAF formed per minute. The intraassay variability was usually below 5%. Protein content was determined with the Bio-Rad kit using BSA as standards (Bio-Rad, Hercules, CA).
Measurement of in vivo half life of PAF in plasma. Anesthetized, catheterized rats were treated with CHX (5 mg/kg, i.v.) or saline (1 mL/kg, i.v.). Thirty minutes later, PAF (1.5 μg/kg) containing 1.4 × 107 dpm of 1-O-[3H]alkyl-2-acetyl-3-phosphorylcholine (37 MBq/mL, Amersham) was injected i.v. to the rat. Blood samples (100 μL) were collected from the carotid artery at 0, 5, 15, and 30 min after PAF injection, and mixed instantly with equal volume of saline containing 0.5% acetic acid solution to inactivate AH. The blood sample was centrifuged at 2000 × g for 10 min at room temperature, total lipid was extracted and was analyzed by TLC, as described in the PAF-AH assay. The ratio between [3H]-lyso-PAF and [3H]-PAF was calculated. The half-life of PAF was calculated from the regression line fitted to a log-linear plot of the data.
MPO activity assay(27). A sample of 0.3 g of intestinal tissue was homogenized in 3 mL of 0.05 M potassium phosphate buffer containing 0.5% hexadecyltrimethyl-ammonium bromide and EDTA (5 mM), and sonicated. An aliquot of the tissue homogenate was mixed with the substrate (O-dianisidine HCl + H2O2 in potassium phosphate buffer), and the change of OD at 460 nm/min was recorded with a spectrophotometer. The final MPO activity was calculated based on a standard curve constructed with human leukocyte MPO standards.
Statistical analysis. Results were expressed as mean ± SEM and were analyzed statistically by ANOVA; p value were assigned by either the Dunnett multiple comparison test or Fisher's exact test. Results were considered significant when p < 0.05.
RESULTS
PAF up-regulates PAF-AH activity in the plasma as well as in the intestine; PAF-AH elevation is independent of protein synthesis. As shown in Figure 1 (upper panel), there was a slight increase of PAF-AH activity in plasma within 2 h of the experimental period in sham-operated animals (0.69 ± 0.1 nmol/mL/min). CHX (5 mg/kg, i.v.) decreased basal PAF-AH activity in the plasma (-4.54 ± 0.90 nmol/mL/min, compared with that of sham-operated rats, p < 0.05, Fig. 1, upper panel), suggesting a rapid turnover of the plasma enzyme, which depends on de novo protein synthesis to maintain its physiologic level. However, CHX exhibited no effect on intestinal PAF-AH (Fig. 1,lower panel; not significantly different from the baseline intestinal PAF-AH, 1.0 ± 0.12 nmol/mg of protein/min).
Effect of PAF and CHX on plasma (upper panel) and intestinal (lower panel) AH activity. Plasma samples were collected before the experiment and at 90 min after PAF injection. Intestinal samples were obtained at 90 min after PAF. *p < 0.05, compared with sham-operated group. (Mean ± SEM, see Table 1 for number of animals in each group.)
PAF (1.5 μg/kg, i.v.) significantly increased PAF-AH activity in the plasma (6.1 ± 1.5 nmol/mL/min) and in the small intestine (2.81 ± 0.42 nmol/mg of protein/min) (Fig. 1). This effect was especially pronounced in the intestine, reaching 2.8-fold of control values at 90 min. The stimulatory effect of PAF on plasma PAF-AH seems to be independent of protein synthesis, because CHX, although lowering the basal level of serum PAF-AH, did not abolish the PAF effect on the CHX-pretreated animal (2.94 ± 0.5 nmol/mL/min). CHX also had no effect on PAF-induced intestinal AH (3.14 ± 0.34 nmol/mg of protein/min) (Fig. 1).
