Abstract
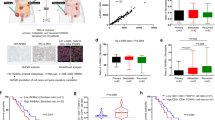
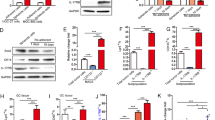
Inflammatory cytokines, components of cancer stem cells (CSCs) niche, could affect the characteristics of CSCs such as self-renewal and metastasis. Interleukin-17 (IL-17) is a new pro-inflammatory cytokine mainly produced by T-helper (Th17) cells and macrophages. The effects of IL-17 on the characteristics of CSCs remain to be explored. Here we first demonstrated a role of IL-17 in promoting the self-renewal of ovarian CD133+ cancer stem-like cells (CSLCs). We detected IL-17-producing cells (CD4+ cells and CD68+ macrophages) in the niche of CD133+CSLCs. Meanwhile, there was IL-17 receptor expression on CD133+CSLCs derived from A2780 cell line and primary ovarian cancer tissues. By recombinant human IL-17 stimulation and IL-17 transfection, the growth and sphere formation capacities of ovarian CD133+CSLCs were significantly enhanced in a dose-dependent manner. Moreover, ovarian CD133+CSLCs transfected with IL-17 showed greater tumorigenesis capacity in nude mice. These data suggest that IL-17 promoted the self-renewal of ovarian CD133+CSLCs. Further investigation through gene profiling revealed that the stimulation function of IL-17 on self-renewal of ovarian CD133+CSLCs might be mediated by the nuclear factor (NF)-κB and p38 mitogen-activated protein kinases (MAPK) signaling pathway. NF-κB and p38 MAPK were activated by IL-17. More importantly, IL-17-promoted self-renewal was inhibited by specific inhibitors of NF-κB and p38 MAPK. Taken together, our data indicate that IL-17 contributed to ovarian cancer malignancy through promoting the self-renewal of CD133+CSLCs and that IL-17 and its signaling pathway might serve as therapeutic targets for the treatment of ovarian cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Zhang S, Balch C, Chan MW, Lai HC, Matei D, Schilder JM et al. Identification and characterization of ovarian cancer-initiating cells from primary human tumors. Cancer Res 2008; 68: 4311–4320.
Hu L, McArthur C, Jaffe RB . Ovarian cancer stem-like side-population cells are tumourigenic and chemoresistant. Br J Cancer 2010; 102: 1276–1283.
Silva IA, Bai S, McLean K, Yang K, Griffith K, Thomas D et al. Aldehyde dehydrogenase in combination with CD133 defines angiogenic ovarian cancer stem cells that portend poor patient survival. Cancer Res 2011; 71: 3991–4001.
Curley MD, Therrien VA, Cummings CL, Sergent PA, Koulouris CR, Friel AM et al. CD133 expression defines a tumor initiating cell population in primary human ovarian cancer. Stem Cells 2009; 27: 2875–2883.
Baba T, Convery PA, Matsumura N, Whitaker RS, Kondoh E, Perry T et al. Epigenetic regulation of CD133 and tumorigenicity of CD133+ ovarian cancer cells. Oncogene 2009; 28: 209–218.
O’Brien CA, Kreso A, Jamieson CH . Cancer stem cells and self-renewal. Clin Cancer Res 2010; 16: 3113–3120.
Hanahan D, Weinberg RA . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–674.
Pasquier J, Rafii A . Role of the microenvironment in ovarian cancer stem cell maintenance. BioMed Res Int 2013; 2013: 630782.
Mantovani A, Allavena P, Sica A, Balkwill F . Cancer-related inflammation. Nature 2008; 454: 436–444.
Yi L, Xiao H, Xu M, Ye X, Hu J, Li F et al. Glioma-initiating cells: a predominant role in microglia/macrophages tropism to glioma. J Neuroimmunol 2011; 232: 75–82.
Sheridan C, Kishimoto H, Fuchs RK, Mehrotra S, Bhat-Nakshatri P, Turner CH et al. CD44+/CD24- breast cancer cells exhibit enhanced invasive properties: an early step necessary for metastasis. Breast Cancer Res 2006; 8: R59.
Todaro M, Alea MP, Di Stefano AB, Cammareri P, Vermeulen L, Iovino F et al. Colon cancer stem cells dictate tumor growth and resist cell death by production of interleukin-4. Cell Stem Cell 2007; 1: 389–402.
Iliopoulos D, Hirsch HA, Struhl K . An epigenetic switch involving NF-kappaB, Lin28, Let-7 MicroRNA, and IL6 links inflammation to cell transformation. Cell 2009; 139: 693–706.
Ginestier C, Liu S, Diebel ME, Korkaya H, Luo M, Brown M et al. CXCR1 blockade selectively targets human breast cancer stem cells in vitro and in xenografts. J Clin Invest 2010; 120: 485–497.
Azzi S, Bruno S, Giron-Michel J, Clay D, Devocelle A, Croce M et al. Differentiation therapy: targeting human renal cancer stem cells with interleukin 15. J Natl Cancer Inst 2011; 103: 1884–1898.
