Abstract
A patient with a deficient voltage-dependent anion channel (VDAC) is reported, presenting clinically with psychomotor retardation and minor dysmorphic features. Biochemical studies on muscle mitochondria showed impaired rates of pyruvate oxidation and ATP production; however, no specific deficient activity of one of the mitochondrial enzymes was involved. Western blotting experiments indicated an almost complete VDAC deficiency in skeletal muscle. The only moderately decreased VDAC content in the patient's fibroblasts might indicate that VDAC is expressed in a tissue-specific manner. The deficiency is likely caused by a mutation in the HVDAC1 gene or by a disturbed posttranslational modification. This is the first described deficiency of a component of the outer mitochondrial membrane associated with the pyruvate oxidation pathway. Defects in this membrane should be considered as a possible cause of otherwise unexplained mitochondrial disorders.
Similar content being viewed by others
Main
A dramatic increase in recognition of disorders of mitochondrial energy metabolism has been seen in the last decade(1, 2). These disorders show diverse clinical symptoms ranging from pure myopathy with lactic acidosis to severe multisystem disease with central nervous system involvement. Frequently, biochemical defects of the pyruvate dehydrogenase complex, or of one or more of the four multisubunit complexes of the respiratory chain, have been demonstrated as underlying causes(1, 3), or they are associated with mutations in the mitochondrial genome(1, 2).
Biochemical investigations in patients suspected to have a respiratory chain defect are commonly performed on skeletal muscle. They may involve measurement of substrate oxidation or oxygen consumption by intact mitochondria to obtain information about the overall flux through the various parts of the mitochondrial energy metabolism. In case of an impaired flux, the different enzyme components of the respiratory chain and of the pyruvate dehydrogenase complex are measured, possibly followed by immunoblotting to precisely define the defect at the enzyme and protein level.
Despite progress in the diagnostic approach, we experienced that, in almost a quarter of the patients with evidently reduced substrate oxidation in muscle mitochondria, these abnormalities could not be ascribed to a known defect in one of the mitochondrial enzymes. We hypothesized that, in at least part of this category of patients, malfunctioning of one of the transport mechanisms across the mitochondrial membranes may cause biochemical and clinical disorders.
For adequate functioning of the mitochondrial energy pathway, metabolites which are directly involved in the process of oxidative phosphorylation, such as ADP, ATP, and phosphate have to be transported across mitochondrial membranes, as well as ions (K+, Na+, H+, Ca2+) to maintain osmotic homeostasis in the mitochondrial matrix(4, 5). Hence, when studying mitochondrial disorders it seems worthwhile to investigate the proteins which are responsible for the transport of the different substances across mitochondrial membranes.
VDAC is one of these transporters in the outer mitochondrial membrane. This channel, also called mitochondrial porin, forms a pore which at low transmembrane voltage is open for anions such as phosphate, chloride, and adenine nucleotides. At a high transmembrane voltage or in the presence of a specific VDAC-modulating protein, VDAC functions as a selective channel for cations and various uncharged molecules(6, 7).
To detect disorders of the mitochondrial transport system, we screened a selected group of patients for abnormalities of VDAC in skeletal muscle. One subject with deficiency of VDAC protein was detected. The preliminary findings of this patient have previously been published(8). The clinical, biochemical, and muscle morphologic features of the patient are described in the present report.
METHODS
Case report. The male patient, the third child of healthy unrelated parents, was born at term after a normal pregnancy. Delivery of the macrosomic child (weight 5.7 kg, length 60 cm, head circumference 43.2 cm) was delayed. The patient presented with mild unspecific facial dysmorphism with prominent forehead, deep nasal bridge, mild hypertelorism, and high arched palatum. The internipple distance was increased, and the hypotonic abdominal wall showed median diastasis and herniation of the umbilical cord. There was a bilateral syndactyly of the 2nd and 3rd toe with hypoplastic nails. Initially due to intrapartum asphyxia, resuscitating measures were required. The Apgar score was 7 after 10 min. Oxygen was administered during the first 10 postnatal days. Brief neonatal seizures subsided after the start of phenobarbital treatment. In the first weeks mild hypothalamic hypothyroidism became obvious, and treatment with thyroxin was started. The psychomotor development was abnormal; the patient showed failure to thrive and had persistent muscular hypotonia with persistent head lag. There was poor visual fixation and no progressive development or milestones.
