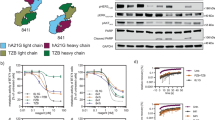
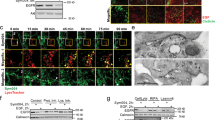
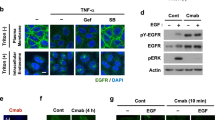
Abstract
Activation of the extensive cross-talk among the receptor tyrosine kinases (RTKs), particularly ErbB family-Met cross-talk, has emerged as a likely source of drug resistance. Notwithstanding brilliant successes were attained while using small-molecule inhibitors or antibody therapeutics against specific RTKs in multiple cancers over recent decades, a high recurrence rate remains unsolved in patients treated with these targeted inhibitors. It is well aligned with multifaceted properties of cancer and cross-talk and convergence of signaling pathways of RTKs. Thereby many therapeutic interventions have been actively developed to overcome inherent or acquired resistance. To date, no bispecific antibody (BsAb) showed complete depletion of dual RTKs from the plasma membrane and efficient dual degradation. In this manuscript, we report the first findings of a target-specific dual internalization and degradation of membrane RTKs induced by designed BsAbs based on the internalizing monoclonal antibodies and the therapeutic values of these BsAbs. Leveraging the anti-Met mAb able to internalize and degrade by a unique mechanism, we generated the BsAbs for Met/epidermal growth factor receptor (EGFR) and Met/HER2 to induce an efficient EGFR or HER2 internalization and degradation in the presence of Met that is frequently overexpressed in the invasive tumors and involved in the resistance against EGFR- or HER2-targeted therapies. We found that Met/EGFR BsAb ME22S induces dissociation of the Met–EGFR complex from Hsp90, followed by significant degradation of Met and EGFR. By employing patient-derived tumor models we demonstrate therapeutic potential of the BsAb-mediated dual degradation in various cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Schlessinger J . Cell signaling by receptor tyrosine kinases. Cell 2000; 103: 211–225.
Zwick E, Bange J, Ullrich A . Receptor tyrosine kinase signalling as a target for cancer intervention strategies. Endocr Relat Cancer 2001; 8: 161–173.
Dhomen NS, Mariadason J, Tebbutt N, Scott AM . Therapeutic targeting of the epidermal growth factor receptor in human cancer. Crit Rev Oncog 2012; 17: 31–50.
Sequist LV, Bell DW, Lynch TJ, Haber DA . Molecular predictors of response to epidermal growth factor receptor antagonists in non-small-cell lung cancer. J Clin Oncol 2007; 25: 587–595.
Doebele RC, Oton AB, Peled N, Camidge DR, Bunn PA Jr . New strategies to overcome limitations of reversible EGFR tyrosine kinase inhibitor therapy in non-small cell lung cancer. Lung Cancer (Amsterdam, Netherlands) 2010; 69: 1–12.
Lin L, Bivona TG . Mechanisms of resistance to epidermal growth factor receptor inhibitors and novel therapeutic strategies to overcome resistance in NSCLC patients. Chemother Res Pract 2012; 2012: 817297.
Guix M, Faber AC, Wang SE, Olivares MG, Song Y, Qu S et al. Acquired resistance to EGFR tyrosine kinase inhibitors in cancer cells is mediated by loss of IGF-binding proteins. J Clin Invest 2008; 118: 2609–2619.
Engelman JA, Zejnullahu K, Mitsudomi T, Song Y, Hyland C, Park JO et al. MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signaling. Science (New York, NY) 2007; 316: 1039–1043.
Wheeler DL, Huang S, Kruser TJ, Nechrebecki MM, Armstrong EA, Benavente S et al. Mechanisms of acquired resistance to cetuximab: role of HER (ErbB) family members. Oncogene 2008; 27: 3944–3956.
Gusenbauer S, Vlaicu P, Ullrich A . HGF induces novel EGFR functions involved in resistance formation to tyrosine kinase inhibitors. Oncogene 2013; 32: 3846–3856.
Liska D, Chen CT, Bachleitner-Hofmann T, Christensen JG, Weiser MR . HGF rescues colorectal cancer cells from EGFR inhibition via MET activation. Clin Cancer Res 2011; 17: 472–482.
Sierra JR, Tsao MS . c-MET as a potential therapeutic target and biomarker in cancer. Ther Adv Med Oncol 2011; 3: S21–S35.
Dulak AM, Gubish CT, Stabile LP, Henry C, Siegfried JM . HGF-independent potentiation of EGFR action by c-Met. Oncogene 2011; 30: 3625–3635.
