Abstract
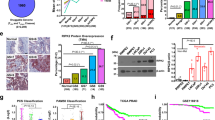
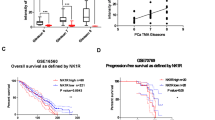
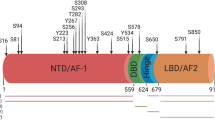
Deregulation of the receptor tyrosine kinase Axl has been implicated in the progression of several human cancers. However, the role of Axl in prostate cancer remains poorly understood, and the therapeutic efficacy of Axl targeting remains untested. In this report we identified Axl as a new therapeutic target for prostate cancer. Axl is consistently overexpressed in prostate cancer cell lines and human prostate tumors. Interestingly, the blockage of Axl gene expression strongly inhibits proliferation, migration, invasion and tumor growth. Furthermore, inhibition of Axl expression by small interfering RNA regulates a transcriptional program of genes involved in cell survival, strikingly all connected to the nuclear factor-κB pathway. Additionally, blockage of Axl expression leads to inhibition of Akt, IKKα and IκBα phosphorylation, increasing IκBα expression and stability. Furthermore, induction of Akt phosphorylation by insulin-like growth factor 1 in Axl knockdown cells restores Akt activity and proliferation. Taken together, our results establish an unambiguous role for Axl in prostate cancer tumorigenesis with implications for prostate cancer treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Haas GP, Delongchamps N, Brawley OW, Wang CY, de la Roza G . The worldwide epidemiology of prostate cancer: perspectives from autopsy studies. Can J Urol 2008; 15: 3866–3871.
O’Bryan JP, Frye RA, Cogswell PC, Neubauer A, Kitch B, Prokop C et al. Axl, a transforming gene isolated from primary human myeloid leukemia cells, encodes a novel receptor tyrosine kinase. Mol Cell Biol 1991; 11: 5016–5031.
Janssen JW, Schulz AS, Steenvoorden AC, Schmidberger M, Strehl S, Ambros PF et al. A novel putative tyrosine kinase receptor with oncogenic potential. Oncogene 1991; 6: 2113–2120.
Ye X, Li Y, Stawicki S, Couto S, Eastham-Anderson J, Kallop D et al. An anti-Axl monoclonal antibody attenuates xenograft tumor growth and enhances the effect of multiple anticancer therapies. Oncogene 2010; 29: 5254–5264.
Holland SJ, Pan A, Franci C, Hu Y, Chang B, Li W et al. R428, a selective small molecule inhibitor of Axl kinase, blocks tumor spread and prolongs survival in models of metastatic breast cancer. Cancer Res 2010; 70: 1544–1554.
Zhang YX, Knyazev PG, Cheburkin YV, Sharma K, Knyazev YP, Orfi L et al. AXL is a potential target for therapeutic intervention in breast cancer progression. Cancer Res 2008; 68: 1905–1915.
Rankin EB, Fuh KC, Taylor TE, Krieg AJ, Musser M, Yuan J et al. AXL is an essential factor and therapeutic target for metastatic ovarian cancer. Cancer Res 2010; 70: 7570–7579.
Koorstra JB, Karikari CA, Feldmann G, Bisht S, Rojas PL, Offerhaus GJ et al. The Axl receptor tyrosine kinase confers an adverse prognostic influence in pancreatic cancer and represents a new therapeutic target. Cancer Biol Ther 2009; 8: 618–626.
Gjerdrum C, Tiron C, Hoiby T, Stefansson I, Haugen H, Sandal T et al. Axl is an essential epithelial-to-mesenchymal transition-induced regulator of breast cancer metastasis and patient survival. Proc Natl Acad Sci USA 2010; 107: 1124–1129.
Varnum BC, Young C, Elliott G, Garcia A, Bartley TD, Fridell YW et al. Axl receptor tyrosine kinase stimulated by the vitamin K-dependent protein encoded by growth-arrest-specific gene 6. Nature 1995; 373: 623–626.