At the age of 5 mo, a magnetic resonance imaging scan of the brain showed mildly dilated and disfigured lateral ventricles (Fig. 1a). An area of cortical dysplasia, probably polymicrogyria, was seen in the region of the right Sylvian fissure (Fig. 1,a and b). The entire cortex showed too many unbranched gyri. The white matter contained hardly any myelin and had a high signal intensity throughout, indicative of a delay in myelination.
At the age of 7 mo a progressive hydrocephalus was diagnosed and treated with ventriculo-peritonal drainage. Electro-encephalography showed epileptic discharges. Visual and auditory evoked potentials showed reduction of central conduction velocities. Motor conduction velocities were normal. Electromyography revealed a myopathic pattern. The patient was strongly suspected to suffer from the Pallister-Killian syndrome, but a tetrasomy 12p was excluded both in blood cells and fibroblasts. The results of repeated routine laboratory tests in blood, cerebrospinal fluid, and urine were within normal limits, and comprehensive metabolic investigations gave normal results as well. Because an i.v. glucose loading test (2 g/kg body weight) showed a pathologic increase in blood lactate concentration (from 1.1 to 2.4 mM), a mitochondrial respiratory chain defect was considered in the differential diagnosis. Therefore a muscle biopsy of the quadriceps was performed at the age of 20 mo. At the age of 4 y the main clinical symptom was psychomotor retardation.
Morphologic studies. For morphologic investigations 8-μm sections of skeletal muscle were used for conventional histology and enzyme histochemistry. The sections were stained for succinate dehydrogenase, myosin-ATPase (pH 4.3, 4.6, and 10.3), and cytochrome oxidase according to standard procedures(9, 10). A small muscle specimen was fixed in glutaraldehyde for electron microscopic studies.
Biochemical studies. For biochemical investigations 10%(wt/vol) muscle homogenate was prepared in SETH medium (250 mM sucrose, 2 mM EDTA, 10 mM Tris-HCl, pH 7.4, 50 U/mL heparin). The 600 × g supernatant was used for biochemical determinations(11). Energy metabolism of intact muscle mitochondria was tested by measuring the rates of oxidation of labeled substrates and of ATP plus creatine phosphate production as described earlier(11).[1-14C]Pyruvate, [U-14C]malate, and [1,4-14C]succinate (1 mM) were used in combination with different co-substrates and specific inhibitors. In parallel experiments mitochondrial ATP and creatine phosphate production from pyruvate as well as from succinate was measured. All incubations were performed in the presence of 2 mM ADP and 20 mM creatine. In one experiment pyruvate oxidation was determined in the presence of the uncoupling agent CCCP (2 μM). Activities of the various enzyme complexes of the respiratory chain were determined in the 600 × g supernatant as previously described(11). Samples were frozen and stored at -80°C after homogenization of fresh muscle. Activity of the pyruvate dehydrogenase complex and content of carnitine were determined in total muscle homogenate(12, 13).
Skin fibroblasts were cultured in Medium 199 with Earle's salts (ICN Biomedicals, Zoetermeer, The Netherlands) to confluency. The lactate/pyruvate ratio in fibroblasts was determined essentially as described by Wijburget al.(14).
For all other studies on fibroblasts, mitochondrial enriched fractions prepared from cultured fibroblasts by homogenization and subsequent differential centrifugation were used.
