Abstract
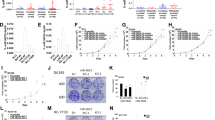
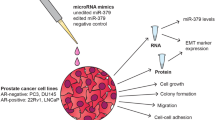
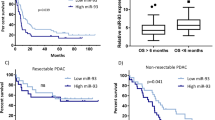
Deregulation of microRNAs (miRs) contributes to progression and metastasis of prostate and other cancers. miR-23b and -27b, encoded in the same miR cluster (miR-23b/-27b), are downregulated in human metastatic prostate cancer compared with primary tumors and benign tissue. Expression of miR-23b/-27b decreases prostate cancer cell migration, invasion and results in anoikis resistance. Conversely, antagomiR-mediated miR-23b and -27b silencing produces the opposite result in a more indolent prostate cancer cell line. However, neither miR-23b/-27b expression or inhibition impacts prostate cancer cell proliferation suggesting that miR-23b/-27b selectively suppresses metastasis. To examine the effects of miR-23b/-27b on prostate cancer metastasis in vivo, orthotopic prostate xenografts were established using aggressive prostate cancer cells transduced with miR-23b/-27b or non-targeting control miRNA. Although primary tumor formation was similar between miR-23b/-27b-transduced cells and controls, miR-23b/-27b expression in prostate cancer cells decreased seminal vesicle invasion and distant metastases. Gene-expression profiling identified the endocytic adaptor, Huntingtin-interacting protein 1-related (HIP1R) as being downregulated by miR-23b/-27b. Increased HIP1R expression in prostate cancer cells inversely phenocopied the effects of miR-23b/-27b overexpression on migration, invasion and anchorage-independent growth. HIP1R rescued miR-23b/-27b-mediated repression of migration in prostate cancer cells. HIP1R mRNA levels were decreased in seminal vesicle tissue from mice bearing miR-23b/-27b-transduced prostate cancer cell xenografts compared with scrambled controls, suggesting HIP1R is a key functional target of miR-23b/-27b. In addition, depletion of HIP1R led to a more rounded, less mesenchymal-like cell morphology, consistent with decreased metastatic properties. Together, these data demonstrate that the miR-23b/-27b cluster functions as a metastasis-suppressor by decreasing HIP1R levels in pre-clinical models of prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Jemal A, Siegel R, Xu J, Ward E . Cancer statistics, 2010. CA Cancer J Clin 2010; 60: 277–300.
Lagos-Quintana M, Rauhurt R, Lendeckel W, Tuschi T . Identification of novel genes coding for small expressed RNAs. Science 2001; 294: 853–858.
Baines AT, Xu D, Der CJ . Inhibition of Ras for cancer treatment: the search continues. Future Med Chem 2011; 3: 1787–1808.
Dalmay T, Edwards DR . MicroRNAs and the hallmarks of cancer. Oncogene 2006; 25: 6170–6175.
Ambs S, Prueitt RL, Yi M, Hudson RS, Howe TM, Petrocca F et al. Genomic profiling of microRNA and messenger RNA reveals deregulated microRNA expression in prostate cancer. Cancer Res 2008; 68: 6162–6170.
Porkka KP, Pfeiffer MJ, Waltering KK, Vessella RL, Tammela TL, Visakorpi T . MicroRNA expression profiling in prostate cancer. Cancer Res 2007; 67: 6130–6135.
Coppola V, Maria RD, Bonci D . MicroRNAs and prostate cancer. Endocr Relat Cancer 2009; 17: F1–F17.
Hurst DR, Edmonds MD, Welch DR . Metastamir: the field of metastasis-regulatory microRNA is spreading. Cancer Res 2009; 69: 7495–7498.
Dykxhoorn DM . MicroRNAs and metastasis: little RNAs go a long way. Cancer Res 2010; 70: 6401–6406.
Gandellini P, Folini M, Zaffaroni N . Towards the definition of prostate cancer-related microRNAs: where are we now? Trends Mol Med 2009; 15: 381–390.
Shi X-B, Tepper CG, White RW . microRNAs and prostate cancer. J Cell Mol Med 2008; 12: 1456–1465.
Wang Y-L, Wu S, Jiang B, Yin F-F, Zheng S-S, Hou S-C . Role of microRNAs in prostate cancer pathogenesis. Clin Genitourin Cancer 2015; 13: 261–270.
Altuvia Y, Landgraf P, Lithwick G, Elefant N, Pfeffer S, Aravin A et al. Clustering and conservation patterns of human microRNAs. Nucleic Acids Res 2005; 33: 2697–2706.