Rouvier E, Luciani MF, Mattei MG, Denizot F, Golstein P . CTLA-8, cloned from an activated T cell, bearing AU-rich messenger RNA instability sequences, and homologous to a herpesvirus saimiri gene. J Immunol 1993; 150: 5445–5456.
Zhu X, Mulcahy LA, Mohammed RA, Lee AH, Franks HA, Kilpatrick L et al. IL-17 expression by breast-cancer-associated macrophages: IL-17 promotes invasiveness of breast cancer cell lines. Breast Cancer Res 2008; 10: R95.
Le Gouvello S, Bastuji-Garin S, Aloulou N, Mansour H, Chaumette MT, Berrehar F et al. High prevalence of Foxp3 and IL17 in MMR-proficient colorectal carcinomas. Gut 2008; 57: 772–779.
Sfanos KS, Bruno TC, Maris CH, Xu L, Thoburn CJ, DeMarzo AM et al. Phenotypic analysis of prostate-infiltrating lymphocytes reveals TH17 and Treg skewing. Clin Cancer Res 2008; 14: 3254–3261.
Steiner GE, Newman ME, Paikl D, Stix U, Memaran-Dagda N, Lee C et al. Expression and function of pro-inflammatory interleukin IL-17 and IL-17 receptor in normal, benign hyperplastic, and malignant prostate. Prostate 2003; 56: 171–182.
Miyahara Y, Odunsi K, Chen W, Peng G, Matsuzaki J, Wang RF . Generation and regulation of human CD4+ IL-17-producing T cells in ovarian cancer. Proc Natl Acad Sci USA 2008; 105: 15505–15510.
Kato T, Furumoto H, Ogura T, Onishi Y, Irahara M, Yamano S et al. Expression of IL-17 mRNA in ovarian cancer. Biochem Biophys Res Commun 2001; 282: 735–738.
Murugaiyan G, Saha B . Protumor vs antitumor functions of IL-17. J Immunol 2009; 183: 4169–4175.
Shalom-Barak T, Quach J, Lotz M . Interleukin-17-induced gene expression in articular chondrocytes is associated with activation of mitogen-activated protein kinases and NF-kappaB. J Biol Chem 1998; 273: 27467–27473.
Subramaniam SV, Cooper RS, Adunyah SE . Evidence for the involvement of JAK/STAT pathway in the signaling mechanism of interleukin-17. Biochem Biophys Res Commun 1999; 262: 14–19.
Subramaniam SV, Pearson LL, Adunyah SE . Interleukin-17 induces rapid tyrosine phosphorylation and activation of raf-1 kinase in human monocytic progenitor cell line U937. Biochem Biophys Res Commun 1999; 259: 172–177.
Chang SH, Park H, Dong C . Act1 adaptor protein is an immediate and essential signaling component of interleukin-17 receptor. J Biol Chem 2006; 281: 35603–35607.
Shostak K, Chariot A . NF-kappaB, stem cells and breast cancer: the links get stronger. Breast Cancer Res 2011; 13: 214.
Armstrong L, Hughes O, Yung S, Hyslop L, Stewart R, Wappler I et al. The role of PI3K/AKT, MAPK/ERK and NFkappabeta signalling in the maintenance of human embryonic stem cell pluripotency and viability highlighted by transcriptional profiling and functional analysis. Hum Mol Genet 2006; 15: 1894–1913.
Long H, Xie R, Xiang T, Zhao Z, Lin S, Liang Z et al. Autocrine CCL5 signaling promotes invasion and migration of CD133+ ovarian cancer stem-like cells via NF-kappaB-mediated MMP-9 upregulation. Stem Cells 2012; 30: 2309–2319.
Kusumbe AP, Mali AM, Bapat SA . CD133-expressing stem cells associated with ovarian metastases establish an endothelial hierarchy and contribute to tumor vasculature. Stem Cells 2009; 27: 498–508.
Numasaki M, Watanabe M, Suzuki T, Takahashi H, Nakamura A, McAllister F et al. IL-17 enhances the net angiogenic activity and in vivo growth of human non-small cell lung cancer in SCID mice through promoting CXCR-2-dependent angiogenesis. J Immunol 2005; 175: 6177–6189.
Wang R, Chadalavada K, Wilshire J, Kowalik U, Hovinga KE, Geber A et al. Glioblastoma stem-like cells give rise to tumour endothelium. Nature 2010; 468: 829–833.
Filatova A, Acker T, Garvalov BK . The cancer stem cell niche(s): the crosstalk between glioma stem cells and their microenvironment. Biochim Biophys Acta 2013; 1830: 2496–2508.
Hamilton TC, Foster BJ, Grotzinger KR, McKoy W, Young RC, Ozols RF . Development of drug-sensitive and -resistant human ovarian cancer cell lines. Proc Am Assoc Cancer Res 1983; 24: 313.