Immunochemical studies. Muscle 600 × g supernatants(11) and isolated mitochondria (14,000× g pellet of the 600 × g fraction) from 70 suspected patients, as well as mitochondrial enriched fractions of skin fibroblasts, were used for immunochemical studies. The samples were suspended at room temperature in sample buffer [4% (wt/vol) SDS, 5 mM aminobenzidine, 6 M urea, 0.01% (wt/vol) bromphenol blue in 6.25 mM Tris-HCl, pH 6.8]. Samples containing 100 mU of the mitochondrial reference enzyme cytochrome c oxidase (unless otherwise indicated), determined according to the method of Cooperstein and Lazarow(15), were applied to 10-15% polyacrylamide gels. The proteins in the samples were separated by SDS-PAGE and electroblotted to 0.2-μm nitrocellulose membranes (Schleicher & Schuell, Dassel, Germany). Both electrophoresis and electroblotting were performed on Phast System equipment (Pharmacia-LKB, Woerden, The Netherlands). The blots were incubated with various antisera against ANT and VDAC. Antigen-antibody complexes were detected using enhanced chemiluminescence(ECL; Amersham International plc, Buckinghamshire, England) with biotinylated horseradish peroxidase as detection ligand (Dako, Glostrup, Denmark).
Antibodies. Two polyclonal antisera against ANT were applied to Western blots. One was raised against ANT isolated from bovine heart(16) and one against the synthetic peptide corresponding to the C-terminal part (amino acids 288-297) of the protein(17). Three different antibodies against VDAC were applied: one polyclonal antiserum against VDAC isolated from bovine heart(18) and two MAb raised against VDAC isolated from human B lymphocytes and cultured in two different cell lines (MAb-4 and MAb-6)(19).
RESULTS
Morphology. Examination of the muscle by light microscopy revealed a normal checkerboard pattern of fiber types with 45% type I and 55% type II fibers. The fiber diameter was smaller (8-19 μm) compared with age-matched controls (10-30 μm). Other stainings (i.e. succinate dehydrogenase, cytochrome oxidase, NADH-dehydrogenase, ATPase) gave normal patterns. Ultrastructural investigations showed normal mitochondria with clearly visible mitochondrial outer membranes (Fig. 2).
Biochemistry. In muscle all measured substrate oxidation rates were decreased (Table 1). Uncoupling by CCCP did not stimulate pyruvate oxidation. The generation of high energetic compounds was decreased to the same extent as the oxidation rates. The activities of the respiratory chain enzymes were related to the mitochondrial reference enzyme citrate synthase because the mitochondrial content of human skeletal muscle can vary to a considerable extent, especially in pathologic tissue. One should distinguish between lowered mitochondrial content and a specific enzyme deficiency. No specific deficiency of a respiratory chain enzyme was detected. The activities of the respiratory chain enzymes were all normal(Table 1). The carnitine content in muscle was slightly decreased: 2.1 μmol/g of muscle [control (n) = 2.7-4.7].
In patient's fibroblasts the lactate/pyruvate ratio after glucose conversion appeared to be normal: 12 (n < 25), which means there is no indication for a disturbance in oxidative phosphorylation in fibroblasts.
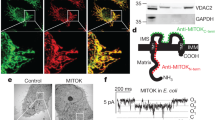
Immunochemistry. A clearly decreased amount of the VDAC protein(35 kD) in the patient's muscle, using both MAb against human VDAC as well as the polyclonal antibovine heart VDAC antiserum, is seen in Figure 3. The ANT content (29 kD) appears to be only slightly reduced. The polyclonal anti-VDAC antiserum also reacts with other muscle proteins. In this polyclonal anti-VDAC panel the amount of all proteins in the patient appeared to be comparable with those in controls except for the VDAC protein. The polyclonal antiserum against total ANT gave similar results(not shown) as the antiserum directed against the C terminus of ANT. The two MAb against VDAC isolated from human B lymphocytes (MAb-4 and MAb-6) reacted comparably in all applications. In the Figures 3,4,and 5, only MAb-6 results are shown.
Immunoblots of patient and control muscle incubated with antibodies against VDAC and ANT. The 600 × g muscle supernatants, containing 100 mU of cytochrome c oxidase, were loaded in each lane. Lanes 3, patient's muscle; lanes 1, 2, and4, three different control muscles; lane V, isolated VDAC(35 kD); lane A, isolated ANT (29 kD). Outer lanes, biotinylated molecular mass marker (39.8, 29.0, and 20.1 kD). The immunostaining was performed with three different antibodies: monoclonal anti-VDAC (mAb-6), polyclonal anti VDAC (pVDAC), and polyclonal anti C-terminal ANT (C-ANT).