Fuse M, Kojima S, Enokida H, Chiypmaru T, Yoshino H, Nohata N et al. Tumor suppressive microRNAs (miR-222 and miR-31) regulate molecular pathways based on microRNA expression signature in prostate cancer. J Hum Genet 2012; 57: 691–699.
Martens-Uzunova ES, Jalava SE, Dits NF, van Leenders GJ, Møller S, Trapman J et al. Diagnostic and prognostic signatures from the small non-coding RNA transcriptome in prostate cancer. Oncogene 2012; 31: 978–991.
Sun T, Yang M, Chen S, Balk S, Pomerantz M, Hsieh C-L et al. The altered expression of MiR-221/-222 and MiR-23b/-27b is associated with the development of human castration resistant prostate cancer. Prostate 2012; 72: 10.
Sun T, Wang Q, Balk S, Brown M, Lee G-SM, kantoff P . The role of microRNA-221 and -222 in androgen-independent prostate cancer cell lines. Cancer Res 2009; 69: 3356–3363.
Ishteiwy RA, Ward TM, Dykxhoorn DM, Burnstein KL . The microRNA-23b/-27b cluster suppresses the metastatic phenotype of castration-resistant prostate cancer cells. PLoS One 2012; 7: e52106.
Stephenson RA, Dinney CP, Gohji K, Ordonez NG, Killion JJ, Fidler IJ . Metastatic model for human prostate cancer using orthotopic implantation in nude mice. J Natl Cancer Inst 1992; 84: 951–957.
Liu W, Vivian CJ, Brinker AE, Hampton KR, Lianidou E, Welch DR . Microenvironmental influences on metastasis suppressor expression and function during a metastatic cell’s journey. Cancer Microenviron 2014; 7: 117–131.
Yates TJ, Lopez LE, Lokeshwar SD, Ortiz N, Kallifatidis G, Jordan A et al. Dietary supplement 4-methylumbelliferone: an effective chemopreventive and therapeutic agent for prostate cancer. J Natl Cancer Inst 2015; 107: 7.
Brett TJ, Legendre-Guillemin V, McPherson PS, Fremont DH . Structural definition of the F-actin-binding THATCH domain from HIP1R. Nat Struct Mol Biol 2006; 13: 121–130.
O'Day E, Lal A . MicroRNAs and their target gene networks in breast cancer. Breast Cancer Res 2010; 12: 201.
Howe EN, Cochrane DR, Richer JK . Targets of miR-200c mediate suppression of cell motility and anoikis resistance. Breast Cancer Res 2011; 13: R45.
D'Amico AV, Whittington R, Malkowicz SB, Schultz D, Schnall M, Tomaszewski JE et al. A multivariate analysis of clinical and pathological factors that predict for prostate specific antigen failure after radical prostatectomy for prostate cancer. J Urol 1995; 154: 131–138.
Thompson DW, Bracken CP, Goodall GJ . Experimental strategies for microRNA target identification. Nucleic Acids Res 2011; 39: 6845–6853.
Tay Y, Zhang J, Thomson AM, Lim B, Rigoutsos I . MicroRNAs to Nanog, Oct4 and Sox2 coding regions modulate embryonic stem cell differentiation. Nature 2008; 455: 1124–1128.
Engqvist-Goldstein AE, Zhang CX, Carreno S, Barroso C, Heuser JE, Drubin DG . RNAi-mediated Hip1R silencing results in stable association between the endocytic machinery and the actin assembly machinery. Mol Biol Cell 2004; 15: 1666–1679.
Gottfried I, Ehrlich M, Ashery U . The Sla2p/HIP1/HIP1R family: similar structure, similar function in endocytosis? Biochem Soc Trans 2010; 38: 187–191.
Seki N, Muramatsu M, Sugano S, Suzuki Y, Nakagawara A, Ohhira M et al. Cloning, expression analysis, and chromosomal localization of HIP1R, an isolog of huntingtin interacting protein (HIP1). J Hum Genet 1998; 43: 268–271.
Boettner DR, Chi RJ, Lemmon SK . Lessons from yeast for clathrin-mediated endocytosis. Nat Cell Biol 2012; 14: 2–10.
Hyun TS, Rao DS, Saint-Dic D, Michael LE, Kumar PD, Bradley SV et al. HIP1 and HIP1r stabilize receptor tyrosine kinases and bind 3-phosphoinositides via epsin N-terminal homology domains. J Biol Chem 2004; 279: 14294–14306.