CHX synergizes with PAF to induce shock, systemic inflammatory response, and intestinal necrosis. PAF alone, at the dose used (1.5 μg/kg, i.v.), caused only a transient drop of blood pressure, which recovered to near the baseline value within 1.5 h (Fig. 2), and no gross intestinal injury (Table 1). PAF also caused mild hemoconcentration (Hct, 44 ± 1; Fig. 3), peripheral leukocytosis (Fig. 4) with increased neutrophil count (Table 2), and elevated intestinal MPO (an indicator of polymorphonuclear leukocyte sequestration) (Fig. 5). Most of these changes were aggravated by pretreatment of rats with CHX (5 mg/kg, i.v.). CHX plus PAF induced intestinal necrosis in 17 of 18 animals tested (Table 1), and caused severe hemoconcentration (Hct, 55 ± 2; Fig. 3) and leukocytosis (342 ± 59% of baseline value, Fig. 4). Although animals receiving CHX plus PAF had a slow, partial recovery of blood pressure after the initial hypotension, blood pressure fell again after 60 min and resulted in shock at 90 min (Fig. 2). CHX, when used alone, caused a mild drop of blood pressure (Fig. 2) and paradoxically decreased intestinal MPO content (Fig. 5, p < 0.05). CHX also increased both peripheral white blood cell count (Fig. 4, p < 0.05) and lymphocyte count (Table 2, p < 0.05), but had no effect on Hct (Fig. 3). No intestinal injury was observed in rats receiving CHX alone (Table 1). No significant difference was found between the half-lives of exogenous PAF in animals receiving PAF (10.5 ± 0.1 min) and receiving combined CHX and PAF (12.7 ± 2.3 min).
Changes of mean arterial blood pressure. (Upper panel) Effect of PAF and CHX: (○, n = 10) sham-operated; (•, n = 10) PAF (1.5 μg/kg, injected i.v. at time 0); (▵, n = 6) CHX (5 mg/kg, i.v., injected at time -30 min); (▴, n = 18) CHX + PAF. (Lower panel) Effect of various treatment: (▴, n = 18) CHX + PAF; (⋄, n = 5) CHX + anti-TNF + PAF (10 mL/kg of anti-TNF antibody was injected i.p. 20 min before PAF); (♦, n = 6) CHX + PAF + allopurinol (10 mg/kg of allopurinol was injected 20 min after PAF); (□, n = 6) CHX + PAF + WEB (1 mg/kg WEB2170 was injected 20 min after PAF). (Mean ± SEM.)
Effect of various treatments on PAF-induced hemoconcentration. Values obtained at 90 min after PAF injection. See Figure 2 legend for symbols and numbers of animals. *p < 0.05, **p < 0.01 compared with sham-operated (left panel) or CHX + PAF group (right panel). (WEB, WEB2170; αTNF, anti-TNF antibody; Alprol, allopurinol).
Endogenous PAF and TNF play a role in the up-regulation of intestinal AH; endogenous PAF is also responsible for the intestinal injury induced by combined treatment of CHX and PAF. The intestinal necrosis induced by PAF in CHX-pretreated animals was significantly ameliorated by WEB2170 (a PAF antagonist, 1 mg/kg, i.v., 20 min after PAF) (Table 1), indicating a central role of endogenous PAF in mediating tissue injury. WEB2170 also partially reversed the hypotension (Fig. 2). The seemingly beneficial effect of anti-TNF antibody (10 mL/kg, 20 min before PAF) on the gross injury (Table 1) of the intestine was not supported by statistical analysis. There was no significant difference in intestinal MPO activity among these groups (Fig. 5). As shown in Figure 6, the stimulatory effect of PAF on the intestinal AH activity of CHX-pretreated rats was partially but significantly inhibited by anti-TNF antibody and WEB2170 (p < 0.05). In contrast, neither treatment was effective in blocking the PAF effect on plasma PAF-AH activity in CHX-treated rats (Fig. 6).
Changes of plasma (upper panel) and intestinal (lower panel) AH activity after various treatments. Plasma samples were taken before the experiment and at 90 min after PAF. Intestinal samples were collected at 90 min after PAF. *p < 0.05, compared with CHX + PAF-treated animals. See Figure 3 legend for abbreviations.