Immunoblot of mitochondrial enriched fractions of cultured skin fibroblasts from patient (lanes 3) and three different controls (lanes 1, 2, and 4), all containing 100 mU of cytochrome c oxidase. Immunostaining was performed with antibodies against VDAC (mAb-6, pVDAC) and ANT (C-ANT). For additional details, see legend to Figure 2.
Investigation of the amount of VDAC in the patient's muscle (left) and fibroblasts (right) by using varying activities of the mitochondrial reference enzyme cytochrome c oxidase (10, 30, 100, and 150 mU). The activity of cytochrome c oxidase in milliunits and source of the samples (con, control;pat, patient) are indicated at the top. The blots were incubated with MAb-6 antiserum.
Figure 4 shows the results of the immunochemical examination of skin fibroblasts. Compared with muscle tissue the patient's fibroblasts show a less decreased VDAC content. The amount of ANT in fibroblasts is normal compared with controls.
The relative VDAC content in the patient's muscle tissue and fibroblasts is presented in Figure 5. The specific amount of VDAC is about 10-fold decreased in the patient's muscle, whereas in fibroblasts the VDAC amount appears to be only slightly decreased.
DISCUSSION
At present, knowledge about the function of mitochondrial membrane carriers in human physiology and pathophysiology is limited. Only a few subjects with a defective carrier have been described: one with ANT deficiency(20), one suspected to have a defective protein import into mitochondria(21), and one likely with a defective malate-aspartate shuttle(22). Lack of the carnitine carrier has been found more frequently(23). To our knowledge no systematic study has been performed to identify deficiencies of other mitochondrial membrane carriers.
By screening a selected group of subjects, we found one with deficient VDAC protein in skeletal muscle. In this subject, investigation of the muscle mitochondria showed impaired substrate oxidation and reduced production of high energetic compounds (Table 1). These data point to a functional defect in one of the many components involved in the mitochondrial energy metabolism, i.e. respiratory chain enzymes or postrespiratory chain energy-converting steps, such as ATPase, mitochondrial creatine kinase, or ANT. An abnormal mitochondrial matrix composition, due to altered ion homeostasis or diminished NAD+ or coenzyme A concentration, could also have resulted in a reduced oxidative phosphorylation capacity.
A defect in the respiratory chain could be excluded by demonstrating normal enzyme activities. A defect in postrespiratory chain energy-converting processes was less presumable because stimulation of pyruvate oxidation by use of the uncoupler CCCP, which is expected in such a case(20, 24), failed to appear.
In contrast, abnormal ion composition and/or altered osmolarity within the mitochondrial matrix could be a logic explanation for an impaired mitochondrial energy metabolism in our patient. In this respect it is well known that a lower pH in the mitochondrial matrix resulting from any disturbance of proton transport across mitochondrial membranes inhibits mitochondrial ATPase and thus ATP production(25), as well as various components of the respiratory chain and mitochondrial transport systems(26). Likewise, maintenance of the volume of the mitochondrial matrix within a narrow range is crucial for proper energy metabolism. Any change of the permeability of the inner or outer mitochondrial membrane may influence the respiratory rate(4, 5, 7).
One of the proteins involved in anion and cation transport across the outer mitochondrial membrane is VDAC. VDAC is an abundant channel-forming protein; the pore size is regulated by the membrane potential(6, 7, 27, 28). The shortage of VDAC in our patient's muscle tissue might have caused an abnormal ion composition of the mitochondrial matrix, thereby deteriorating the process of oxidative phosphorylation. Because VDAC is also responsible for the transport of ATP and ADP across the outer mitochondrial membrane(6, 27), one would expect in our patient a reduced transport of ADP and inorganic phosphate inward, as well as ATP out of the mitochondria, thereby hampering the energy metabolism. From the fact that the uncoupler CCCP did not stimulate substrate oxidation in muscle tissue, it could be hypothesized that biochemical abnormalities in VDAC-deficient mitochondria have been the consequence of malfunctioning of ion transport rather than the consequence of a blockade of ADP and ATP transport.