Ciucci A, De Stefano I, Vellone VG, Lisi L, Bottoni C, Scambia G et al. Expression of the glioma-associated oncogene homolog 1 (gli1) in advanced serous ovarian cancer is associated with unfavorable overall survival. PLoS One 2013; 8: e60145.
Domcke S, Sinha R, Levine DA, Sander C, Schultz N . Evaluating cell lines as tumour models by comparison of genomic profiles. Nat Commun 2013; 4: 2126.
Jovanovic DV, Di Battista JA, Martel-Pelletier J, Jolicoeur FC, He Y, Zhang M et al. IL-17 stimulates the production and expression of proinflammatory cytokines, IL-beta and TNF-alpha, by human macrophages. J Immunol 1998; 160: 3513–3521.
Antonysamy MA, Fanslow WC, Fu F, Li W, Qian S, Troutt AB et al. Evidence for a role of IL-17 in organ allograft rejection: IL-17 promotes the functional differentiation of dendritic cell progenitors. J Immunol 1999; 162: 577–584.
Lai D, Wang F, Chen Y, Wang C, Liu S, Lu B et al. Human ovarian cancer stem-like cells can be efficiently killed by gammadelta T lymphocytes. Cancer Immunol Immunother 2012; 61: 979–989.
Lan C, Huang X, Lin S, Huang H, Cai Q, Lu J et al. High density of IL-17-producing cells is associated with improved prognosis for advanced epithelial ovarian cancer. Cell Tissue Res 2013; 352: 351–359.
Takahashi H, Numasaki M, Lotze MT, Sasaki H . Interleukin-17 enhances bFGF-, HGF- and VEGF-induced growth of vascular endothelial cells. Immunol Lett 2005; 98: 189–193.
Numasaki M, Lotze MT, Sasaki H . Interleukin-17 augments tumor necrosis factor-alpha-induced elaboration of proangiogenic factors from fibroblasts. Immunol Lett 2004; 93: 39–43.
Chen X, Xie Q, Cheng X, Diao X, Cheng Y, Liu J et al. Role of interleukin-17 in lymphangiogenesis in non-small-cell lung cancer: enhanced production of vascular endothelial growth factor C in non-small-cell lung carcinoma cells. Cancer Sci 2010; 101: 2384–2390.
Du JW, Xu KY, Fang LY, Qi XL . Interleukin-17, produced by lymphocytes, promotes tumor growth and angiogenesis in a mouse model of breast cancer. Mol Med Rep 2012; 6: 1099–1102.
Kang Z, Wang C, Zepp J, Wu L, Sun K, Zhao J et al. Act1 mediates IL-17-induced EAE pathogenesis selectively in NG2(+) glial cells. Nat Neurosci 2013; 16: 1401–1408.
Villalva C, Martin-Lanneree S, Cortes U, Dkhissi F, Wager M, Le Corf A et al. STAT3 is essential for the maintenance of neurosphere-initiating tumor cells in patients with glioblastomas: a potential for targeted therapy? Int J Cancer 2011; 128: 826–838.
Naka K, Hoshii T, Muraguchi T, Tadokoro Y, Ooshio T, Kondo Y et al. TGF-beta-FOXO signalling maintains leukaemia-initiating cells in chronic myeloid leukaemia. Nature 2010; 463: 676–680.
Laine J, Kunstle G, Obata T, Sha M, Noguchi M . The protooncogene TCL1 is an Akt kinase coactivator. Mol Cell 2000; 6: 395–407.
Bleau AM, Hambardzumyan D, Ozawa T, Fomchenko EI, Huse JT, Brennan CW et al. PTEN/PI3K/Akt pathway regulates the side population phenotype and ABCG2 activity in glioma tumor stem-like cells. Cell Stem Cell 2009; 4: 226–235.
Acknowledgements
This work was supported by National Nature Science Foundation of China (grant nos. 81070018, 81071772 and 81222031), by the outstanding Youth Scientist Foundation of Chongqing (no. CSTC, 2008BA5035) and by National Key Basic Research Program of China (973 program, grant nos. 2010CB529404 and 2012CB526603).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Xiang, T., Long, H., He, L. et al. Interleukin-17 produced by tumor microenvironment promotes self-renewal of CD133+ cancer stem-like cells in ovarian cancer. Oncogene 34, 165–176 (2015). https://doi.org/10.1038/onc.2013.537
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.537
Keywords
This article is cited by
-
Functional Roles of CD133: More than Stemness Associated Factor Regulated by the Microenvironment
Stem Cell Reviews and Reports (2024)
-
Targeting ovarian cancer stem cells: a new way out
Stem Cell Research & Therapy (2023)
-
IL-17A-mediated mitochondrial dysfunction induces pyroptosis in colorectal cancer cells and promotes CD8 + T-cell tumour infiltration
Journal of Translational Medicine (2023)
-
The IL-17 family in diseases: from bench to bedside
Signal Transduction and Targeted Therapy (2023)
-
IL-17A promotes Helicobacter pylori-induced gastric carcinogenesis via interactions with IL-17RC
Gastric Cancer (2023)