Abstract
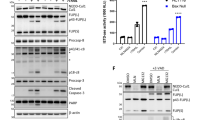
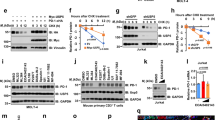
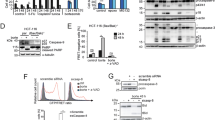
FLICE-like inhibitory protein (FLIP) is a critical regulator of death receptor-mediated apoptosis. Here, we found ubiquitin-specific peptidase 8 (USP8) to be a novel deubiquitylase of the long isoform of FLIP (FLIPL). USP8 directly deubiquitylates and stabilizes FLIPL, but not the short isoform. USP8 depletion induces FLIPL destabilization, promoting anti-Fas-, tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)- and tumor necrosis factor alpha-induced extrinsic apoptosis by facilitating death-inducing signaling complex or TNFR1 complex II formation, which results in the activation of caspase-8 and caspase-3. USP8 mRNA levels are elevated in melanoma and cervical cancers, and the protein levels of USP8 and FLIPL are positively correlated in these cancer cell lines. Xenograft analyses using ME-180 cervical cancer cells showed that USP8 depletion attenuated tumor growth upon TRAIL injection. Taken together, our data indicate that USP8 functions as a novel deubiquitylase of FLIPL and inhibits extrinsic apoptosis by stabilizing FLIPL.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Mahmood Z, Shukla Y . Death receptors: targets for cancer therapy. Exp Cell Res 2010; 316: 887–899.
French LE, Tschopp J . Protein-based therapeutic approaches targeting death receptors. Cell Death Differ 2003; 10: 117–123.
Wajant H . Death receptors. Essays Biochem 2003; 39: 53–71.
Kischkel FC, Hellbardt S, Behrmann I, Germer M, Pawlita M, Krammer PH et al. Cytotoxicity-dependent APO-1 (Fas/CD95)-associated proteins form a death-inducing signaling complex (DISC) with the receptor. EMBO J 1995; 14: 5579–5588.
Dickens LS, Boyd RS, Jukes-Jones R, Hughes MA, Robinson GL, Fairall L et al. A death effector domain chain DISC model reveals a crucial role for caspase-8 chain assembly in mediating apoptotic cell death. Mol Cell 2012; 47: 291–305.
Schleich K, Warnken U, Fricker N, Ozturk S, Richter P, Kammerer K et al. Stoichiometry of the CD95 death-inducing signaling complex: experimental and modeling evidence for a death effector domain chain model. Mol Cell 2012; 47: 306–319.
Lavrik I, Golks A, Krammer PH . Death receptor signaling. J Cell Sci 2005; 118: 265–267.
Wilson NS, Dixit V, Ashkenazi A . Death receptor signal transducers: nodes of coordination in immune signaling networks. Nat Immunol 2009; 10: 348–355.
Symons A, Beinke S, Ley SC . MAP kinase kinase kinases and innate immunity. Trends Immunol 2006; 27: 40–48.
Varfolomeev EE, Ashkenazi A . Tumor necrosis factor: an apoptosis JuNKie? Cell 2004; 116: 491–497.
Wang L, Du F, Wang X . TNF-alpha induces two distinct caspase-8 activation pathways. Cell 2008; 133: 693–703.
Schutze S, Tchikov V, Schneider-Brachert W . Regulation of TNFR1 and CD95 signalling by receptor compartmentalization. Nat Rev Mol Cell Biol 2008; 9: 655–662.
Yu JW, Shi Y . FLIP and the death effector domain family. Oncogene 2008; 27: 6216–6227.
Tschopp J, Irmler M, Thome M . Inhibition of fas death signals by FLIPs. Curr Opin Immunol 1998; 10: 552–558.
Inohara N, Koseki T, Hu Y, Chen S, Nunez G . CLARP, a death effector domain-containing protein interacts with caspase-8 and regulates apoptosis. Proc Natl Acad Sci U S A 1997; 94: 10717–10722.
Hu S, Vincenz C, Ni J, Gentz R, Dixit VM . I-FLICE, a novel inhibitor of tumor necrosis factor receptor-1- and CD-95-induced apoptosis. J Biol Chem 1997; 272: 17255–17257.