The important role of VDAC in energetic and metabolic function of mitochondria is illustrated with VDAC-deficient mutants, which have been generated only in yeast mitochondria. These mutants showed a disturbed respiration, whereas the respiration in the uncoupled state was normal. The mutants showed an increased requirement for ADP and Mg2+ ions(29–31). To comply with the requirements, the size of VDAC pores may be regulated, either by a specific modulator protein(7, 32), by NADH concentration, or by the colloidal osmotic pressure, shifting the membrane potential required for opening and closure of the pores(5, 33–35). Also the fact that VDAC interacts with kinases, ANT, and benzodiazepine receptors in important mitochondrial structures, such as special contact sites and megachannels, might underline its regulatory role in mitochondrial energy metabolism(5, 27, 35, 36).
In our patient VDAC deficiency was expressed in a tissue-specific manner. VDAC protein was severely decreased in skeletal muscle, but only moderately decreased in cultured fibroblasts. With respect to human VDAC, five partly established cDNA sequences, encoded by four genes, have been reported(37–39). The occurrence of isoforms might be an explanation for the tissue-specific VDAC disturbances in our patient. However, the monoclonal anti-VDAC antibody which was used in our experiments was shown to recognize only the HVDAC1 isoform and more specifically the acetylated N-terminal region(40). So, the most likely explanation is that the HVDAC1 protein in muscle tissue of our patient is almost completely absent, mutated, or nonacetylated. The gene of HVDAC1 is located on chromosome X in the interval Xq13-q21(38). High resolution analysis of the patient's X chromosome did not demonstrate abnormalities. Studies are in progress to detect the defect at molecular level.
In our patient, a damage of the mitochondrial outer membrane in which VDAC is incorporated could also have induced a lack of the VDAC protein. However, ultrastructural investigations clearly showed the presence of a normal mitochondrial outer membrane. Unfortunately, not enough muscle was available to estimate the presence of other components of the mitochondrial outer membrane. In our opinion the VDAC deficiency is not caused by the absence of(large parts of) the outer membrane, because oxidative phosphorylation remains usually unimpaired in mitochondria devoid of the outer membrane(5, 6, 31). Removal of the mitochondrial outer membrane in VDAC-deficient mutants even induces normalization of energy metabolism(31).
VDAC has also been reported to be a constituent of the plasma membrane of mammalian brain cells(41, 42), human T and B lymphocytes(19, 43, 44), muscle cells(19), and an epithelial carcinoma cell line(45). It seems to occur also in the sarcoplasmatic reticulum(46). It remains obscure whether the multitopologic localization of VDAC and its deficiency is of importance for the clinical findings in our patient. It is unknown whether VDAC is also deficient in cerebral tissues and whether the abnormalities in magnetic resonance imaging findings and psychomotor development are related to such a defect.
In conclusion, this report on a patient with a deficient VDAC protein together with a recently reported ANT defect(20) suggests that an immmunochemical approach provides a useful extension of the diagnostic program for detecting mitochondrial disorders.
Abbreviations
- ANT:
-
adenine nucleotide translocator
- CCCP:
-
carbonyl cyanide-p-(trifluoromethoxy)phenylhydrazone
- VDAC:
-
voltage-dependent anion channel
References
DiMauro S 1993 Mitochondrial encephalomyopathies. In: Rosenburg RN, Prusiner SB, DiMauro S, Barchi LR, Kunkel LM (eds) The Molecular and Genetic Basis of Neurological Disease. Butterworth-Heinemann, Boston, pp 665–694.
Shoffner JM, Wallace DC 1994 Oxidative phosphorylation diseases and mitochondrial DNA mutations: diagnosis and treatment. Annu Rev Nutr 14: 535–568.
Scholte HR 1988 The biochemical basis of mitochondrial diseases. J Bioenerg Biomembr 20: 161–192.
Halestrap AP, Connern CP, Griffiths EJ 1992 Mechanisms involved in the regulation of mitochondrial inner membrane permeability and their physiological and pathological significance. In: Quagliariello E, Palmieri F (eds) Molecular Mechanisms of Transport. Elsevier Science Publishers BV, Amsterdam, pp 259–266.
Brdiczka D, Wallimann T 1994 The importance of the outer mitochondrial compartment in regulation of energy metabolism. Mol Cell Biochem 133/134: 69–83.