Manfioletti G, Brancolini C, Avanzi G, Schneider C . The protein encoded by a growth arrest-specific gene (gas6) is a new member of the vitamin K-dependent proteins related to protein S, a negative coregulator in the blood coagulation cascade. Mol Cell Biol 1993; 13: 4976–4985.
Healy AM, Schwartz JJ, Zhu X, Herrick BE, Varnum B, Farber HW . Gas 6 promotes Axl-mediated survival in pulmonary endothelial cells. Am J Physiol Lung Cell Mol Physiol 2001; 280: L1273–L1281.
Lee WP, Wen Y, Varnum B, Hung MC . Akt is required for Axl-Gas6 signaling to protect cells from E1A-mediated apoptosis. Oncogene 2002; 21: 329–336.
Bellosta P, Costa M, Lin DA, Basilico C . The receptor tyrosine kinase ARK mediates cell aggregation by homophilic binding. Mol Cell Biol 1995; 15: 614–625.
Gallicchio M, Mitola S, Valdembri D, Fantozzi R, Varnum B, Avanzi GC et al. Inhibition of vascular endothelial growth factor receptor 2-mediated endothelial cell activation by Axl tyrosine kinase receptor. Blood 2005; 105: 1970–1976.
Goruppi S, Chiaruttini C, Ruaro ME, Varnum B, Schneider C . Gas6 induces growth, beta-catenin stabilization, and T-cell factor transcriptional activation in contact-inhibited C57 mammary cells. Mol Cell Biol 2001; 21: 902–915.
Goruppi S, Ruaro E, Varnum B, Schneider C . Requirement of phosphatidylinositol 3-kinase-dependent pathway and Src for Gas6-Axl mitogenic and survival activities in NIH 3T3 fibroblasts. Mol Cell Biol 1997; 17: 4442–4453.
Vajkoczy P, Knyazev P, Kunkel A, Capelle HH, Behrndt S, von Tengg-Kobligk H et al. Dominant-negative inhibition of the Axl receptor tyrosine kinase suppresses brain tumor cell growth and invasion and prolongs survival. Proc Natl Acad Sci USA 2006; 103: 5799–5804.
Li Y, Ye X, Tan C, Hongo JA, Zha J, Liu J et al. Axl as a potential therapeutic target in cancer: role of Axl in tumor growth, metastasis and angiogenesis. Oncogene 2009; 28: 3442–3455.
Shieh YS, Lai CY, Kao YR, Shiah SG, Chu YW, Lee HS et al. Expression of axl in lung adenocarcinoma and correlation with tumor progression. Neoplasia 2005; 7: 1058–1064.
Wimmel A, Glitz D, Kraus A, Roeder J, Schuermann M . Axl receptor tyrosine kinase expression in human lung cancer cell lines correlates with cellular adhesion. Eur J Cancer 2001; 37: 2264–2274.
Harwood FG, Kasibhatla S, Petak I, Vernes R, Green DR, Houghton JA . Regulation of FasL by NF-kappaB and AP-1 in Fas-dependent thymineless death of human colon carcinoma cells. J Biol Chem 2000; 275: 10023–10029.
Mitsiades N, Mitsiades CS, Poulaki V, Chauhan D, Richardson PG, Hideshima T et al. Biologic sequelae of nuclear factor-kappaB blockade in multiple myeloma: therapeutic applications. Blood 2002; 99: 4079–4086.
Li ZW, Chu W, Hu Y, Delhase M, Deerinck T, Ellisman M et al. The IKKbeta subunit of IkappaB kinase (IKK) is essential for nuclear factor kappaB activation and prevention of apoptosis. J Exp Med 1999; 189: 1839–1845.
