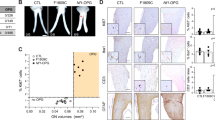
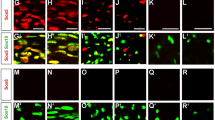
Abstract
Neurofibromatosis type 1 (NF1) is an inherited disease in which affected patients are predisposed to develop benign Schwann cell (SC) tumours called neurofibromas. In the mouse, loss of Nf1 in the SC lineage causes neurofibroma formation. The tyrosine kinase receptor EGFR is expressed in Schwann cell precursors (SCP), which have been implicated in plexiform neurofibroma initiation. To test if EGFR activity affects neurofibroma initiation, size, and/or number, we studied mice expressing human EGFR in SCs and SCP in the context of mice that form neurofibromas. Neurofibroma number increased in homozygous CNP-hEGFR mice versus heterozygous littermates, and neurofibroma number and size increased when CNP-hEGFR was crossed to Nf1fl/fl;DhhCre mice. Conversely, diminished EGFR signalling in Nf1fl/fl;DhhCre;Wa2/+ mice decreased neurofibroma number. In vivo transplantation verified the correlation between EGFR activity and neurofibroma formation. Mechanistically, expression of CNP-hEGFR increased SCP/neurofibroma-initiating cell self-renewal, a surrogate for tumour initiation, and activated P-Stat3. Further, Il-6 reinforced Jak2/Stat3 activation in SCPs and SCs. These gain- and loss-of function assays show that levels of tyrosine kinase expression in SCPs modify neurofibroma initiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Boyd KP, Korf BR, Theos A . Neurofibromatosis type 1. J Am Acad Dermatol 2009; 61: 1–14.
Zhu Y, Ghosh P, Charnay P, Burns D, Parada L . Neurofibromas in NF1: Schwann cell origin and role of tumor environment. Science 2002; 296: 920–922.
Rizvi T, Huang Y, Sidani A, Atit R, Largaespada D, Boissy R et al. A novel cytokine pathway suppresses glial cell melanogenesis after injury to adult nerve. J Neurosci 2002; 22: 9831–9840.
Ribeiro S, Napoli I, White IJ, Parrinello S, Flanagan AM, Suter U et al. Injury signals cooperate with Nf1 loss to relieve the tumor-suppressive environment of adult peripheral nerve. Cell Rep 2013; 5: 126–136.
Vogel KS, Klesse LJ, Velasco-Miguel S, Meyers K, Rushing EJ, Parada LF . Mouse tumor model for neurofibromatosis type 1. Science 1999; 286: 2176–2179.
Wu J, Williams JP, Rizvi TA, Kordich JJ, Witte D, Meijer D et al. Plexiform and dermal neurofibromas and pigmentation are caused by Nf1 loss in desert hedgehog-expressing cells. Cancer Cell 2008; 13: 105–116.
Mayes DA, Rizvi TA, Cancelas JA, Kolasinski NT, Ciraolo GM, Stemmer-Rachamimov AO et al. Perinatal or adult Nf1 inactivation using tamoxifen-inducible PlpCre each cause neurofibroma formation. Cancer Res 2011; 71: 4675–4685.
Le L, Liu C, Shipman T, Chen Z, Suter U, Parada L . Susceptible stages in Schwann cells for NF1-associated plexiform neurofibroma development. Cancer Res 2011; 71: 4686–4695.
Chen Z, Liu C, Patel AJ, Liao CP, Wang Y, Le LQ . Cells of origin in the embryonic nerve roots for NF1-associated plexiform neurofibroma. Cancer Cell 2014; 26: 695–706.
Carroll SL, Ratner N . How does the Schwann cell lineage form tumors in NF1? Glia 2008; 56: 1590–1605.
Gomez-Sanchez JA, Lopez de Armentia M, Lujan R, Kessaris N, Richardson WD, Cabedo H . Sustained axon-glial signaling induces Schwann cell hyperproliferation, Remak bundle myelination, and tumorigenesis. J Neurosci 2009; 29: 11304–11315.
Huijbregts R, Roth K, Schmidt R, Carroll S . Hypertrophic neuropathies and malignant peripheral nerve sheath tumors in transgenic mice overexpressing glial growth factor beta3 in myelinating Schwann cells. J Neurosci 2003; 23: 7269–7280.
Ling B, Wu J, Miller S, Monk K, Shamekh R, Rizvi T et al. Role for the epidermal growth factor receptor in neurofibromatosis-related peripheral nerve tumorigenesis. Cancer Cell 2005; 7: 65–75.