Mannella CA 1992 The ins andouts of mitochondrial membrane channels. Trends Biochem Sci 17: 315–320.
Liu MY, Colombini M 1992 Regulation of mitochondrial respiration by controlling the permeability of the outer membrane through the mitochondrial channel, VDAC. Biochim Biophys Acta 1098: 255–260.
Huizing M, Ruitenbeek W, Thinnes FP, DePinto V 1994 Lack of voltage-dependent anion channel in human mitochondrial myopathies. Lancet 344: 762
Dubowitz V, Brooke MH, Neville HE, (eds) 1973 Muscle biopsy: a modern approach. In: Major Problems in Neurology, Vol 2. WB Saunders, London
Novikoff AB, Goldfischer S 1969 Visualisation of peroxisomes (microbodies) and mitochondria with diaminobenzidine. J Histochem Cytochem 17: 675–680.
Fischer JC, Ruitenbeek W, Gabreels FJM, Janssen AJM, Renier WO, Sengers RCA, Stadhouders AM, ter Laak HJ, Trijbels JMF, Veerkamp JH 1986 A mitochondrial encephalomyopathy: the first case with an established defect at the level of coenzyme Q. Eur J Pediatr 144: 441–444.
Sperl WA, Ruitenbeek W, Kerkhof CMC, Sengers RCA, Trijbels JMF, Guggenbichler JP, Janssen AJM, Bakkeren JAJM 1990 Deficiency of the and subunits of pyruvate dehydrogenase in a patient with lactic acidosis and unexpected sudden death. Eur J Pediatr 149: 487–492.
Parvin R, Pande SV 1977 Microdetermination of(-)carnitine and carnitineacetyl-transferase activity. Anal Biochem 79: 190–201.
Wijburg FA, Feller N, Ruitenbeek W, Trijbels JMF, Sengers RCA, Scholte HR, Przyrembel H, Wanders RJ 1990 Detection of respiratory chain dysfunction by measuring lactate and pyruvate production in cultured fibroblasts. J Inherit Metab Dis 13: 355–358.
Cooperstein SJ, Lazarow A 1951 A microspectrophotometric method for the determination of cytochrome oxidase. J Biol Chem 189: 665–670.
Bentlage HACM, Janssen AJM, Chomyn A, Attardi G, Walker JE, Schagger H, Sengers RCA, Trijbels JMF 1995 Multiple deficiencies of mitochondrial DNA- and nuclear-encoded subunits of respiratory NADH dehydrogenase detected with peptide- and subunit-specific antibodies in mitochondrial myopathies. Biochim Biophys Acta 1234: 62–72.
Brandolin G, Boulay F, Dalbon P, Vignais PV 1989 Orientation of the N-terminal region of the membrane-bound ADP/ATP carrier protein explored by antipeptide antibodies and an arginine-specific endoprotease. Evidence that the accessibility of the N-terminal residues depends on the conformational state of the carrier. Biochemistry 28: 1093–1100.
DePinto V, Prezioso G, Thinnes FP, Link TA, Palmieri F 1991 Peptide-specific antibodies and proteases as probes of the transmembrane topology of the bovine heart mitochondrial porin. Biochemistry 30: 10191–10200.
Babel D, Walter G, Gotz H, Thinnes FP, Jurgens L, Konig U, Hilschmann N 1991 Studies on human porin. VI. Production and characterization of eight monoclonal mouse antibodies against the human VDAC“porin 31HL” and their application for histotopological studies in human skeletal muscle. Biol Chem Hoppe Seyler 372: 1027–1034.
Bakker HD, Scholte HR, van den Bogert C, Ruitenbeek W, Jeneson JAL, Wanders RJA, Abeling NGGM, Dorland B, Sengers RCA, van Gennip AH 1993 Deficiency of the adenine nucleotide translocator in muscle of a patient with myopathy and lactic acidosis: a new mitochondrial defect. Pediatr Res 33: 412–417.
Schapira AHV, Cooper JM, Morgan-Hughes JA, Landon DN, Clark JB 1990 Mitochondrial myopathy with a defect of mitochondrial-protein transport. N Engl J Med 323: 37–42.