ROS mediates the injurious effect of PAF and CHX, but does not contribute to the regulation of AH by PAF. To examine whether the effects of PAF on AH stimulation and intestinal injury in CHX-pretreated rats are due to ROS formation, we treated rats with allopurinol (10 mg/kg, i.p.), an XO inhibitor, 20 min after PAF. Allopurinol was given after PAF to avoid early PAF effect such as leukocyte adhesion(28), because it has previously been shown that leukocyteendothelial adhesion may depend on endothelial XO activation(29). We found that allopurinol almost completely prevented PAF-induced intestinal injury (Table 1) in CHX-pretreated rats, and significantly alleviated the hemoconcentration (Fig. 3, p < 0.05, compared with the CHX + PAF group). However, it failed to modulate the hypotensive and leukocytosis changes induced by PAF (Figs. 2 and 4). It also had no effect on plasma or tissue AH (Fig. 6).
DISCUSSION
PAF-AH is a key enzyme for the catabolism of PAF and PAF analogs derived from phospholipid oxidation. It has been found in multiple types of cells(24, 30–32) and in different tissues(15, 33, 34). Two forms of PAF-AH, i.e. intracellular and plasmatic, have been cloned, but no sequence similarity was found between the two(15). Macrophages(15) and hepatocyte(20, 32) have been suggested as the cellular source of plasma AH. A recent study showed that intestinal epithelial cells synthesize and secrete PAF-AH in vitro(24), and epithelial cells isolated from intestine with active ulcerative colitis had a higher level of secreted PAF-AH than did cells from histologically normal tissues(24), suggesting that intestinal epithelia may be an additional source of plasma PAF-AH in vivo.
In the rat model of NEC produced by injecting PAF, we found that a low dose of PAF (1.5 μg/kg) up-regulates PAF-AH activity in both the plasma and intestinal tissue within 90 min, suggesting that PAF up-regulates tissue AH activity and its secretion (into plasma). It has been shown that both the synthesis and release of PAF-AH by macrophages and HL-60 cells are dependent on protein synthesis(25, 26). The present study showed that plasma PAF-AH activity decreased within 2 h after CHX treatment, whereas the intestinal PAF-AH was not affected. This observation suggests that there is a rapid turnover of plasma PAF-AH, whose homeostasis depends on new protein synthesis, whereas the tissue AH, probably of the intracellular, nonsecretory form, is stable and not subjected to a constant degradation process, as is the case of the plasma.
Although lowering the plasma PAF-AH level by blocking protein synthesis does not induce a systemic inflammatory response, we found that this treatment rendered the animal more sensitive to PAF stimulation. Low dose PAF (1.5 μg/kg), insufficient to cause injury, induced a systemic inflammatory response and tissue damage when given after CHX pretreatment. Such an exaggerated response may be due, at least in part, to lowered plasma PAF-AH. We reported previously that PAF administration induced endogenous PAF formation in intestinal tissue(8) and resulted in an elevated serum PAF level(6). Because the half-life of exogenous PAF (which is very short)(35) did not differ significantly in the PAF and CHX + PAF treatment groups, it may be reasoned that it is the catabolism of the endogenous PAF, rather than the injected PAF, that was affected by the decreased plasma PAF-AH activity. This may account for the late, second phase drop (60 min after PAF) of blood pressure in CHX-pretreated rats, in contrast to the rapid, profound, and irreversible hypotension induced by a high dose (2.5-3 μg/kg) of PAF(14).
It has been shown that release of PAF-AH by macrophages is regulated by several inflammatory mediators, such as PAF, macrophage colony-stimulating factor, and granulocyte/macrophage colony-stimulating factor(36). However, the role of inflammatory mediators in the regulation of PAF-AH in vivo has been examined only rarely. Our earlier studies showed that PAF induces endogenous synthesis of PAF(8) in the small intestine to mediate injury. Thus, it is not surprising that posttreatment with PAF antagonists reduced intestinal injury. The present study also demonstrates that both endogenous TNF and endogenous PAF were involved in the PAF-induced up-regulation of AH activity. In the presence of low dose PAF, this may be an important mechanism to protect the animal from tissue injury by counteracting the increased endogenous PAF production. It is possible that, at high dose PAF, the endogenous PAF production exceeds the capacity of tissue AH, and injury develops. Interestingly, plasma AH appears independent of these endogenous mediators.
