Abstract
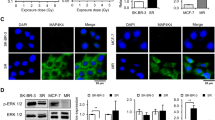
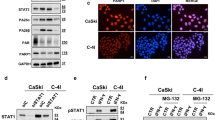
Radiation therapy is a staple approach for cancer treatment, whereas radioresistance of cancer cells remains a substantial clinical problem. In response to ionizing radiation (IR) induced DNA damage, cancer cells can sustain/activate pro-survival signaling pathways, leading to apoptotic resistance and induction of cell cycle checkpoint/DNA repair. Previous studies show that Rac1 GTPase is overexpressed/hyperactivated in breast cancer cells and is associated with poor prognosis. Studies from our laboratory reveal that Rac1 activity is necessary for G2/M checkpoint activation and cell survival in response to IR exposure of breast and pancreatic cancer cells. In this study, we investigated the effect of Rac1 on the survival of breast cancer cells treated with hyper-fractionated radiation (HFR), which is used clinically for cancer treatment. Results in this report indicate that Rac1 protein expression is increased in the breast cancer cells that survived HFR compared with parental cells. Furthermore, this increase of Rac1 is associated with enhanced activities of extracellular signal-regulated kinases 1 and 2 (ERK1/2) and nuclear factor-κB (NF-κB) signaling pathways and increased levels of anti-apoptotic protein Bcl-xL and Mcl-1, which are downstream targets of ERK1/2 and NF-κB signaling pathways. Using Rac1-specific inhibitor and dominant-negative mutant N17Rac1, here we demonstrate that Rac1 inhibition decreases the phosphorylation of ERK1/2 and inhibitory κBα (IκBα), as well as the levels of Bcl-xL and Mcl-1 protein in the HFR-selected breast cancer cells. Moreover, inhibition of Rac1 using either small molecule inhibitor or dominant-negative N17Rac1 abrogates clonogenic survival of HFR-selected breast cancer cells and decreases the level of intact poly(ADP-ribose) polymerase, which is indicative of apoptosis induction. Collectively, results in this report suggest that Rac1 signaling is essential for the survival of breast cancer cells subjected to HFR and implicate Rac1 in radioresistance of breast cancer cells. These studies also provide the basis to explore Rac1 as a therapeutic target for radioresistant breast cancer cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
DeSantis C, Siegel R, Jemal A . Cancer treatment and survivorship: facts and figures 2014-2015. Am Cancer Soc 2014. 3–6.
Skvortsova I, Debbage P, Kumar V, Skvortsov S . Radiation resistance: cancer stem cells (CSCs) and their enigmatic pro-survival signaling. Semin Cancer Biol 2015; 35: 39–44.
Valerie K, Yacoub A, Hagan MP, Curiel DT, Fisher PB, Grant S et al. Radiation-induced cell signaling: inside-out and outside-in. Mol Cancer Ther 2007; 6: 789–801.
Hein AL, Ouellette MM, Yan Y . Radiation-induced signaling pathways that promote cancer cell survival (review). Int J Oncol 2014; 45: 1813–1819.
Sancar A, Lindsey-Boltz LA, Unsal-Kacmaz K, Linn S . Molecular mechanisms of mammalian DNA repair and the DNA damage checkpoints. Annu Rev Biochem 2004; 73: 39–85.
Wang W, Nag SA, Zhang R . Targeting the NFkappaB signaling pathways for breast cancer prevention and therapy. Curr Med Chem 2015; 22: 264–289.
Chen BPC, Li M, Asaithamby A . New insights into the roles of ATM and DNA-PKcs in the cellular response to oxidative stress. Cancer Lett 2012; 327: 103–110.
Baldwin AS . Regulation of cell death and autophagy by IKK and NF-κB: critical mechanisms in immune function and cancer. Immunol Rev 2012; 246: 327–345.
Bosco E, Mulloy J, Zheng Y . Rac1 GTPase: a “Rac” of all trades. Cell Mol Life Sci 2009; 66: 370.
Wertheimer E, Gutierrez-Uzquiza A, Rosemblit C, Lopez-Haber C, Sosa MS, Kazanietz MG . Rac signaling in breast cancer: a tale of GEFs and GAPs. Cell Signal 2012; 24: 353–362.
Heasman SJ, Ridley AJ . Mammalian Rho GTPases: new insights into their functions from in vivo studies. Nat Rev Mol Cell Biol 2008; 9: 690.
Brown JH, Del Re DP, Sussman MA . The Rac and Rho hall of fame: a decade of hypertrophic signaling hits. Circ Res 2006; 98: 730–742.