Sgambato A, Casaluce F, Maione P, Rossi A, Rossi E, Napolitano A et al. The c-Met inhibitors: a new class of drugs in the battle against advanced nonsmall-cell lung cancer. Curr Pharm Des 2012; 18: 6155–6168.
Goffin JR, Zbuk K . Epidermal growth factor receptor: pathway, therapies, and pipeline. Clin Ther 2013; 35: 1282–1303.
Martens T, Schmidt NO, Eckerich C, Fillbrandt R, Merchant M, Schwall R et al. A novel one-armed anti-c-Met antibody inhibits glioblastoma growth in vivo. Clin Cancer Res 2006; 12: 6144–6152.
Oh YM, Song YJ, Lee SB, Jeong Y, Kim B, Kim GW et al. A new anti-c-Met antibody selected by a mechanism-based dual-screening method: therapeutic potential in cancer. Mol Cells 2012; 34: 523–529.
Lee JM, Kim B, Lee SB, Jeong Y, Oh YM, Song YJ et al. Cbl-independent degradation of Met: ways to avoid agonism of bivalent Met-targeting antibody. Oncogene 2014; 33: 34–43.
Kim B, Wang S, Lee JM, Jeong Y, Ahn T, Son DS et al. Synthetic lethal screening reveals FGFR as one of the combinatorial targets to overcome resistance to Met-targeted therapy. Oncogene 2015; 34: 1083–1093.
Oh YM, Lee SB, Choi J, Suh HY, Shim S, Song YJ et al. USP8 modulates ubiquitination of LRIG1 for Met degradation. Sci Rep 2014; 4: 4980.
Jin H, Yang R, Zheng Z, Romero M, Ross J, Bou-Reslan H et al. MetMAb, the one-armed 5D5 anti-c-Met antibody, inhibits orthotopic pancreatic tumor growth and improves survival. Cancer Res 2008; 68: 4360–4368.
Greenall SA, Gherardi E, Liu Z, Donoghue JF, Vitali AA, Li Q et al. Non-agonistic bivalent antibodies that promote c-MET degradation and inhibit tumor growth and others specific for tumor related c-MET. PloS One 2012; 7: e34658.
Doody JF, Wang Y, Patel SN, Joynes C, Lee SP, Gerlak J et al. Inhibitory activity of cetuximab on epidermal growth factor receptor mutations in non small cell lung cancers. Mol Cancer Ther 2007; 6: 2642–2651.
Jo M, Stolz DB, Esplen JE, Dorko K, Michalopoulos GK, Strom SC . Cross-talk between epidermal growth factor receptor and c-Met signal pathways in transformed cells. J Biol Chem 2000; 275: 8806–8811.
Karamouzis MV, Konstantinopoulos PA, Papavassiliou AG . Targeting MET as a strategy to overcome crosstalk-related resistance to EGFR inhibitors. Lancet Oncol 2009; 10: 709–717.
McDermott U, Pusapati RV, Christensen JG, Gray NS, Settleman J . Acquired resistance of non-small cell lung cancer cells to MET kinase inhibition is mediated by a switch to epidermal growth factor receptor dependency. Cancer Res 2010; 70: 1625–1634.
Annamalai B, Liu X, Gopal U, Isaacs JS . Hsp90 is an essential regulator of EphA2 receptor stability and signaling: implications for cancer cell migration and metastasis. Mol Cancer Res 2009; 7: 1021–1032.
Krishnamoorthy GP, Guida T, Alfano L, Avilla E, Santoro M, Carlomagno F et al. Molecular mechanism of 17-allylamino-17-demethoxygeldanamycin (17-AAG)-induced AXL receptor tyrosine kinase degradation. J Biol Chem 2013; 288: 17481–17494.
Mahalingam D, Swords R, Carew JS, Nawrocki ST, Bhalla K, Giles FJ . Targeting HSP90 for cancer therapy. Br J Cancer 2009; 100: 1523–1529.
Miyata Y, Nakamoto H, Neckers L . The therapeutic target Hsp90 and cancer hallmarks. Curr Pharm Des 2013; 19: 347–365.
Lee JY, Kim SY, Park C, Kim NK, Jang J, Park K et al. Patient-derived cell models as preclinical tools for genome-directed targeted therapy. Oncotarget 2015; 6: 25619–25630.
Troiani T, Martinelli E, Napolitano S, Vitagliano D, Ciuffreda LP, Costantino S et al. Increased TGF-alpha as a mechanism of acquired resistance to the anti-EGFR inhibitor cetuximab through EGFR-MET interaction and activation of MET signaling in colon cancer cells. Clin Cancer Res 2013; 19: 6751–6765.