Zerbini LF, Wang Y, Cho JY, Libermann TA . Constitutive activation of nuclear factor kappaB p50/p65 and Fra-1 and JunD is essential for deregulated interleukin 6 expression in prostate cancer. Cancer Res 2003; 63: 2206–2215.
Grall F, Gu X, Tan L, Cho JY, Inan MS, Pettit AR et al. Responses to the proinflammatory cytokines interleukin-1 and tumor necrosis factor alpha in cells derived from rheumatoid synovium and other joint tissues involve nuclear factor kappaB-mediated induction of the Ets transcription factor ESE-1. Arthritis Rheum 2003; 48: 1249–1260.
Walz A, Schmutz P, Mueller C, Schnyder-Candrian S . Regulation and function of the CXC chemokine ENA-78 in monocytes and its role in disease. J Leukoc Biol 1997; 62: 604–611.
Koch AE . Chemokines and their receptors in rheumatoid arthritis: future targets? Arthritis Rheum 2005; 52: 710–721.
Zerbini LF, Wang Y, Czibere A, Correa RG, Cho JY, Ijiri K et al. NF-kappa B-mediated repression of growth arrest- and DNA-damage-inducible proteins 45alpha and gamma is essential for cancer cell survival. Proc Natl Acad Sci USA 2004; 101: 13618–13623.
Tsai KC, Teng LW, Shao YM, Chen YC, Lee YC, Li M et al. The first pharmacophore model for potent NF-kappaB inhibitors. Bioorg Med Chem Lett 2009; 19: 5665–5669.
Tobe M, Isobe Y, Tomizawa H, Nagasaki T, Takahashi H, Fukazawa T et al. Discovery of quinazolines as a novel structural class of potent inhibitors of NF-kappa B activation. Bioorg Med Chem 2003; 11: 383–391.
Kobori M, Yang Z, Gong D, Heissmeyer V, Zhu H, Jung YK et al. Wedelolactone suppresses LPS-induced caspase-11 expression by directly inhibiting the IKK complex. Cell Death Differ 2004; 11: 123–130.
Sarker D, Reid AH, Yap TA, de Bono JS . Targeting the PI3K/AKT pathway for the treatment of prostate cancer. Clin Cancer Res 2009; 15: 4799–4805.
Romashkova JA, Makarov SS . NF-kappaB is a target of AKT in anti-apoptotic PDGF signalling. Nature 1999; 401: 86–90.
Zheng WH, Kar S, Quirion R . Insulin-like growth factor-1-induced phosphorylation of transcription factor FKHRL1 is mediated by phosphatidylinositol 3-kinase/Akt kinase and role of this pathway in insulin-like growth factor-1-induced survival of cultured hippocampal neurons. Mol Pharmacol 2002; 62: 225–233.
Okamoto M, Lee C, Oyasu R . Interleukin-6 as a paracrine and autocrine growth factor in human prostatic carcinoma cells in vitro. Cancer Res 1997; 57: 141–146.
Hutterer M, Knyazev P, Abate A, Reschke M, Maier H, Stefanova N et al. Axl and growth arrest-specific gene 6 are frequently overexpressed in human gliomas and predict poor prognosis in patients with glioblastoma multiforme. Clin Cancer Res 2008; 14: 130–138.
Sainaghi PP, Castello L, Bergamasco L, Galletti M, Bellosta P, Avanzi GC . Gas6 induces proliferation in prostate carcinoma cell lines expressing the Axl receptor. J Cell Physiol 2005; 204: 36–44.
Shiozawa Y, Pedersen EA, Patel LR, Ziegler AM, Havens AM, Jung Y et al. GAS6/AXL axis regulates prostate cancer invasion, proliferation, and survival in the bone marrow niche. Neoplasia 2010; 12: 116–127.
Jacob AN, Kalapurakal J, Davidson WR, Kandpal G, Dunson N, Prashar Y et al. A receptor tyrosine kinase, UFO/Axl, and other genes isolated by a modified differential display PCR are overexpressed in metastatic prostatic carcinoma cell line DU145. Cancer Detect Prev 1999; 23: 325–332.