Hyun TS, Li L, Oravecz-Wilson KI, Bradley SV, Provot MM, Munaco AJ et al. Hip1-related mutant mice grow and develop normally but have accelerated spinal abnormalities and dwarfism in the absence of HIP1. Mol Cell Biol 2004; 24: 4329–4340.
Rao DS, Hyun TS, Kumar PD, Mizukami IF, Rubin MA, Lucas PC et al. Huntingtin-interacting protein 1 is overexpressed in prostate and colon cancer and is critical for cellular survival. J Clin Invest 2002; 110: 351–360.
Wang J, Yu W, Cai Y, Ren C, Ittmann MM . Altered fibroblast growth factor receptor 4 stability promotes prostate cancer progression. Neoplasia 2008; 10: 847–856.
Mosesson Y, Mills GB, Yarden Y . Derailed endocytosis: an emerging feature of cancer. Nat Rev Cancer 2008; 8: 835–850.
Loop SM, Rozanski TA, Ostenson RC . Human primary prostate tumor cell line, ALVA-21: a new model for studying the hormonal regulation of prostate tumor cell growth. Prostate 1993; 22: 93–108.
Wang M, Stearns M . Isolation and characterization of PC-3 human prostatic tumor sublines which preferentially metastasize to select organs in S.C.I.D mice. Differentiation 1991; 48: 115–125.
D'Ambrosio J, Fatatis A . Osteoblasts modulate Ca2+ signaling in bone-metastatic prostate and breast cancer cells. Clin Exp Metastasis 2009; 26: 955–964.
Peacock SO, Fahrenholtz CD, Burnstein KL . Vav3 enhances androgen receptor splice variant activity and is critical for castration-resistant prostate cancer growth and survival. Mol Endocrinol 2012; 26: 1967–1979.
Engqvist-Goldstein AE, Kessels MM, Chopra VS, Hayden MR, Drubin DG . An Actin-binding protein of the Sla2/Huntingtin interacting protein 1 family is a novel component of clathrin-coated pits and vesicles. J Cell Biol 1999; 147: 1503–1518.
Lyons LS, Rao S, Balkan W, Faysal J, Maiorino CA, Burnstein KL . Ligand-independent activation of androgen receptors by Rho GTPase signaling in prostate cancer. Mol Endocrinol 2008; 22: 597–608.
Grasso CS, Wu Y, Robinson DR, Cao X, Dhanasekaran SM, Khan AP et al. The mutational landscape of lethal castration-resistant prostate cancer. Nature 2012; 487: 239–243.
Chandran UR, Ma C, Dhir R, Bisceglia M, Lyons-Weiler M, Liang W et al. Gene expression profiles of prostate cancer reveal involvement of multiple molecular pathways in the metastatic process. BMC Cancer 2007; 7: 64.
Yu YP, Landsittel D, Jing L, Ren B, Liu L, McDonald C et al. Gene expression alterations in prostate cancer predicting tumor aggression and preceding development of malignancy. J Clin Oncol 2004; 22: 2790–2799.
Acknowledgements
Funding provided by DOD pre-doctoral fellowship W81XWH-11-1-0314 (RAI/MAR), NIH R01CA132200 and Women’s Cancer Association of the University of Miami (KLB). We thank UM investigators Drs Cale Fahrenholtz, Stephanie Peacock and Ning Zhao, as well as Annie Greene and Govindi Samaranayake for helpful technical assistance and guidance. Dr Michael Henry at the University of Iowa and Dr David Drubin at University of California at Berkeley generously provided the PQCXIN-luciferase and HIP1R-6myc constructs respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Rice, M., Ishteiwy, R., Magani, F. et al. The microRNA-23b/-27b cluster suppresses prostate cancer metastasis via Huntingtin-interacting protein 1-related. Oncogene 35, 4752–4761 (2016). https://doi.org/10.1038/onc.2016.6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.6
This article is cited by
-
Quantitative proteomics analysis in small cell carcinoma of cervix reveals novel therapeutic targets
Clinical Proteomics (2023)
-
Flurbiprofen inhibits cell proliferation in thyroid cancer through interrupting HIP1R-induced endocytosis of PTEN
European Journal of Medical Research (2022)
-
The miR-23–27–24 cluster: an emerging target in NAFLD pathogenesis
Acta Pharmacologica Sinica (2022)
-
A novel miRNA inhibits metastasis of prostate cancer via decreasing CREBBP-mediated histone acetylation
Journal of Cancer Research and Clinical Oncology (2021)
-
Reduced Arginyltransferase 1 is a driver and a potential prognostic indicator of prostate cancer metastasis
Oncogene (2019)