Administration of allopurinol, a competitive inhibitor of XO, prevented the intestinal injury induced by PAF in CHX-pretreated animals. The small intestine is a rich source of xanthine dehydrogenase, the precursor of XO, an enzyme responsible for the production of ROS during ischemic/reperfusion injury(37). We and other investigators have shown that pretreatment with allopurinol significantly ameliorated PAF(38)- or indomethacin(39)-induced intestinal injury, suggesting that intestinal ROS formation catalyzed by XO is an important factor causing tissue injury. A previous report demonstrates that ROS reduces plasma PAF-AH within 60 s and completely inactivates it in 10 min(40). To our surprise, we failed to find a significant difference in AH activity, either in plasma or intestinal tissue, between the groups with and without allopurinol treatment. Thus, the protective effect of allopurinol is not due to an elevated plasma or intestinal AH activity, but may be due to its direct action on the abrogation of ROS formation. It is speculated that the exaggerated injury in response to PAF in CHX-treated animals may also be related to a decreased production of proteins involved in the bodily defense, such as an endogenous antioxidant system.
Abbreviations
- PAF:
-
platelet-activating factor
- NEC:
-
necrotizing enterocolitis
- TNF:
-
tumor necrosis factor
- AH:
-
acetylhydrolase
- CHX:
-
cycloheximide
- XO:
-
xanthine oxidase
- MPO:
-
myeloperoxidase
- Hct:
-
hematocrit
- ROS:
-
reactive oxygen species
References
Snyder F 1990 Platelet-activating factor and related acetylated lipids as potent biologically active cellular mediators. Am J Physiol 259:C697–C708.
Prescott SM, Zimmerman GA, McIntyre TM 1990 Platelet -activating factor. J Biol Chem 265: 17381–17384.
Rosam AC, Wallace JL, Whittle BJ 1986 Potent ulcerogenic actions of platelet-activating factor on the stomach. Nature 319: 54–56.
Nassif A, Longo WE, Mazuski JE, Vernava AM, Kaminski DL 1996 Role of cytokines and platelet-activating factor in inflammatory bowel disease. Implications for therapy. Dis Colon Rectum 39: 217–223.
Caplan MS, Sun XM, Hsueh W, Hageman JR 1990 Role of platelet activating factor and tumor necrosis factor-alpha in neonatal necrotizing enterocolitis. J Pediatr 116: 960–964.
Sun X, Caplan MS, Liu Y, Hsueh W 1995 Endotoxin-resistant mice are protected from PAF-induced bowel injury and death. Role of TNF, complement activation, and endogenous PAF production. Dig Dis Sci 40: 495–502.
Qu XW, Huang L, Burthart T, Crawford SE, Caplan MS, Hsuch W 1996 PAF production in the rat ileum: quantitation of tissue PAF by an improved method. Prostaglandins 51: 249–262.
Zhang C, Hsueh W, Caplan MS, Kelly A 1991 Platelet activating factor-induced shock and intestinal necrosis in the rat: role of endogenous platelet-activating factor and effect of saline infusion. Crit Care Med 19: 1067–1072.
Doebber TW, Wu MS, Robbins JC, Choy BM, Chang MN, Shen TY 1985 Platelet-activating factor (PAF) involvement in endotoxin-induced hypotension in rats. Studies with PAF-receptor antagonist Kadsurenone. Biochem Biophys Res Common 127: 799–808.
Chang SW, Feddersen CO, Henson PM, Voelkel NF 1987 Platelet-activating factor mediates hemodynamic changes and lung injury in endotoxin treated rat. J Clin Invest 797: 1498–1509.