Eblen ST, Slack JK, Weber MJ, Catling AD . Rac-PAK signaling stimulates extracellular signal-regulated kinase (ERK) activation by regulating formation of MEK1-ERK complexes. Mol Cell Biol 2002; 22: 6023–6033.
King AJ, Sun H, Diaz B, Barnard D, Miao W, Bagrodia S et al. The protein kinase Pak3 positively regulates Raf-1 activity through phosphorylation of serine 338. Nature 1998; 396: 180–183.
Slack-Davis JK, Eblen ST, Zecevic M, Boerner SA, Tarcsafalvi A, Diaz HB et al. PAK1 phosphorylation of MEK1 regulates fibronectin-stimulated MAPK activation. J Cell Biol 2003; 162: 281–291.
Bokoch GM, Vlahos CJ, Wang Y, Knaus UG, Traynor-Kaplan AE . Rac GTPase interacts specifically with phosphatidylinositol 3-kinase. Biochem J 1996; 315 (Pt 3): 775–779.
Tolias KF, Cantley LC, Carpenter CL . Rho family GTPases bind to phosphoinositide kinases. J Biol Chem 1995; 270: 17656–17659.
Murga C, Zohar M, Teramoto H, Gutkind JS . Rac1 and RhoG promote cell survival by the activation of PI3K and Akt, independently of their ability to stimulate JNK and NF-kappaB. Oncogene 2002; 21: 207–216.
Gonzalez E, Kou R, Michel T . Rac1 modulates sphingosine 1-phosphate-mediated activation of phosphoinositide 3-kinase/Akt signaling pathways in vascular endothelial cells. J Biol Chem 2006; 281: 3210–3216.
Abbott DW, Holt JT . Mitogen-activated protein kinase kinase 2 activation is essential for progression through the G2/M checkpoint arrest in cells exposed to ionizing radiation. J Biol Chem 1999; 274: 2732–2742.
Tang D, Wu D, Hirao A, Lahti JM, Liu L, Mazza B et al. ERK activation mediates cell cycle arrest and apoptosis after DNA damage independently of p53. J Biol Chem 2002; 277: 12710–12717.
Yan Y, Black CP, Cowan KH . Irradiation-induced G2/M checkpoint response requires ERK1/2 activation. Oncogene 2007; 26: 4689–4698.
Toulany M, Lee K-J, Fattah KR, Lin Y-F, Fehrenbacher B, Schaller M et al. Akt promotes post-irradiation survival of human tumor cells through initiation, progression, and termination of DNA-PKcs–dependent DNA double-strand break repair. Mol Cancer Res 2012; 10: 945–957.
Sahlberg SH, Gustafsson AS, Pendekanti PN, Glimelius B, Stenerlow B . The influence of AKT isoforms on radiation sensitivity and DNA repair in colon cancer cell lines. Tumour Biol 2014; 35: 3525–3534.
Shimura T, Kakuda S, Ochiai Y, Kuwahara Y, Takai Y, Fukumoto M . Targeting the AKT/GSK3β/cyclin D1/Cdk4 survival signaling pathway for eradication of tumor radioresistance acquired by fractionated radiotherapy. Int J Radiat Oncol Biol Phys 2011; 80: 540–548.
Yan Y, Greer PM, Cao PT, Kolb RH, Cowan KH . RAC1 GTPase plays an important role in gamma-irradiation induced G2/M checkpoint activation. Breast Cancer Res 2012; 14: R60.
Yan Y, Hein AL, Etekpo A, Burchett KM, Lin C, Enke CA et al. Inhibition of RAC1 GTPase sensitizes pancreatic cancer cells to gamma-irradiation. Oncotarget 2014; 5: 10251–10270.
Magné N, Toillon R-A, Bottero V, Didelot C, Houtte PV, Gérard J-P et al. NF-κB modulation and ionizing radiation: mechanisms and future directions for cancer treatment. Cancer Lett 2006; 231: 158–168.
Bid HK, Roberts RD, Manchanda PK, Houghton PJ . RAC1: an emerging therapeutic option for targeting cancer angiogenesis and metastasis. Mol Cancer Ther 2013; 12: 1925–1934.
Wetterskog D, Shiu KK, Chong I, Meijer T, Mackay A, Lambros M et al. Identification of novel determinants of resistance to lapatinib in ERBB2-amplified cancers. Oncogene 2014; 33: 966–976.
