Abstract
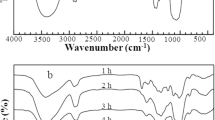
Oleoylated hemicelluloses with degrees of substitution between 0.24 and 1.67 were prepared by a rapid esterification of wheat straw hemicelluloses with oleoyl chloride using 4-dimethylaminopyridine as a catalyst and triethylamine as a neutralizer in homogeneous N,N-dimethylformamide and lithium chloride system. The effects of oleoyl chloride concentration, reaction duration, and triethylamine concentration have been studied. Under an optimum condition (sample 12, molar ratio 1 : 3), over 90% of the free hydroxyl groups in native hemicelluloses were oleoylated in 35 min at 75°C. The new materials were characterized by FT-IR. GPC, and thermal analysis. The molecular weight measurements (39700–110900) showed only a minimal degradation of hemicellulosic chains during the rapid reactions at 75°C for 15–40 min.
Similar content being viewed by others
Article PDF
References
J. Aburto, S. Thiebaud, I. Alric, E. Borredon, D. Bikiaris, J. Prinos, and C. Panayiotou, Carbohydr. Polym., 34, 101 (1998).
D. Montane, X. Farriol, J. Salvado, P. Jollez, and E. Chornet, Biomass and Bioenery, 14, 261 (1998).
B. M. Gatewood, J. C. Wu, A. S. Roberts, A. C. Lumley, and A. M. Lewis, Textile Chemist and Colorist, 30, 39 (1998).
J. M. Lawther, R. C. Sun, and W. B. Banks, J. Agric. Food Chem., 43, 667 (1995).
R. Kohn, Z. Hromadkova, and A. Ebringerova, Collection Czechoslovak Chem. Commun., 51, 2250 (1986).
C. P. J. Glaudemans and T. E. Timell, Svensk Papperstidn., 61, 1 (1958).
T. E. Timell, Adv. Carbohyd. Chem., 19, 247 (1964).
B. Philipp, W. Wagenknecht, and I. Nehls, in “Cellulose, Structural and Fundational Aspects,” J. F. Kennedy, G. O. Philipps, and P. A. Williams, Ed., Ellis Horwood, Chichester, 1989, p 173.
N. A. El-Shinnawy and S. F. El-Kalyoubi, J. Appl. Polym. Sci., 30, 2171 (1988).
C. L. McCormick and P. A. Callais, Polymer, 28, 2317 (1987).
M. Shigematsu, A. Goto, S. Yoshida, M. Tanahashi, and Y. Shinoda, Mokuzai Gakkaishi, 40, 1214 (1994).
J. F. Carson and W. D. Maclay, J. Am. Chem. Soc., 70, 293 (1948).
A. B. Blakeney, P. J. Harris, R. J. Henry, and B. A. Stone, Carbohydr. Res., 113, 291 (1983).
R. C. Sun, J. M. Fang, P. Rowlands, and J. Bolton, J. Agric. Food Chem., 46, 2804 (1998).
R. C. Sun, J. M. Lawther, and W. B. Banks, Ind. Crops Prod., 4, 127 (1995).
R. C. Sun, J. M. Lawther, and W. B. Banks, Carbohydr. Polym., 29, 325 (1996).
B. Focher, A. Marzetti, A. Naggi, and G. Torri, Makromol. Chem., 190, 129 (1989).
D. L. Williams, H. A. Pretus, R. B. McNamee, E. L. Jones, H. E. Ensley, and I. W. Browder, Carbohydr. Res., 235, 247 (1992).
A. Kato, J. Azuma, and T. Koshijima, Agric. Biol. Chem., 51, 1691 (1987).
A. Ebringerova, Z. Hromadkova, J. Alfoldi, and G. Berth, Carbohydr. Polym., 19, 99 (1992).
T. Imamura, T. Watanabe, M. Kuwahara, and T. Koshijima, Phytochemistry, 37, 1165 (1994).
M. I. Khalil, A. Hashem, and A. Hebeish, Starch/Stärke, 47, 394 (1995).
S. Gupta, R. N. Madan, and M. C. Bansal, Tappi J., 70, 113 (1987).
C. Q. Yang and X. Wang, Textile Res. J., 66, 595 (1996).
K. Rahn, M. Diamantoglou, D. Klemm, H. Berghmans, and T. Heinze, Die Angew. Makromol. Chem., 238, 143 (1996).
S. Lepeniotis and B. I. Feuer, Chemometrics and Intelligent Laboratory Systems, 36, 229 (1997).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sun, R., Tomkinson, J., Liu, J. et al. Oleoylation of Wheat Straw Hemicelluloses in New Homogeneous System. Polym J 31, 857–863 (1999). https://doi.org/10.1295/polymj.31.857
Issue Date:
DOI: https://doi.org/10.1295/polymj.31.857
Keywords
This article is cited by
-
A promising strategy for preparation of cationic xylan by environment-friendly semi-dry oven process
Fibers and Polymers (2014)