Irmler M, Thome M, Hahne M, Schneider P, Hofmann K, Steiner V et al. Inhibition of death receptor signals by cellular FLIP. Nature 1997; 388: 190–195.
Goltsev YV, Kovalenko AV, Arnold E, Varfolomeev EE, Brodianskii VM, Wallach D . CASH a novel caspase homologue with death effector domains. J Biol Chem 1997; 272: 19641–19644.
Srinivasula SM, Ahmad M, Ottilie S, Bullrich F, Banks S, Wang Y et al. FLAME-1, a novel FADD-like anti-apoptotic molecule that regulates Fas/TNFR1-induced apoptosis. J Biol Chem 1997; 272: 18542–18545.
Shu HB, Halpin DR, Goeddel DV . Casper is a FADD- and caspase-related inducer of apoptosis. Immunity 1997; 6: 751–763.
Thome M, Tschopp J . Regulation of lymphocyte proliferation and death by FLIP. Nat Rev Immunol 2001; 1: 50–58.
Krueger A, Schmitz I, Baumann S, Krammer PH, Kirchhoff S . Cellular FLICE-inhibitory protein splice variants inhibit different steps of caspase-8 activation at the CD95 death-inducing signaling complex. J Biol Chem 2001; 276: 20633–20640.
Micheau O, Thome M, Schneider P, Holler N, Tschopp J, Nicholson DW et al. The long form of FLIP is an activator of caspase-8 at the Fas death-inducing signaling complex. J Biol Chem 2002; 277: 45162–45171.
Boatright KM, Deis C, Denault JB, Sutherlin DP, Salvesen GS . Activation of caspases-8 and -10 by FLIP(L). Biochem J 2004; 382: 651–657.
Pop C, Oberst A, Drag M, Van Raam BJ, Riedl SJ, Green DR et al. FLIP(L) induces caspase 8 activity in the absence of interdomain caspase 8 cleavage and alters substrate specificity. Biochem J 2011; 433: 447–457.
Weinlich R, Dillon CP, Green DR . Ripped to death. Trends Cell Biol 2011; 21: 630–637.
Hughes MA, Powley IR, Jukes-Jones R, Horn S, Feoktistova M, Fairall L et al. Co-operative and hierarchical binding of c-FLIP and caspase-8: a unified model defines how c-FLIP isoforms differentially control cell fate. Mol Cell 2016; 61: 834–849.
Panka DJ, Mano T, Suhara T, Walsh K, Mier JW . Phosphatidylinositol 3-kinase/Akt activity regulates c-FLIP expression in tumor cells. J Biol Chem 2001; 276: 6893–6896.
Kreuz S, Siegmund D, Scheurich P, Wajant H . NF-kappaB inducers upregulate cFLIP, a cycloheximide-sensitive inhibitor of death receptor signaling. Mol Cell Biol 2001; 21: 3964–3973.
Yerbes R, Lopez-Rivas A, Reginato MJ, Palacios C . Control of FLIP(L) expression and TRAIL resistance by the extracellular signal-regulated kinase1/2 pathway in breast epithelial cells. Cell Death Differ 2012; 19: 1908–1916.
Chang L, Kamata H, Solinas G, Luo JL, Maeda S, Venuprasad K et al. The E3 ubiquitin ligase itch couples JNK activation to TNFalpha-induced cell death by inducing c-FLIP(L) turnover. Cell 2006; 124: 601–613.
Chanvorachote P, Nimmannit U, Wang L, Stehlik C, Lu B, Azad N et al. Nitric oxide negatively regulates Fas CD95-induced apoptosis through inhibition of ubiquitin-proteasome-mediated degradation of FLICE inhibitory protein. J Biol Chem 2005; 280: 42044–42050.
Kaunisto A, Kochin V, Asaoka T, Mikhailov A, Poukkula M, Meinander A et al. PKC-mediated phosphorylation regulates c-FLIP ubiquitylation and stability. Cell Death Differ 2009; 16: 1215–1226.
Poukkula M, Kaunisto A, Hietakangas V, Denessiouk K, Katajamaki T, Johnson MS et al. Rapid turnover of c-FLIPshort is determined by its unique C-terminal tail. J Biol Chem 2005; 280: 27345–27355.