Skvortsov S, Dudás J, Eichberger P, Witsch-Baumgartner M, Loeffler-Ragg J, Pritz C et al. Rac1 as a potential therapeutic target for chemo-radioresistant head and neck squamous cell carcinomas (HNSCC). Br J Cancer 2014; 110: 2677–2687.
Arnold CR, Abdelmoez A, Thurner G, Debbage P, Lukas P, Skvortsov S et al. Rac1 as a multifunctional therapeutic target to prevent and combat cancer metastasis. Oncoscience 2014; 1: 513–521.
Dokmanovic M, Hirsch DS, Shen Y, Wu WJ . Rac1 contributes to trastuzumab resistance of breast cancer cells: Rac1 as a potential therapeutic target for the treatment of trastuzumab-resistant breast cancer. Mol Cancer Ther 2009; 8: 1557–1569.
Sooman L, Ekman S, Andersson C, Kultima H, Isaksson A, Johansson F et al. Synergistic interactions between camptothecin and EGFR or RAC1 inhibitors and between imatinib and Notch signaling or RAC1 inhibitors in glioblastoma cell lines. Cancer Chemother Pharmacol 2013; 72: 329–340.
Zhao Y, Wang Z, Jiang Y, Yang C . Inactivation of Rac1 reduces trastuzumab resistance in PTEN deficient and insulin-like growth factor I receptor overexpressing human breast cancer SKBR3 cells. Cancer Lett 2011; 313: 54–63.
Espinha G, Osaki J, Magalhaes Y, Forti F . Rac1 GTPase-deficient HeLa cells present reduced DNA repair, proliferation, and survival under UV or gamma irradiation. Mol Cell Biochem 2015; 404: 281–297.
Zhao X, Malhotra GK, Lele SM, Lele MS, West WW, Eudy JD et al. Telomerase-immortalized human mammary stem/progenitor cells with ability to self-renew and differentiate. Proc Natl Acad Sci USA 2010; 107: 14146–14151.
Neve RM, Chin K, Fridlyand J, Yeh J, Baehner FL, Fevr T et al. A collection of breast cancer cell lines for the study of functionally distinct cancer subtypes. Cancer Cell 2006; 10: 515–527.
Subik K, Lee JF, Baxter L, Strzepek T, Costello D, Crowley P et al. The expression patterns of ER, PR, HER2, CK5/6, EGFR, Ki-67 and AR by immunohistochemical analysis in breast cancer cell lines. Breast Cancer (Auckl) 2010; 4: 35–41.
Schnelzer A, Prechtel D, Knaus U, Dehne K, Gerhard M, Graeff H et al. Rac1 in human breast cancer: overexpression, mutation analysis, and characterization of a new isoform, Rac1b. Oncogene 2000; 19: 3013–3020.
Moore KA, Sethi R, Doanes AM, Johnson TM, Pracyk JB, Kirby M et al. Rac1 is required for cell proliferation and G2/M progression. Biochem J 1997; 326 (Pt 1): 17–20.
Kroemer G, Galluzzi L, Vandenabeele P, Abrams J, Alnemri ES, Baehrecke EH et al. Classification of cell death: recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ 2009; 16: 3–11.
Lazebnik YA, Kaufmann SH, Desnoyers S, Poirier GG, Earnshaw WC . Cleavage of poly(ADP-ribose) polymerase by a proteinase with properties like ICE. Nature 1994; 371: 346–347.
Leve F, Morgado-Díaz JA . Rho GTPase signaling in the development of colorectal cancer. J Cell Biochem 2012; 113: 2549–2559.
Kunz M . Oncogenes in melanoma: an update. Eur J Cell Biol 2014; 93: 1–10.
Bussink J, van der Kogel AJ, Kaanders JHAM . Activation of the PI3-K/AKT pathway and implications for radioresistance mechanisms in head and neck cancer. Lancet Oncol 2008; 9: 288–296.
Munshi A, Ramesh R . Mitogen-activated protein kinases and their role in radiation response. Genes Cancer 2013; 4: 401–408.
Jiang D, Li D, Cao L, Wang L, Zhu S, Xu T et al. Positive feedback regulation of proliferation in vascular smooth muscle cells stimulated by lipopolysaccharide is mediated through the TLR 4/Rac1/Akt pathway. PLoS One 2014; 9: e92398.
Wang Z, Pedersen E, Basse A, Lefever T, Peyrollier K, Kapoor S et al. Rac1 is crucial for Ras-dependent skin tumor formation by controlling Pak1-Mek-Erk hyperactivation and hyperproliferation in vivo. Oncogene 2010; 29: 3362–3373.