Lay JD, Hong CC, Huang JS, Yang YY, Pao CY, Liu CH et al. Sulfasalazine suppresses drug resistance and invasiveness of lung adenocarcinoma cells expressing AXL. Cancer Res 2007; 67: 3878–3887.
Demarchi F, Verardo R, Varnum B, Brancolini C, Schneider C . Gas6 anti-apoptotic signaling requires NF-kappa B activation. J Biol Chem 2001; 276: 31738–31744.
Hasanbasic I, Cuerquis J, Varnum B, Blostein MD . Intracellular signaling pathways involved in Gas6-Axl-mediated survival of endothelial cells. Am J Physiol Heart Circ Physiol 2004; 287: H1207–H1213.
Seaton A, Scullin P, Maxwell PJ, Wilson C, Pettigrew J, Gallagher R et al. Interleukin-8 signaling promotes androgen-independent proliferation of prostate cancer cells via induction of androgen receptor expression and activation. Carcinogenesis 2008; 29: 1148–1156.
Begley LA, Kasina S, Mehra R, Adsule S, Admon AJ, Lonigro RJ et al. CXCL5 promotes prostate cancer progression. Neoplasia 2008; 10: 244–254.
Voronov E, Shouval DS, Krelin Y, Cagnano E, Benharroch D, Iwakura Y et al. IL-1 is required for tumor invasiveness and angiogenesis. Proc Natl Acad Sci USA 2003; 100: 2645–2650.
De Smaele E, Zazzeroni F, Papa S, Nguyen DU, Jin R, Jones J et al. Induction of gadd45beta by NF-kappaB downregulates pro-apoptotic JNK signalling. Nature 2001; 414: 308–313.
Sawabu T, Seno H, Kawashima T, Fukuda A, Uenoyama Y, Kawada M et al. Growth arrest-specific gene 6 and Axl signaling enhances gastric cancer cell survival via Akt pathway. Mol Carcinog 2007; 46: 155–164.
Rawlings JS, Rosler KM, Harrison DA . The JAK/STAT signaling pathway. J Cell Sci 2004; 117 (Part 8): 1281–1283.
Zerbini LF, Czibere A, Wang Y, Correa RG, Otu H, Joseph M et al. A novel pathway involving melanoma differentiation associated gene-7/interleukin-24 mediates nonsteroidal anti-inflammatory drug-induced apoptosis and growth arrest of cancer cells. Cancer Res 2006; 66: 11922–11931.
Acknowledgements
JDP is recipient of the ICGEB post-doctoral fellowship. JFV is recipient of CAPES international fellowship. This study was supported by the Department of Defense grant PC051217 (LFZ), NIH grants 1RO1 CA85467 (TAL) and the Prostate Cancer Foundation (TAL).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Paccez, J., Vasques, G., Correa, R. et al. The receptor tyrosine kinase Axl is an essential regulator of prostate cancer proliferation and tumor growth and represents a new therapeutic target. Oncogene 32, 689–698 (2013). https://doi.org/10.1038/onc.2012.89
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.89
Keywords
This article is cited by
-
The effect of inhibition of receptor tyrosine kinase AXL on DNA damage response in ovarian cancer
Communications Biology (2023)
-
AXL, along with PROS1, is overexpressed in papillary thyroid carcinoma and regulates its biological behaviour
World Journal of Surgical Oncology (2022)
-
Ribonuclease 4 is associated with aggressiveness and progression of prostate cancer
Communications Biology (2022)
-
Epstein–Barr virus miR-BART4-3p regulates cell proliferation, apoptosis, and migration by targeting AXL in gastric carcinoma
Virus Genes (2022)
-
MicroRNAs as the critical regulators of protein kinases in prostate and bladder cancers
Egyptian Journal of Medical Human Genetics (2021)