Abstract
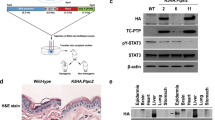
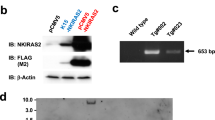
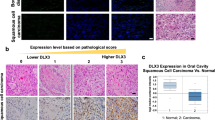
Somatic mutations in the gene encoding the catalytic subunit of protein phosphatase 6 (Ppp6c) have been identified in malignant melanoma and are thought to function as a driver in B-raf- or N-ras-driven tumorigenesis. To assess the role of Ppp6c in carcinogenesis, we generated skin keratinocyte-specific Ppp6c conditional knockout mice and performed two-stage skin carcinogenesis analysis. Ppp6c deficiency induced papilloma formation with 7,12-dimethylbenz (a) anthracene (DMBA) only, and development of those papillomas was significantly accelerated compared with that seen following DMBA/TPA (12-O-tetradecanoylphorbol 13-acetate) treatment of wild-type mice. NF-κB activation either by tumor necrosis factor (TNF)-α or interleukin (IL)-1β was enhanced in Ppp6c-deficient keratinocytes. Overall, we conclude that Ppp6c deficiency predisposes mice to skin carcinogenesis initiated by DMBA. This is the first report showing that such deficiency promotes tumor formation in mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Shi Y . Serine/threonine phosphatases: mechanism through structure. Cell 2009; 139: 468–484.
Brautigan DL . Protein Ser/Thr phosphatases—the ugly ducklings of cell signalling. FEBS J 2013; 280: 324–345.
Stefansson B, Ohama T, Daugherty AE, Brautigan DL . Protein phosphatase 6 regulatory subunits composed of ankyrin repeat domains. Biochemistry 2008; 47: 1442–1451.
Bastians H, Ponstingl H . The novel human protein serine/threonine phosphatase 6 is a functional homologue of budding yeast Sit4p and fission yeast ppe1. J Cell Sci 1996; 109: 2865–2874.
Goshima G, Iwasaki O, Obuse C, Yanagida M . The role of Ppe1/PP6 phosphatase for equal chromosome segregation in fission yeast kinetochore. EMBO J 2003; 22: 2752–2763.
Luke MM, Della Seta F, Di Como CJ, Sugimoto H, Kobayashi R, Arndt KT . The SAP, a new family of proteins, associate and function positively with the SIT4 phosphatase. Mol Cell Biol 1996; 16: 2744–2755.
Sutton A, Immanuel D, Arndt KT . The SIT4 protein phosphatase functions in late G1 for progression into S phase. Mol Cell Biol 1991; 11: 2133–2148.
Afshar K, Werner ME, Tse YC, Glotzer M, Gönczy P . Regulation of cortical contractility and spindle positioning by the protein phosphatase 6 PPH-6 in one-cell stage C. elegans embryos. Development 2010; 137: 237–247.
Kajino T, Ren H, Iemura S, Natsume T, Stefansson B, Brautigan DL et al. Protein phosphatase 6 down-regulates TAK1 kinase activation in the IL-1 signaling pathway. J Biol Chem 2006; 281: 39891–39896.
Stefansson B, Brautigan DL . Protein phosphatase 6 subunit with conserved Sit4-associated protein domain targets IκBɛ. J Biol Chem 2006; 281: 22624–22634.
Mi J, Dziegielewski J, Bolesta E, Brautigan DL, Larner JM . Activation of DNA-PK by ionizing radiation is mediated by protein phosphatase 6. PLoS ONE 2009; 4: e4395.
Hodis E, Watson IR, Kryukov GV, Arold ST, Imielinski M, Theurillat JP et al. A landscape of driver mutations in melanoma. Cell 2012; 150: 251–263.
Krauthammer M, Kong Y, Ha BH, Evans P, Bacchiocchi A, McCusker JP et al. Exome sequencing identifies recurrent somatic RAC1 mutations in melanoma. Nat Genet 2012; 44: 1006–1014.
Abel EL, Angel JM, Kiguchi K, DiGiovanni J . Multi-stage chemical carcinogenesis in mouse skin: fundamentals and applications. Nat Protoc 2009; 4: 1350–1362.
Suganuma M, Fujiki H, Suguri H, Yoshizawa S, Hirota M, Nakayasu M et al. Okadaic acid: an additional non-phorbol-12-tetradecanoate-13-acetate-type tumor promoter. Proc Natl Acad Sci USA 1988; 85: 1768–1771.
Fujiki H, Suganuma M . Tumor promoters - microcystin-LR, nodularin and TNF-α and human cancer development. Anticancer Agents Med Chem 2011; 11: 4–18.
Suganuma M, Okabe S, Marino MW, Sakai A, Sueoka E, Fujiki H . Essential role of tumor necrosis factor alpha (TNF-alpha) in tumor promotion as revealed by TNF-alpha-deficient mice. Cancer Res 1999; 59: 4516–4518.
Komori A, Suganuma M, Okabe S, Zou X, Tius MA, Fujiki H . Canventol inhibits tumor promotion in CD-1 mouse skin through inhibition of tumor necrosis factor alpha release and of protein isoprenylation. Cancer Res 1993; 53: 3462–3464.
Schioppa T, Moore R, Thompson RG, Rosser EC, Kulbe H, Nedospasov S et al. B regulatory cells and the tumor-promoting actions of TNF-α during squamous carcinogenesis. Proc Natl Acad Sci USA 2011; 108: 10662–10667.