Hayes DJ, Taylor DJ, Bore PJ, Hilton-Jones D, Arnold DL, Squier MV, Gent AE, Radda GK 1987 An unusual metabolic myopathy: a malate-aspartate shuttle defect. J Neurol Sci 82: 27–39.
Pande SV, Murthy MRS 1994 Carnitine-acylcarnitine translocase deficiency: implications in human pathology. Biochim Biophys Acta 1226: 269–276.
Ruitenbeek W, Huizing M, DePinto V, Thinnes FP, Trijbels JMF, Wendel U, Sengers RCA 1995 Defects of mitochondrial membrane-bound transport proteins in human mitochondriopathies: a biochemical approach. In: Palmieri F (ed) Progress in Cell Research. Elsevier Science Publishers BV, Amsterdam, in press
Senior AE 1988 ATP synthesis by oxidative phosphorylation. Physiol Rev 68: 177–231.
Hak JB, van Beek JHGM, Westerhof N 1993 Acidosis slows the response of oxidative phosphorylation to metabolic demand in isolated rabbit heart. Pflugers Arch 423: 324–329.
Benz R 1994 Permeation of hydrophilic solutes through mitochondrial outer membranes: review on mitochondrial porins. Biochim Biophys Acta 1197: 167–196.
Sorgato MC, Moran O 1993 Channels in mitochondrial membranes: knowns, unknowns, and prospects for the future. Crit Rev Biochem Mol Biol 18: 127–171.
Dihanich M, Suta K, Schatz G 1987 A yeast mutant lacking mitochondrial porin is respiratory-deficient, but can recover respiration with simultaneous accumulation of an 86-kD extramitochondrial protein. EMBO J 6: 723–728.
Michejda J, Guo XJ, Lauquin GJ 1990 The respiration of cells and mitochondria of porin deficient yeast mutants is coupled. Biochem Biophys Res Commun 171: 354–361.
Michejda J, Kmita H, Stobienia O, Budzinska M, Lauquin GJM 1994 Restrictions of metabolite permeation through the outer mitochondrial membrane of porin-deficient yeast mutant. In: Forte M, Colombini M (eds) Molecular Biology of Mitochondrial Transport Systems. NATO ASI Series, Vol H83. Springer-Verlag Berlin, pp 341–356.
Lui MY, Torgrimson A, Colombini M 1994 Characterization and partial purification of the VDAC-channel-modulating protein from calf liver mitochondria. Biochim Biophys Acta 1185: 203–212.
Zizi M, Forte M, Blachly-Dyson E, Colombini M 1994 NADH regulates the gating of VDAC, the mitochondrial outer membrane channel. J Biol Chem 269: 1614–1616.
Zimmerberg J, Parsegian VA 1986 Polymer inaccessible volume changes during opening and closing of a voltage-dependent ionic channel. Nature 323: 36–39.
Gellerich FN, Kapischke M, Kunz W, Neumann W, Kuznetsov A, Brdiczka D, Nicolay K 1994 The influence of the cytosolic oncotic pressure on the permeability of the mitochondrial outer membrane for ADP: implications for the kinetic properties of mitochondrial creatine kinase and for ADP channelling into the intermembrane space. Mol Cell Biochem 133/134: 85–104.
McEnery MW, Dawson TM, Verma A, Gurley D, Colombini M, Synder SH 1993 Mitochondrial voltage-dependent anion channel. J Biol Chem 268: 23289–23296.
Blachly-Dyson E, Zambronicz EB, Yu WH, Adams V, McCabe ERB, Adelman J, Colombini M, Forte M 1993 Cloning and functional expression in yeast of two human isoforms of the outer mitochondrial membrane channel, the voltage-dependent anion channel. J Biol Chem 268: 1835–1841.
Blachly-Dyson E, Baldini A, Litt M, McCabe ERB, Forte M 1994 Human genes encoding the voltage-dependent anion channel (VDAC) of the outer mitochondrial membrane: mapping and identification of two new isoforms. Genomics 20: 62–67.
