Abstract
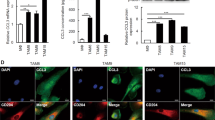
Tumor-associated macrophage (TAM)-related chronic inflammation and interleukin-6 (IL-6) contribute to the progression of nasopharyngeal carcinoma (NPC). In this study, we characterized TAMs and IL-6 expression in 212 biopsied NPC and 119 non-tumor nasopharyngeal epithelium (NPE) tissues by tissue array. In comparison with that in the NPE tissues, more TAM infiltrates and a higher density of IL-6 expression were detected in NPC tissues, which were associated with the poor survival of NPC patients. In contrast, little or no LPLUNC1, a regulator of inflammation, expression was detected in NPC tissues, and the levels of LPLUNC1 expression in the NPC were associated negatively with the numbers of TAMs and the levels of IL-6 expression, but positively with the survival of NPC patients. Induction of LPLUNC1 overexpression in NPC cells mitigated lipopolysaccharide (LPS)-induced IL-6, IL-8, tumor necrosis factor-α and IL-1β expression or treatment of THP-1 macrophages with LPLUNC1 inhibited spontaneous and LPS-induced IL-6 expression in vitro. IL-6-promoted NPC cell proliferation in a dose- and time-dependent manner, accompanied by increasing cyclin D1 and Bcl-2 expression and the Stat3 activation, but inhibiting Bax and p21 expression. Induction of LPLUNC1 overexpression inhibited NPC cell proliferation, induced NPC cell arrest, promoted NPC cell apoptosis even after IL-6 stimulation and inhibited the growth of implanted NPC tumors in vivo, which were associated with decreasing cyclin D1 and Bcl-2 expression and the Janus kinase 2 (JAK2)/Stat3 activation, but enhancing Bax and p21 expression. These results suggest that LPLUNC1 can inhibit inflammation and NPC growth by downregulating the Stat3 pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Mantovani A, Allavena P, Sica A, Balkwill F . Cancer-related inflammation. Nature 2008; 454: 436–444.
Lu H, Ouyang W, Huang C . Inflammation, a key event in cancer development. Mol Cancer Res 2006; 4: 221–233.
Candido J, Hagemann T . Cancer-related inflammation. J Clin Immunol 2012; 33 (Suppl 1): 79–84.
Balkwill FR, Mantovani A . Cancer-related inflammation: common themes and therapeutic opportunities. Semin Cancer Biol 2012; 22: 33–40.
Hussain SP, Harris CC . Inflammation and cancer: an ancient link with novel potentials. Int J Cancer 2007; 121: 2373–2380.
de Visser KE, Coussens LM . The inflammatory tumor microenvironment and its impact on cancer development. Contrib Microbiol 2006; 13: 118–137.
Mantovani A . Cancer: inflaming metastasis. Nature 2009; 457: 36–37.
Yang Y, Zhou H, Li W, Zhou M, Zeng Z, Xiong W et al. Lipopolysaccharide (LPS) regulates TLR4 signal transduction in nasopharynx epithelial cell line 5-8F via NFkappaB and MAPKs signaling pathways. Mol Immunol 2007; 44: 984–992.
Liao Q, Guo X, Li X, Chen P, Liang F, Tang H et al. Analysis of the contribution of nasopharyngeal epithelial cancer cells to the induction of a local inflammatory response. J Cancer Res Clin Oncol 2011; 138: 57–64.
Shih J, Yuan A, Chen JJW, Yang P . Tumor-associated macrophage: its role in cancer invasion and metastasis. J Cancer Mol 2006; 2: 6.
Cocco C, Airoldi I . Tumor-associated macrophages: from cancer supporters to tumoricidal effectors. Immunotherapy 2012; 4: 667–668.
Mantovani A, Schioppa T, Porta C, Allavena P, Sica A . Role of tumor-associated macrophages in tumor progression and invasion. Cancer Metastasis Rev 2006; 25: 315–322.
Hao NB, Lu MH, Fan YH, Cao YL, Zhang ZR, Yang SM . Macrophages in tumor microenvironments and the progression of tumors. Clin Dev Immunol 2012, 2012: 948098.
Steidl C, Lee T, Shah SP, Farinha P, Han G, Nayar T et al. Tumor-associated macrophages and survival in classic Hodgkin's lymphoma. N Engl J Med 2010; 362: 875–885.
Takayama H, Nishimura K, Tsujimura A, Nakai Y, Nakayama M, Aozasa K et al. Increased infiltration of tumor associated macrophages is associated with poor prognosis of bladder carcinoma in situ after intravesical bacillus Calmette-Guerin instillation. J Urol 2009; 181: 1894–1900.
Solinas G, Germano G, Mantovani A, Allavena P . Tumor-associated macrophages (TAM) as major players of the cancer-related inflammation. J Leukoc Biol 2009; 86: 1065–1073.
Heinrich PC, Behrmann I, Haan S, Hermanns HM, Muller-Newen G, Schaper F . Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem J 2003; 374: 1–20.