Castoldi R, Ecker V, Wiehle L, Majety M, Busl-Schuller R, Asmussen M et al. A novel bispecific EGFR/Met antibody blocks tumor-promoting phenotypic effects induced by resistance to EGFR inhibition and has potent antitumor activity. Oncogene 2013; 32: 5593–5601.
Ha SY, Lee J, Jang J, Hong JY, Do IG, Park SH et al. HER2-positive gastric cancer with concomitant MET and/or EGFR overexpression: a distinct subset of patients for dual inhibition therapy. Int J Cancer 2015; 136: 1629–1635.
Kim S, Lee J, Hong ME, Do IG, Kang SY, Ha SY et al. High-throughput sequencing and copy number variation detection using formalin fixed embedded tissue in metastatic gastric cancer. PloS One 2014; 9: e111693.
Reungwetwattana T, Dy GK . Targeted therapies in development for non-small cell lung cancer. J Carcinog 2013; 12: 22.
Spigel DR, Ervin TJ, Ramlau RA, Daniel DB, Goldschmidt Jr JH, Blumenschein Jr GR et al. Randomized phase II trial of Onartuzumab in combination with erlotinib in patients with advanced non-small-cell lung cancer. J Clin Oncol 2013; 31: 4105–4114.
Modjtahedi H, Hickish T, Nicolson M, Moore J, Styles J, Eccles S et al. Phase I trial and tumour localisation of the anti-EGFR monoclonal antibody ICR62 in head and neck or lung cancer. Br J Cancer 1996; 73: 228–235.
Weatherill EE, Cain KL, Heywood SP, Compson JE, Heads JT, Adams R et al. Towards a universal disulphide stabilised single chain Fv format: importance of interchain disulphide bond location and vL-vH orientation. Protein Eng Des Sel 2012; 25: 321–329.
Yang HY, Kang KJ, Chung JE, Shim H . Construction of a large synthetic human scFv library with six diversified CDRs and high functional diversity. Mol Cells 2009; 27: 225–235.
Adams GP, Weiner LM . Monoclonal antibody therapy of cancer. Nat Biotechnol 2005; 23: 1147–1157.
Sliwkowski MX, Mellman I . Antibody therapeutics in cancer. Science (New York, NY) 2013; 341: 1192–1198.
Duan Y, Gu TJ, Jiang CL, Yuan RS, Zhang HF, Hou HJ et al. A novel disulfide-stabilized single-chain variable antibody fragment against rabies virus G protein with enhanced in vivo neutralizing potency. Mol Immunol 2012; 51: 188–196.
Reiter Y, Brinkmann U, Kreitman RJ, Jung SH, Lee B, Pastan I . Stabilization of the Fv fragments in recombinant immunotoxins by disulfide bonds engineered into conserved framework regions. Biochemistry 1994; 33: 5451–5459.
Stevenson M . Therapeutic potential of RNA interference. N Engl J Med 2004; 351: 1772–1777.
Davidson BL, McCray PB Jr . Current prospects for RNA interference-based therapies. Nat Rev Genet 2011; 12: 329–340.
Pai SI, Lin YY, Macaes B, Meneshian A, Hung CF, Wu TC . Prospects of RNA interference therapy for cancer. Gene Ther 2006; 13: 464–477.
Straussman R, Morikawa T, Shee K, Barzily-Rokni M, Qian ZR, Du J et al. Tumour micro-environment elicits innate resistance to RAF inhibitors through HGF secretion. Nature 2012; 487: 500–504.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The following authors are employed by the Samsung Advanced Institute of Technology or Samsung Bioepis: JML, SHL, J-WH, SJO, BK, SJ, S-hS, PWL, SBL, M-YC, YJK, JC and K-AK.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Lee, J., Lee, S., Hwang, JW. et al. Novel strategy for a bispecific antibody: induction of dual target internalization and degradation. Oncogene 35, 4437–4446 (2016). https://doi.org/10.1038/onc.2015.514
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.514
This article is cited by
-
CD20/TNFR1 dual-targeting antibody enhances lysosome rupture-mediated cell death in B cell lymphoma
Cancer Immunology, Immunotherapy (2023)
-
Antibody drug conjugates as targeted cancer therapy: past development, present challenges and future opportunities
Archives of Pharmacal Research (2023)
-
Management and Treatment of Non-small Cell Lung Cancer with MET Alteration and Mechanisms of Resistance
Current Treatment Options in Oncology (2022)
-
Imaging EGFR and HER3 through 89Zr-labeled MEHD7945A (Duligotuzumab)
Scientific Reports (2018)
-
A phase I study of LY3164530, a bispecific antibody targeting MET and EGFR, in patients with advanced or metastatic cancer
Cancer Chemotherapy and Pharmacology (2018)