Omori E, Morioka S, Matsumoto K, Ninomiya-Tsuji J . TAK1 regulates reactive oxygen species and cell death in keratinocytes, which is essential for skin integrity. J Biol Chem 2008; 283: 26161–26168.
Sundberg JP, Sundberg BA, Beamer WG . Comparison of chemical carcinogen skin tumor induction efficacy in inbred, mutant, and hybrid strains of mice: morphologic variations of induced tumors and absence of a papillomavirus cocarcinogen. Mol Carcinog 1997; 20: 19–32.
Kolář M, Szabo P, Dvořánková B, Lacina L, Gabius HJ, Strnad H et al. Upregulation of IL-6, IL-8 and CXCL-1 production in dermal fibroblasts by normal/malignant epithelial cells in vitro: immunohistochemical and transcriptomic analyses. Biol Cell 2012; 104: 738–751.
Davalos AR, Coppe JP, Campisi J, Desprez PY . Senescent cells as a source of inflammatory factors for tumor progression. Cancer Metastasis Rev 2010; 29: 273–283.
Ancrile BB, O'Hayer KM, Counter CM . Oncogenic ras-induced expression of cytokines: a new target of anti-cancer therapeutics. Mol Interv 2008; 8: 22–27.
Baldin V, Lukas J, Marcote MJ, Pagano M, Draetta G . Cyclin D1 is a nuclear protein required for cell cycle progression in G1. Genes Dev 1993; 7: 812–821.
Valdiglesias V, Giunta S, Fenech M, Neri M, Bonassi S . γH2AX as a marker of DNA double strand breaks and genomic instability in human population studies. Mutat Res 2013; 753: 24–40.
Zhang B, Satoh M, Nishimura N, Suzuki JS, Sone H, Aoki Y et al. Metallothionein deficiency promotes mouse skin carcinogenesis induced by 7,12-dimethylbenz[a]anthracene. Cancer Res 1998; 58: 4044–4046.
Coussens LM, Werb Z . Inflammation and cancer. Nature 2002; 420: 860–867.
Mantovani A, Allavena P, Sica A, Balkwill F . Cancer-related inflammation. Nature 2008; 454: 436–444.
Vandercappellen J, Van Damme J, Struyf S . The role of CXC chemokines and their receptors in cancer. Cancer Lett 2008; 267: 226–244.
Fimmel S, Devermann L, Herrmann A, Zouboulis C . GRO-alpha: a potential marker for cancer and aging silenced by RNA interference. Ann N Y Acad Sci 2007; 1119: 176–189.
Hinz M, Krappmann D, Eichten A, Heder A, Scheidereit C, Strauss M . NF-kappaB function in growth control: regulation of cyclin D1 expression and G0/G1-to-S-phase transition. Mol Cell Biol 1999; 19: 2690–2698.
Karin M . Nuclear factor-kappaB in cancer development and progression. Nature 2006; 441: 431–436.
Tachibana K, Scheuer PJ, Tsukitani Y, Kikuchi H, Engen DV, Clardy J et al. Okadaic acid, a cytotoxic polyether from two marine sponges of the genus Halichondria. J Am Chem Soc 1981; 103: 2469–2471.
Suzuki JS, Nishimura N, Zhang B, Nakatsuru Y, Kobayashi S, Satoh M et al. Metallothionein deficiency enhances skin carcinogenesis induced by 7,12-dimethylbenz[a]anthracene and 12-O-tetradecanoylphorbol-13-acetate in metallothionein-null mice. Carcinogenesis 2003; 24: 1123–1132.
Lichti U, Anders J, Yuspa SH . Isolation and short-term culture of primary keratinocytes, hair follicle populations and dermal cells from newborn mice and keratinocytes from adult mice for in vitro analysis and for grafting to immunodeficient mice. Nat Protoc 2008; 3: 799–810.
Masuda K, Katagiri C, Nomura M, Sato M, Kakumoto K, Akagi T et al. MKP-7, a JNK phosphatase, blocks ERK-dependent gene activation by anchoring phosphorylated ERK in the cytoplasm. Biochem Biophys Res Commun 2010; 393: 201–206.
Acknowledgements
We thank Dr Hirota Fujiki for critical advice and Dr Yoshikazu Nishino for statistical analyses. We thank Nozomi Sasaki, Kuniko Komuro and Miyuki Ueki for technical assistance. We thank Dr Elise Lamar for English editing. This work was supported by JSPS KAKENHI grant numbers 24591928 to Yoichiro Kakugawa, 25861168 to Kayoko Fukamachi and 22590298 to Hiroshi Shima and by a Nara Women’s University Intramural Grant for Project Research to Toshio Watanabe.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Hayashi, K., Momoi, Y., Tanuma, N. et al. Abrogation of protein phosphatase 6 promotes skin carcinogenesis induced by DMBA. Oncogene 34, 4647–4655 (2015). https://doi.org/10.1038/onc.2014.398
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.398
This article is cited by
-
Targeting protein phosphatases for the treatment of inflammation-related diseases: From signaling to therapy
Signal Transduction and Targeted Therapy (2022)
-
PP6 negatively modulates LUBAC-mediated M1-ubiquitination of RIPK1 and c-FLIPL to promote TNFα-mediated cell death
Cell Death & Disease (2022)
-
PpV, acting via the JNK pathway, represses apoptosis during normal development of Drosophila wing
Apoptosis (2018)