Holtkamp N, Malzer E, Zietsch J, Okuducu AF, Mucha J, Mawrin C et al. EGFR and erbB2 in malignant peripheral nerve sheath tumors and implications for targeted therapy. Neuro-Oncol 2008; 10: 946–957.
DeClue JE, Heffelfinger S, Benvenuto G, Ling B, Li S, Rui W et al. Epidermal growth factor receptor expression in neurofibromatosis type-1 related tumors and NF1 animal models. J Clin Invest 2000; 105: 1233–1241.
Li H, Zhao X, Yan X, Jessen WJ, Kim MO, Dombi E et al. Runx1 contributes to neurofibromatosis type 1 neurofibroma formation. Oncogene 2016; 35: 1468–1474.
Yarden Y . The EGFR family and its ligands in human cancer. Signalling mechanisms and therapeutic opportunities. Eur J Cancer 2001; 37 (Suppl): S3–S8.
Williams JP, Wu J, Johansson G, Rizvi TA, Miller SC, Geiger H et al. Nf1 mutation expands an EGFR-dependent peripheral nerve progenitor that confers neurofibroma tumorigenic potential. Cell Stem Cell 2008; 3: 658–669.
Wu J, Keng VW, Patmore DM, Kendall JJ, Patel AV, Jousma E et al. Insertional mutagenesis identifies a STAT3/Arid1b/beta-catenin pathway driving neurofibroma initiation. Cell Rep 2016; 14: 1979–1990.
Battle TE, Frank DA . The role of STATs in apoptosis. Curr Mol Med 2002; 2: 381–392.
Wu J, Crimmins J, Monk K, Williams J, Fitzgerald M, Tedesco S et al. Perinatal epidermal growth factor receptor blockade prevents peripheral nerve disruption in a mouse model reminiscent of benign world health organization grade I neurofibroma. Am J Pathol 2006; 168: 1686–1696.
Wu J, Patmore DM, Jousma E, Eaves DW, Breving K, Patel AV et al. EGFR-STAT3 signaling promotes formation of malignant peripheral nerve sheath tumors. Oncogene 2014; 33: 173–180.
Luetteke N, Phillips H, Qiu T, Copeland N, Earp H, Jenkins N et al. The mouse waved-2 phenotype results from a point mutation in the EGF receptor tyrosine kinase. Genes Dev 1994; 8: 399–413.
Joseph NM, Mosher JT, Buchstaller J, Snider P, McKeever PE, Lim M et al. The loss of Nf1 transiently promotes self-renewal but not tumorigenesis by neural crest stem cells. Cancer Cell 2008; 13: 129–140.
Li H, Velasco-Miguel S, Vass W, Parada L, DeClue J . Epidermal growth factor receptor signaling pathways are associated with tumorigenesis in the Nf1:p53 mouse tumor model. Cancer Res 2002; 62: 4507–4513.
Jessen WJ, Miller SJ, Jousma E, Wu J, Rizvi TA, Brundage ME et al. MEK inhibition exhibits efficacy in human and mouse neurofibromatosis tumors. J Clin Invest 2013; 123: 340–347.
Wang L, Lee HK, Seo IA, Shin YK, Lee KY, Park HT . Cell type-specific STAT3 activation by gp130-related cytokines in the peripheral nerves. Neuroreport 2009; 20: 663–668.
Zheng H, Chang L, Patel N, Yang J, Lowe L, Burns DK et al. Induction of abnormal proliferation by nonmyelinating Schwann cells triggers neurofibroma formation. Cancer Cell 2008; 13: 117–128.
Chen S, Rio C, Ji RR, Dikkes P, Coggeshall RE, Woolf CJ et al. Disruption of ErbB receptor signaling in adult non-myelinating Schwann cells causes progressive sensory loss. Nat Neurosci 2003; 6: 1186–1193.
Bos JL, Rehmann H, Wittinghofer A . GEFs and GAPs: critical elements in the control of small G proteins. Cell 2007; 129: 865–877.
De Bruin EC, Cowell C, Warne PH, Jiang M, Saunders RE, Melnick MA et al. Reduced NF1 expression confers resistance to EGFR inhibition in lung cancer. Cancer Discov 2014; 4: 606–619.
Harvey M, McArthur MJ, Montgomery CA Jr., Bradley A, Donehower LA . Genetic background alters the spectrum of tumors that develop in p53-deficient mice. FASEB J 1993; 7: 938–943.