Cuadrado A, Martín-Moldes Z, Ye J, Lastres-Becker I . Transcription factors NRF2 and NF-κB are coordinated effectors of the Rho family, GTP-binding protein RAC1 during inflammation. J Biol Chem 2014; 289: 15244–15258.
Myant Kevin B, Cammareri P, McGhee Ewan J, Ridgway Rachel A, Huels David J, Cordero Julia B et al. ROS production and NF-κB activation triggered by RAC1 facilitate WNT-driven intestinal stem cell proliferation and colorectal cancer initiation. Cell Stem Cell 2013; 12: 761–773.
Opferman JT . Attacking cancer's Achilles heel: antagonism of anti-apoptotic BCL-2 family members. FEBS J 2015, e-pub ahead of print 21 August 2015 doi:10.1111/febs.13472.
Hata AN, Engelman JA, Faber AC . The BCL2 family: key mediators of the apoptotic response to targeted anticancer therapeutics. Cancer Discov 2015; 5: 475–487.
Fiebig AA, Zhu W, Hollerbach C, Leber B, Andrews DW . Bcl-XL is qualitatively different from and ten times more effective than Bcl-2 when expressed in a breast cancer cell line. BMC Cancer 2006; 6: 213.
Gao Y, Dickerson JB, Guo F, Zheng J, Zheng Y . Rational design and characterization of a Rac GTPase-specific small molecule inhibitor. Proc Natl Acad Sci USA 2004; 101: 7618–7623.
Yan Y, Spieker RS, Kim M, Stoeger SM, Cowan KH . BRCA1-mediated G2/M cell cycle arrest requires ERK1/2 kinase activation. Oncogene 2005; 24: 3285–3296.
Shiao YH, Palli D, Caporaso NE, Alvord WG, Amorosi A, Nesi G et al. Genetic and immunohistochemical analyses of p53 independently predict regional metastasis of gastric cancers. Cancer Epidemiol Biomarkers Prev 2000; 9: 631–633.
Charafe-Jauffret E, Tarpin C, Bardou VJ, Bertucci F, Ginestier C, Braud AC et al. Immunophenotypic analysis of inflammatory breast cancers: identification of an 'inflammatory signature'. J Pathol 2004; 202: 265–273.
Pessetto ZY, Yan Y, Bessho T, Natarajan A . Inhibition of BRCT(BRCA1)-phosphoprotein interaction enhances the cytotoxic effect of olaparib in breast cancer cells: a proof of concept study for synthetic lethal therapeutic option. Breast Cancer Res Treat 2012; 134: 511–517.
Ridley AJ, Paterson HF, Johnston CL, Diekmann D, Hall A . The small GTP-binding protein rac regulates growth factor-induced membrane ruffling. Cell 1992; 70: 401–410.
Yan Y, Haas JP, Kim M, Sgagias MK, Cowan KH . BRCA1-induced apoptosis involves inactivation of ERK1/2 activities. J Biol Chem 2002; 277: 33422–33430.
Cai Z, Chattopadhyay N, Liu WJ, Chan C, Pignol J-P, Reilly RM . Optimized digital counting colonies of clonogenic assays using ImageJ software and customized macros: comparison with manual counting. Int J Radiat Biol 2011; 87: 1135–1146.
Acknowledgements
We thank Dr Vimla Band for 76N cells and Dr Toren Finkel for Ad.N17Rac1 and Ad.dl312 adenoviral vectors. This work was supported, in parts, by Pilot Project Funding from 5P30GM106397 to YY and AN, and P50CA127297 to SKB, MMO and YY.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Hein, A., Post, C., Sheinin, Y. et al. RAC1 GTPase promotes the survival of breast cancer cells in response to hyper-fractionated radiation treatment. Oncogene 35, 6319–6329 (2016). https://doi.org/10.1038/onc.2016.163
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.163
This article is cited by
-
Benzimidazole carbamate induces cytotoxicity in breast cancer cells via two distinct cell death mechanisms
Cell Death Discovery (2023)
-
RAC1B function is essential for breast cancer stem cell maintenance and chemoresistance of breast tumor cells
Oncogene (2023)
-
Effect of radiotherapy on the DNA cargo and cellular uptake mechanisms of extracellular vesicles
Strahlentherapie und Onkologie (2023)
-
KLF5-trancripted miR-125b-5p is involved in enhancing the radio-sensitivity of breast cancer cells by targeting BRCA1
Molecular & Cellular Toxicology (2022)
-
RAC1 plays an essential role in estrogen receptor alpha function in breast cancer cells
Oncogene (2021)