Sun XM, Hsueh W 1988 Bowel necrosis induced by tumor necrosis induced by tumor necrosis factor in rats is mediated by platelet-activating factor. J Clin Invest 81: 1328–1331.
Warren JS, Barton PA, Mandel DM, Matrosic K 1990 Intrapulmonary tumor necrosis factor triggers local platelet activating factor production in rat immune complex alveolitis. Lab Invest 63: 746–754.
Caplan MS, Sun XM, Hsueh W 1990 Hypoxia causes ischemic bowel necrosis in rats: the role of platelet-activating factor (PAF-acether). Gastroenterology 99: 979–986.
Hsueh W, Caplan MS, Sun X, Tan X, MacKendrick W, Gonzalez-Crussi F 1994 Platelet-activating factor, tumor necrosis factor, hypoxia and necrotizing enterocolitis. Acta Paediatr 396: 11–17.
Tjoelker LW, Wilder C, Eberhardt C, Stafforini DM, Dietsch G, Schimpf B, Hooper S, Le Trong H, Cousens LS, Zimmerman GA, Yamada Y, McIntyre TM, Prescott SM, Gray PW 1995 Anti-inflammatory properties of a platelet-activating factor acetylhydrolase. Nature 374: 549–543.
Caplan M, Hsueh W, Kelly A, Donovan M 1990 Serum PAF acetylhydrolase increase during neonatal maturation. Prostaglandins 39: 705–714.
Lucas A, Cole TJ 1990 Breast milk and neonatal necrotizing enterocolitis. Lancet 336: 1519–1523.
Furukawa M, Narahara H, Yasuda K, Johnston JM 1993 Presence of platelet-activating factor-acetylhydrolase in milk. J Lipid Res 34: 1603–1609.
Furukawa M, Lee EL, Johnston JM 1993 Platelet-activating factor-induced ischemic bowel necrosis: the effect of platelet-activating factor acetylhydrolase. Pediatr Res 34: 237–241.
Svetlov SI, Howard KM, Miwa M, Flickinger BD, Olson MS 1996 Interaction of platelet-activating factor with rat hepatocytes: uptake, translocation, metabolism, and effects on PAF-acetylhydrolase secretion and protein tyrosine phosphorylation. Arch Biochem Biophys 327: 113–122.
Satoh K, Imaizumi T, Kawamura Y, Yoshida H, Hiramoto M, Takamatsu S, Takamatsu M 1991 Platelet-activating factor (PAF) stimulates the production of PAF acetylhydrolase by the human hepatoma cell line. J Clin Invest 87: 476–481.
Huang L, Tan X, Crawford SE, Hsueh W 1994 Platelet-activating factor and endotoxin induce tumor necrosis factor gene expression in rat intestine and liver. Immunology 83: 65–69.
Tan XD, Wang H, Gonzales-Crussi FX, Chang H, Gonzales-Crussi F, Hsueh W 1996 Platelet activating factor and endotoxin increase the enzyme activity and gene expression of type II phospholipase A2 in the rat intestine. Role of polymorphonuclear leukocytes. J Immunol 156: 2985–2990.
Riehl TE, Stenson WF 1995 Platelet-activating factor acetylhydrolases in Caco-2 cells and epithelium of normal and ulcerative colitis patients. Gastroenterology 109: 1826–1834.
Narahara H, Nishioka Y, Johnston JM 1993 Secretion of platelet-activating factor acetylhydrolase by human decidual macrophages. J Clin Endocr Metab 77: 1258–1262.
Lee TC, Fitzgerald V, Chatterjee R, Malone B, Snyder F 1994 Differentiation induced increase of platelet-activating factor acetylhydrolase in HL-60 cells. J Lipid Mediator 9: 267–283.
Sun XM, Qu XW, Huang W, Granger DN, Bree M, Hsueh W 1996 Role of leukocyte β2-integrin in PAF-induced shock and intestinal injury. Am J Physiol 270:G184–G190.
Kubes P, Ibbotson G, Russell J, Wallace JL, Granger DN 1990 Role of platelet-activating factor in ischemia/reperfusion-induced leukocyte adherence. Am J Physiol 259:G300–G305.