Walrath JC, Fox K, Truffer E, Gregory Alvord W, Quinones OA, Reilly KM . Chr 19(A/J) modifies tumor resistance in a sex- and parent-of-origin-specific manner. Mamm Genome 2009; 20: 214–223.
Kazmi SJ, Byer SJ, Eckert JM, Turk AN, Huijbregts RP, Brossier NM et al. Transgenic mice overexpressing neuregulin-1 model neurofibroma-malignant peripheral nerve sheath tumor progression and implicate specific chromosomal copy number variations in tumorigenesis. Am J Pathol 2013; 182: 646–667.
Rieley MB, Stevenson DA, Viskochil DH, Tinkle BT, Martin LJ, Schorry EK . Variable expression of neurofibromatosis 1 in monozygotic twins. Am J Med Genet 2011; 155A: 478–485.
Upadhyaya M, Huson SM, Davies M, Thomas N, Chuzhanova N, Giovannini S et al. An absence of cutaneous neurofibromas associated with a 3-bp inframe deletion in exon 17 of the NF1 gene (c.2970-2972 delAAT): evidence of a clinically significant NF1 genotype-phenotype correlation. Am J Hum Genet 2007; 80: 140–151.
Leppig KA, Kaplan P, Viskochil D, Weaver M, Ortenberg J, Stephens K . Familial neurofibromatosis 1 microdeletions: cosegregation with distinct facial phenotype and early onset of cutaneous neurofibromata. Am J Med Genet 1997; 73: 197–204.
Mautner VF, Kluwe L, Friedrich RE, Roehl AC, Bammert S, Hogel J et al. Clinical characterisation of 29 neurofibromatosis type-1 patients with molecularly ascertained 1.4 Mb type-1 NF1 deletions. J Med Genet 2010; 47: 623–630.
Easton DF, Ponder MA, Huson SM, Ponder BA . An analysis of variation in expression of neurofibromatosis (NF) type 1 (NF1): evidence for modifying genes. Am J Hum Genet 1993; 53: 305–313.
Ryan MA, Nattamai KJ, Xing E, Schleimer D, Daria D, Sengupta A et al. Pharmacological inhibition of EGFR signaling enhances G-CSF-induced hematopoietic stem cell mobilization. Nat Med 2010; 16: 1141–1146.
Lesina M, Kurkowski M, Ludes K, Rose-John S, Treiber M, Klöppel G et al. Stat3/Socs3 activation by IL-6 transsignaling promotes progression of pancreatic intraepithelial neoplasia and development of pancreatic cancer. Cancer Cell 2011; 19: 456–469.
Corcoran RB, Contino G, Deshpande V, Tzatsos A, Conrad C, Benes CH et al. STAT3 plays a critical role in KRAS-induced pancreatic tumorigenesis. Cancer Res 2011; 71: 5020–5029.
Ratner N, Williams JP, Kordich JJ, Kim HA . Schwann cell preparation from single mouse embryos: analyses of neurofibromin function in Schwann cells. Methods Enzymol 2006; 407:22–33.
Stemmer-Rachamimov A, Louis D, Nielsen G, Antonescu C, Borowsky A, Bronson R et al. Comparative pathology of nerve sheath tumors in mouse models and humans. Cancer Res 2004; 64: 3718–3724.
Acknowledgements
We thank Dr Luis F Parada (Memorial Sloan Kettering, NY) for Nf1fl/fl mice and Dr Anat O Stemmer-Rachamimov (Massachusetts General Hospital) for assistance with mouse pathology. We thank Dr Adam Lane (CCHMC) for consultation on statistical analysis. This work was supported by a DAMD New Investigator Award (W81XWH-11-1-0259) (Jianqiang Wu) and NIH R01 NS28840 (Nancy Ratner).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Wu, J., Liu, W., Williams, J. et al. EGFR-Stat3 signalling in nerve glial cells modifies neurofibroma initiation. Oncogene 36, 1669–1677 (2017). https://doi.org/10.1038/onc.2016.386
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.386
This article is cited by
-
Translating current basic research into future therapies for neurofibromatosis type 1
British Journal of Cancer (2020)
-
CUL1 Knockdown Attenuates the Adhesion, Invasion, and Migration of Triple-Negative Breast Cancer Cells via Inhibition of Epithelial-Mesenchymal Transition
Pathology & Oncology Research (2020)
-
STAT3 inhibition reduces macrophage number and tumor growth in neurofibroma
Oncogene (2019)
-
A nanomedicine approach enables co-delivery of cyclosporin A and gefitinib to potentiate the therapeutic efficacy in drug-resistant lung cancer
Signal Transduction and Targeted Therapy (2018)