Sullivan NJ, Sasser AK, Axel AE, Vesuna F, Raman V, Ramirez N et al. Interleukin-6 induces an epithelial-mesenchymal transition phenotype in human breast cancer cells. Oncogene 2009; 28: 2940–2947.
Tan EL, Selvaratnam G, Kananathan R, Sam CK . Quantification of Epstein-Barr virus DNA load, interleukin-6, interleukin-10, transforming growth factor-beta1 and stem cell factor in plasma of patients with nasopharyngeal carcinoma. BMC Cancer 2006; 6: 227.
Chow KC, Chiou SH, Ho SP, Tsai MH, Chen CL, Wang LS et al. The elevated serum interleukin-6 correlates with the increased serum butyrate level in patients with nasopharyngeal carcinoma. Oncol Rep 2003; 10: 813–819.
Bingle CD, Wilson K, Lunn H, Barnes FA, High AS, Wallace WA et al. Human LPLUNC1 is a secreted product of goblet cells and minor glands of the respiratory and upper aerodigestive tracts. Histochem Cell Biol 2010; 133: 505–515.
Bingle CD, Craven CJ . PLUNC: a novel family of candidate host defence proteins expressed in the upper airways and nasopharynx. Hum Mol Genet 2002; 11: 937–943.
Bingle L, Bingle CD . Distribution of human PLUNC/BPI fold-containing (BPIF) proteins. Biochem Soc Trans 2011; 39: 1023–1027.
Shin OS, Uddin T, Citorik R, Wang JP, Della Pelle P, Kradin RL et al. LPLUNC1 modulates innate immune responses to Vibrio cholerae. J Infect Dis 2011; 204: 1349–1357.
Vargas PA, Speight PM, Bingle CD, Barrett AW, Bingle L . Expression of PLUNC family members in benign and malignant salivary gland tumours. Oral Dis 2008; 14: 613–619.
Zhang B, Nie X, Xiao B, Xiang J, Shen S, Gong J et al. Identification of tissue-specific genes in nasopharyngeal epithelial tissue and differentially expressed genes in nasopharyngeal carcinoma by suppression subtractive hybridization and cDNA microarray. Genes Chromosomes Cancer 2003; 38: 80–90.
Baay M, Brouwer A, Pauwels P, Peeters M, Lardon F . Tumor cells and tumor-associated macrophages: secreted proteins as potential targets for therapy. Clin Dev Immunol 2011, 2011: 565187.
Sgambato A, Cittadini A . Inflammation and cancer: a multifaceted link. Eur Rev Med Pharmacol Sci 2011; 14: 263–268.
Rodriguez-Vita J, Lawrence T . The resolution of inflammation and cancer. Cytokine Growth Factor Rev 2011; 21: 61–65.
Motilva V . Inflammation and cancer: new targets and novel therapeutic approach. Curr Pharm Des 2012; 18: 3829–3830.
Fukuda K, Kobayashi A, Watabe K . The role of tumor-associated macrophage in tumor progression. Front Biosci (Schol Ed) 2012; 4: 787–798.
Sica A, Allavena P, Mantovani A . Cancer related inflammation: the macrophage connection. Cancer Lett 2008; 267: 204–215.
Balkwill F, Charles KA, Mantovani A . Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer Cell 2005; 7: 211–217.
Zhang BC, Gao J, Wang J, Rao ZG, Wang BC, Gao JF . Tumor-associated macrophages infiltration is associated with peritumoral lymphangiogenesis and poor prognosis in lung adenocarcinoma. Med Oncol 2010; 28: 1447–1452.
Allavena P, Sica A, Solinas G, Porta C, Mantovani A . The inflammatory micro-environment in tumor progression: the role of tumor-associated macrophages. Crit Rev Oncol Hematol 2008; 66: 1–9.
Wang R, Zhang J, Chen S, Lu M, Luo X, Yao S et al. Tumor-associated macrophages provide a suitable microenvironment for non-small lung cancer invasion and progression. Lung Cancer 2011; 74: 188–196.
Bingle CD, Bingle L . Characterisation of the human plunc gene, a gene product with an upper airways and nasopharyngeal restricted expression pattern. Biochim Biophys Acta 2000; 1493: 363–367.
Di YP, Harper R, Zhao Y, Pahlavan N, Finkbeiner W, Wu R . Molecular cloning and characterization of spurt, a human novel gene that is retinoic acid-inducible and encodes a secretory protein specific in upper respiratory tracts. J Biol Chem 2003; 278: 1165–1173.
Musa M, Wilson K, Sun L, Mulay A, Bingle L, Marriott HM et al. Differential localisation of BPIFA1 (SPLUNC1) and BPIFB1 (LPLUNC1) in the nasal and oral cavities of mice. Cell Tissue Res 2012; 350: 455–464.
Canny G, Levy O . Bactericidal/permeability-increasing protein (BPI) and BPI homologs at mucosal sites. Trends Immunol 2008; 29: 541–547.