Kim Y, Suh N, Sporn M, Reed JC . An inducible pathway for degradation of FLIP protein sensitizes tumor cells to TRAIL-induced apoptosis. J Biol Chem 2002; 277: 22320–22329.
Perez D, White E . E1A sensitizes cells to tumor necrosis factor alpha by downregulating c-FLIP S. J Virol 2003; 77: 2651–2662.
Fukazawa T, Fujiwara T, Uno F, Teraishi F, Kadowaki Y, Itoshima T et al. Accelerated degradation of cellular FLIP protein through the ubiquitin-proteasome pathway in p53-mediated apoptosis of human cancer cells. Oncogene 2001; 20: 5225–5231.
Naviglio S, Mattecucci C, Matoskova B, Nagase T, Nomura N, Di Fiore PP et al. UBPY: a growth-regulated human ubiquitin isopeptidase. EMBO J 1998; 17: 3241–3250.
Byun S, Lee SY, Lee J, Jeong CH, Farrand L, Lim S et al. USP8 is a novel target for overcoming gefitinib resistance in lung cancer. Clin Cancer Res 2013; 19: 3894–3904.
Mizuno E, Iura T, Mukai A, Yoshimori T, Kitamura N, Komada M . Regulation of epidermal growth factor receptor down-regulation by UBPY-mediated deubiquitination at endosomes. Mol Biol Cell 2005; 16: 5163–5174.
Niendorf S, Oksche A, Kisser A, Lohler J, Prinz M, Schorle H et al. Essential role of ubiquitin-specific protease 8 for receptor tyrosine kinase stability and endocytic trafficking in vivo. Mol Cell Biol 2007; 27: 5029–5039.
Shirley S, Micheau O . Targeting c-FLIP in cancer. Cancer Lett 2013; 332: 141–150.
Kavuri SM, Geserick P, Berg D, Dimitrova DP, Feoktistova M, Siegmund D et al. Cellular FLICE-inhibitory protein (cFLIP) isoforms block CD95- and TRAIL death receptor-induced gene induction irrespective of processing of caspase-8 or cFLIP in the death-inducing signaling complex. J Biol Chem 2011; 286: 16631–16646.
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 2004; 6: 1–6.
Yeh WC, Itie A, Elia AJ, Ng M, Shu HB, Wakeham A et al. Requirement for casper (c-FLIP) in regulation of death receptor-induced apoptosis and embryonic development. Immunity 2000; 12: 633–642.
Dillon CP, Oberst A, Weinlich R, Janke LJ, Kang TB, Ben-Moshe T et al. Survival function of the FADD-CASPASE-8-cFLIP(L) complex. Cell Rep 2012; 1: 401–407.
Vandenabeele P, Melino G . The flick of a switch: which death program to choose? Cell Death Differ 2012; 19: 1093–1095.
Feoktistova M, Geserick P, Kellert B, Dimitrova DP, Langlais C, Hupe M et al. cIAPs block ripoptosome formation, a RIP1/caspase-8 containing intracellular cell death complex differentially regulated by cFLIP isoforms. Mol Cell 2011; 43: 449–463.
Tenev T, Bianchi K, Darding M, Broemer M, Langlais C, Wallberg F et al. The Ripoptosome, a signaling platform that assembles in response to genotoxic stress and loss of IAPs. Mol Cell 2011; 43: 432–448.
Oberst A, Dillon CP, Weinlich R, McCormick LL, Fitzgerald P, Pop C et al. Catalytic activity of the caspase-8-FLIP(L) complex inhibits RIPK3-dependent necrosis. Nature 2011; 471: 363–367.
O'Donnell MA, Perez-Jimenez E, Oberst A, Ng A, Massoumi R, Xavier R et al. Caspase 8 inhibits programmed necrosis by processing CYLD. Nat Cell Biol 2011; 13: 1437–1442.
Geserick P, Hupe M, Moulin M, Wong WW, Feoktistova M, Kellert B et al. Cellular IAPs inhibit a cryptic CD95-induced cell death by limiting RIP1 kinase recruitment. J Cell Biol 2009; 187: 1037–1054.