Gaboury J, Woodman RC, Granger DN, Reinhardt P, Kubes P 1993 Nitric oxide prevents leukocyte adherence: role of superoxide. Am J Physiol 265:H862–H867.
Lee TC, Malone B, Wasserman SI, Fitzgerald V, Snyder F 1982 Activities of enzymes that metabolize platelet-activating factor (1-alkyl-2-acetyl-sn-glycero-3-phosphocholine) in neutrophils and eosinophils from humans and the effect of a calcium ionophore. Biochem Biophys Res Commun 105: 1303–1308.
Stafforini DM, Rollins EN, Prescott SM, McIntyre TM 1993 The platelet-activating factor acetylhydrolase from human erythrocytes. Purification and properties. J Biol Chem 268: 3857–3865.
Tarbet EB, Stafforini DM, Elstad MR, Zimmerman GA, McIntyre TM, Prescott SM 1991 Liver cells secrete the plasma form of platelet-activating factor acetylhydrolase. J Biol Chem 266: 16667–16673.
Nijssen JG, Roosenboom CF, van den Bosch H 1986 Identification of a calcium-independent phospholipase A2 in rat lung cytosol and differentiation from acetylhydrolase for 1-alkyl-2-acetyl-sn-glycero-3-phosphocholine (PAF-acether). Biochim Biophys Acta 876: 611–618.
Adachi H, Tsujimoto M, Hattori M, Arai H, Inoue K 1995 cDNA cloning of human cytosolic platelet-activating factor acetylhydrolase γ-subunit and its mRNA expression in human tissues. Biochem Biophy Res Commun 214: 180–187.
Graham RM, Stephens CJ, Silvester W, Leong LL, Sturm MJ, Taylor RR 1994 Plasma degradation of platelet-activating factor in severely ill patients with clinical sepsis. Crit Care Med 22: 204–212.
Dupuis F, Denizot Y, Fixe P, Dulery C, Praloran V 1996 PAF and haematopoiesis. Macrophage colony-stimulating factor and granulocyte macrophage colony-stimulating factor enhance platelet-activating factor acetylhydrolase production by human blood-derived macrophages. Biochim Biophys Acta 1311: 27–32.
Parks DA, Williams TK, Beckman JS 1988 Conversion of xanthine dehydrogenase to oxidase in ischemic rat intestine: a reevaluation. Am J Physiol 254:G768–G774.
Cueva JP, Hsueh W 1988 Role of oxygen derived free radicals in platelet activating factor induced bowel necrosis. Gut 29: 1207–1212.
Zahavi I, Fisher S, Marcus H, Heckelman B, Kiro A, Dinari G 1995 Oxygen radical scavengers are protective against indomethacin-induced intestinal ulceration in the rat. J Pediatr Gastroenerol Nutr 21: 154–157.
Ambrosio G, Oriente A, Napoli C, Palumbo G, Chicariello P, Marone G, Condorelli M, Chiariello M, Triggiani M 1994 Oxygen radicals inhibit human plasma acetylhydrolase, the enzyme that catabolizes platelet-activating factor. J Clin Invest 93: 2408–2416.
Acknowledgements
The authors thank Hsiao-Ting Chiu, M.S., Children's Memorial Institute for Education and Research (CMIER), for assistance with statistical analysis.
Author information
Authors and Affiliations
Additional information
Supported by National Institutes of Health Grants HD31840 and DK34574.
Rights and permissions
About this article
Cite this article
Wang, H., Tan, XD., Qu, XW. et al. Platelet-Activating Factor (PAF) Up-Regulates Plasma and Tissue PAF-Acetylhydrolase Activity in the Rat: Effect of Cycloheximide. Pediatr Res 42, 597–603 (1997). https://doi.org/10.1203/00006450-199711000-00008
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199711000-00008
This article is cited by
-
Platelet-activating factor and endotoxin activate CCAAT/enhancer binding protein in rat small intestine
British Journal of Pharmacology (2001)