Abstract
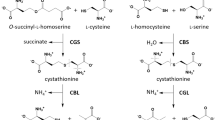
Summary: The thermostability of cystathionine β-synthase and the effect of pyridoxal phosphate (PLP) and other B6 vitamers on this thermostability were studied in extracts of cultured skin fibro-blasts from normal subjects, from heterozygotes for synthase deficiency, and from patients with homocystinuria due to synthase deficiency.
Incubation of crude extracts of normal fibroblasts at 55° (preincubation) for short periods prior to assay consistently resulted in an increase of cystathionine synthase activity (activation) that reached a maximum at 2 min (Fig. 1). Further preincubation resulted in inactivation. Addition of 0.4 mM PLP to the preincubation mixture resulted in greater activation that reached a maximum at 3–5 min. The subsequent inactivation phase proceeded at a slower rate than had occurred in the absence of PLP, resulting in a doubling of the half-life of thermal inactivation at 55°. PLP was the only one of the B6 vitamers to have a protective effect.
No significant activation of synthase was observed when extracts of fibroblasts from synthase-deficient patients were preincubated at 55°. Cells from eight patients were studied: extracts from those of four patients demonstrated activity too low to permit measurement of thermostability; extracts of cells from two patients were less thermostable than those from control subjects and showed a protective effect of PLP; extracts of cells from two other patients (sibs) showed very high heat stability in the absence of PLP, but significant inactivation of synthase in the presence of the coenzye (Fig. 2, A and A).
Three patterns of response to heating were observed with extracts of fibroblasts from seven obligate hétérozygotes (Fig. 3): 1) no activation; 2) activation with and without prior addition of PLP to the preincubation mixture; and 3) activation only in the presence of PLP. The half-life of inactivation at 55° of the extracts from heterozygotes was similar to that seen in extracts from control subjects.
Synthase from cultured long term lymphoid cells also showed activation followed by inactivation when preincubated at 55°, as well as protection by PLP.
Speculation: Heat-induced activation of cystathionine synthase is a physical characteristic of the normal enzyme molecule which is altered in enzyme from patients and from some heterozygotes for synthase deficiency. Although in vivo vitamin B6 therapy restores the ability of hepatic synthase from patients to be activated, this restoration appears not to be the result of a direct stabilizing effect of the coenzyme PLP on the apoenzyme molecule. Furthermore, since PLP does not consistently protect enzyme from patients against heat inactivation in vitro, the increase of hepatic synthase activity observed in some patients receiving megavitamin therapy may not be mediated by a protective or stabilizing effect of the coenzyme.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fleisher, L., Longhi, R., Tallan, H. et al. Cystathionine β-Synthase Deficiency: Differences in Thermostability between Normal and Abnormal Enzyme from Cultured Human Cells. Pediatr Res 12, 293–296 (1978). https://doi.org/10.1203/00006450-197804000-00009
Issue Date:
DOI: https://doi.org/10.1203/00006450-197804000-00009
Keywords
This article is cited by
-
Recent advances in the mechanism of pyridoxine‐responsive disorders
Journal of Inherited Metabolic Disease (1985)
-
Homocystinuria: Studies on cystathionine β‐synthase,S‐adenosylmethionine synthetase and cystathionase activities in skin fibroblasts
Journal of Inherited Metabolic Disease (1981)