Panner A, Crane CA, Weng C, Feletti A, Fang S, Parsa AT et al. Ubiquitin-specific protease 8 links the PTEN-Akt-AIP4 pathway to the control of FLIPS stability and TRAIL sensitivity in glioblastoma multiforme. Cancer Res 2010; 70: 5046–5053.
den Hollander MW, Gietema JA, de Jong S, Walenkamp AM, Reyners AK, Oldenhuis CN et al. Translating TRAIL-receptor targeting agents to the clinic. Cancer Lett 2013; 332: 194–201.
Stuckey DW, Shah K . TRAIL on trial: preclinical advances in cancer therapy. Trends Mol Med 2013; 19: 685–694.
Ashkenazi A, Pai RC, Fong S, Leung S, Lawrence DA, Marsters SA et al. Safety and antitumor activity of recombinant soluble Apo2 ligand. J Clin Invest 1999; 104: 155–162.
Maksimovic-Ivanic D, Stosic-Grujicic S, Nicoletti F, Mijatovic S . Resistance to TRAIL and how to surmount it. Immunol Res 2012; 52: 157–168.
Soria JC, Mark Z, Zatloukal P, Szima B, Albert I, Juhasz E et al. Randomized phase II study of dulanermin in combination with paclitaxel, carboplatin, and bevacizumab in advanced non-small-cell lung cancer. J Clin Oncol 2011; 29: 4442–4451.
Soria JC, Smit E, Khayat D, Besse B, Yang X, Hsu CP et al. Phase 1b study of dulanermin (recombinant human Apo2L/TRAIL) in combination with paclitaxel, carboplatin, and bevacizumab in patients with advanced non-squamous non-small-cell lung cancer. J Clin Oncol 2010; 28: 1527–1533.
Oh YM, Lee SB, Choi J, Suh HY, Shim S, Song YJ et al. USP8 modulates ubiquitination of LRIG1 for Met degradation. Sci Rep 2014; 4: 4980.
Guedat P, Colland F . Patented small molecule inhibitors in the ubiquitin proteasome system. BMC Biochem 2007; 8: S14.
Pyeon D, Newton MA, Lambert PF, den Boon JA, Sengupta S, Marsit CJ et al. Fundamental differences in cell cycle deregulation in human papillomavirus-positive and human papillomavirus-negative head/neck and cervical cancers. Cancer Res 2007; 67: 4605–4619.
Talantov D, Mazumder A, Yu JX, Briggs T, Jiang Y, Backus J et al. Novel genes associated with malignant melanoma but not benign melanocytic lesions. Clin Cancer Res 2005; 11: 7234–7242.
Lee EW, Seong D, Seo J, Jeong M, Lee HK, Song J . USP11-dependent selective cIAP2 deubiquitylation and stabilization determine sensitivity to Smac mimetics. Cell Death Differ 2015; 22: 1463–1476.
Acknowledgements
This work was supported by a grant from the National Research Foundation of Korea (NRF), funded by the Ministry of Science, ICT and Future Planning (NRF-2015R1A3A2066581) (to J Song) and by the Ministry of Education (2012R1A6A3A04040105) (to E-WL).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website .
Supplementary information
Rights and permissions
About this article
Cite this article
Jeong, M., Lee, EW., Seong, D. et al. USP8 suppresses death receptor-mediated apoptosis by enhancing FLIPL stability. Oncogene 36, 458–470 (2017). https://doi.org/10.1038/onc.2016.215
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.215
This article is cited by
-
Spotlights on ubiquitin-specific protease 12 (USP12) in diseases: from multifaceted roles to pathophysiological mechanisms
Journal of Translational Medicine (2023)
-
USP8 inhibitor–induced DNA damage activates cell cycle arrest, apoptosis, and autophagy in esophageal squamous cell carcinoma
Cell Biology and Toxicology (2023)
-
USP8 inhibition reshapes an inflamed tumor microenvironment that potentiates the immunotherapy
Nature Communications (2022)
-
The emerging role of ubiquitin-specific protease 20 in tumorigenesis and cancer therapeutics
Cell Death & Disease (2022)
-
Pro-apoptotic and anti-apoptotic regulation mediated by deubiquitinating enzymes
Cellular and Molecular Life Sciences (2022)