Ha H, Hajek P, Bedwell DM, Burrows PD 1993 A mitochondrial porin cDNA predicts the existence of multiple human porins. J Biol Chem 268: 12143–12149.
Winkelbach H, Walter G, Morys-Wortmann C, Paetzold G, Hesse D, Zimmermann B, Florke H, Reymann S, Stadtmuller U, Thinnes FP, Hilschmann N 1994 Studies on human porin. XII. Eight monoclonal mouse anti-porin 31HL antibodies discriminate type 1 and type 2 mammalian porin channels/VDACs in Western blotting and enzyme-linked immunosorbent assays. Biochem Med Metab Biol 52: 120–127.
Bureau MH, Khrestchatisky M, Heeren MA, Zambrowicz EB, Kim H, Grisar TM, Colombini M, Tobin AJ, Olsen RW 1992 Isolation and cloning of a voltage-dependent anion channel-like Mr 36,000 polypeptide from mammalian Brain. J Biol Chem 267: 8679–8684.
Dermietzel R, Hwang TK, Buettner R, Hofer A, Dotzler E, Kremer M, Deutzmann R, Thinnes FP, Fishman GI, Spray DC, Siemen D 1994 Cloning and in situ localization of a brain-derived porin that constitutes a large-conductance anion channel in astrocytic plasma membranes. Proc Natl Acad Sci USA 91: 499–503.
Thinnes FP 1992 Evidence for extra-mitochondrial localization of the VDAC/porin channel in eucaryotic cells. J Bioenerg Biomembr 24: 71–75.
Kayser H, Kratzin HD, Thinnes FP, Gotz H, Schmidt WE, Eckart K, Hilschmann N 1989 To the knowledge of human porins. Characterization and primary structure of a 31-kDa porin from human B lymphocytes (porin 31HL). Biol Chem Hoppe-Seyler 370: 1265–1278.
Konig U, Gotz H, Walter G, Babel D, Hohmeier HE, Thinnes FP, Hilschmann N 1991 Studies on human porin. V. The expression ofporin 31HL in the plasmalemma is not caused by cell transformation. Biol Chem Hoppe-Seyler 372: 565–572.
Junankar PR, Curtis SM, Pace SM, Dulhunty AF 1994 Immunogold localisation of the VDAC channel in mammalian skeletal and cardiac muscle. Proc Aust Physiol Pharmacol Soc 24: 179
Acknowledgements
The authors acknowledge U. Stadtmüller, S. Reymann, Prof. Dr. H. Götz (Göttingen, Germany) for helpful discussions and practical cooperation; Dr. G. Brandolin (Grenoble, France) and Dr. H. Bentlage (Nijmegen, The Netherlands) for providing antisera; Dr. M. S. van der Knaap for magnetic resonance imaging studies (Amsterdam, The Netherlands); and Dr. H. Brunner (Nijmegen, The Netherlands) and Prof. F. Palmieri (Bari, Italy) for stimulating discussions.
Author information
Authors and Affiliations
Additional information
Supported by the Prinses Beatrix Fonds Grants 93-018 and 95-0501 (to M.H.) and by Telethon Italy Grant 711 (to V.D.P.). The Department of Pediatrics Nijmegen is participating in the Institute of Fundamental and Clinical Human Movement Sciences (IFKB).
Rights and permissions
About this article
Cite this article
Huizing, M., Ruitenbeek, W., Thinnes, F. et al. Deficiency of the Voltage-Dependent Anion Channel: A Novel Cause of Mitochondriopathy. Pediatr Res 39, 760–765 (1996). https://doi.org/10.1203/00006450-199605000-00003
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199605000-00003
This article is cited by
-
Kastor and Polluks polypeptides encoded by a single gene locus cooperatively regulate VDAC and spermatogenesis
Nature Communications (2022)
-
Regulation of VDAC trafficking modulates cell death
Cell Death Discovery (2016)
-
The long and winding road to the mitochondrial pyruvate carrier
Cancer & Metabolism (2013)
-
VDAC contributes to mRNA levels in Saccharomyces cerevisiae cells by the intracellular reduction/oxidation state dependent and independent mechanisms
Journal of Bioenergetics and Biomembranes (2010)