Ishihara K, Hirano T . IL-6 in autoimmune disease and chronic inflammatory proliferative disease. Cytokine Growth Factor Rev 2002; 13: 357–368.
Hong DS, Angelo LS, Kurzrock R . Interleukin-6 and its receptor in cancer: implications for Translational Therapeutics. Cancer 2007; 110: 1911–1928.
Strassmann G, Fong M, Kenney JS, Jacob CO . Evidence for the involvement of interleukin 6 in experimental cancer cachexia. J Clin Invest 1992; 89: 1681–1684.
Becker C, Fantini MC, Wirtz S, Nikolaev A, Lehr HA, Galle PR et al. IL-6 signaling promotes tumor growth in colorectal cancer. Cell Cycle 2005; 4: 217–220.
Hsu HS, Lin JH, Hsu TW, Su K, Wang CW, Yang KY et al. Mesenchymal stem cells enhance lung cancer initiation through activation of IL-6/JAK2/STAT3 pathway. Lung Cancer 2012; 75: 167–177.
Yu H, Pardoll D, Jove R . STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer 2009; 9: 798–809.
Kishimoto T . Interleukin-6: from basic science to medicine–40 years in immunology. Annu Rev Immunol 2005; 23: 1–21.
Chen H, Lee JM, Zong Y, Borowitz M, Ng MH, Ambinder RF et al. Linkage between STAT regulation and Epstein-Barr virus gene expression in tumors. J Virol 2001; 75: 2929–2937.
Hsiao JR, Jin YT, Tsai ST, Shiau AL, Wu CL, Su WC . Constitutive activation of STAT3 and STAT5 is present in the majority of nasopharyngeal carcinoma and correlates with better prognosis. Br J Cancer 2003; 89: 344–349.
Lin CM, Shyu KG, Wang BW, Chang H, Chen YH, Chiu JH . Chrysin suppresses IL-6-induced angiogenesis via down-regulation of JAK1/STAT3 and VEGF: an in vitro and in ovo approach. J Agric Food Chem 2010; 58: 7082–7087.
Kim SR, Bae MK, Kim JY, Wee HJ, Yoo MA, Bae SK . Aspirin induces apoptosis through the blockade of IL-6-STAT3 signaling pathway in human glioblastoma A172 cells. Biochem Biophys Res Commun 2009; 387: 342–347.
Ataie-Kachoie P, Pourgholami MH, Morris DL . Inhibition of the IL-6 signaling pathway: A strategy to combat chronic inflammatory diseases and cancer. Cytokine Growth Factor Rev 2012; 24: 163–173.
Fan SQ, Ma J, Zhou J, Xiong W, Xiao BY, Zhang WL et al. Differential expression of Epstein-Barr virus-encoded RNA and several tumor-related genes in various types of nasopharyngeal epithelial lesions and nasopharyngeal carcinoma using tissue microarray analysis. Hum Pathol 2006; 37: 593–605.
Park EK, Jung HS, Yang HI, Yoo MC, Kim C, Kim KS . Optimized THP-1 differentiation is required for the detection of responses to weak stimuli. Inflamm Res 2007; 56: 45–50.
Zhang X, Xiao W, Wang L, Tian Z, Zhang J . Deactivation of signal transducer and activator of transcription 3 reverses chemotherapeutics resistance of leukemia cells via down-regulating P-gp. PLoS One 2011; 6: e20965.
Acknowledgements
This study was supported by grants from the National High Technology Research and Development Program of China (2012AA02A206), the National Natural Science Foundation of China (91229122, 81071644, 81172189, 81101509, 81171930, 81272254 and 81272298), the 111 project (111-2-12), the Program for New Century Excellent Talents in University (NCET-11-0520) and the Hunan Province Natural Sciences Foundation of China (10JJ7003).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Liao, Q., Zeng, Z., Guo, X. et al. LPLUNC1 suppresses IL-6-induced nasopharyngeal carcinoma cell proliferation via inhibiting the Stat3 activation. Oncogene 33, 2098–2109 (2014). https://doi.org/10.1038/onc.2013.161
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.161
Keywords
This article is cited by
-
BPIFB1, Serving as a Downstream Effector of EBV-miR-BART4, Blocks Immune Escape of Nasopharyngeal Carcinoma via Inhibiting PD-L1 Expression
Biochemical Genetics (2024)
-
USP7 inhibits the progression of nasopharyngeal carcinoma via promoting SPLUNC1-mediated M1 macrophage polarization through TRIM24
Cell Death & Disease (2023)
-
ZIC2 induces pro-tumor macrophage polarization in nasopharyngeal carcinoma by activating the JUNB/MCSF axis
Cell Death & Disease (2023)
-
BPIFB1 inhibits vasculogenic mimicry via downregulation of GLUT1-mediated H3K27 acetylation in nasopharyngeal carcinoma
Oncogene (2022)
-
Reduced O-GlcNAcylation of SNAP-23 promotes cisplatin resistance by inducing exosome secretion in ovarian cancer
Cell Death Discovery (2021)