Abstract
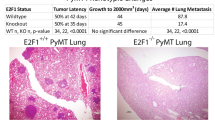
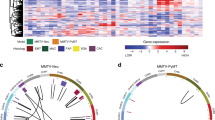
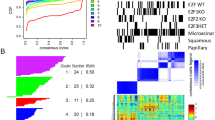
Flotillin microdomains, specialized lipid raft domains in cell membranes, serve as physical platforms for many different molecules important in crucial intracellular signaling pathways. Flotillin-2 (Flot2), together with flotillin-1, is a marker for lipid raft microdomains distinct from caveolar lipid rafts, and has been implicated in the progression of cancer and metastasis formation. Based largely on studies in xenograft models, flotillin-2 has been implicated in the progression of multiple types of human tumors, including breast cancer. In our studies, we identified flotillin-2 as highly amplified in a high-throughput comparative genomic hybridization screen of human breast cancer cell lines and breast tumor samples. Short hairpin RNA-mediated reduction of flotillin-2 protein levels significantly reduced the tumorigenicity and metastatic capability of a human breast cancer cell line in vivo. We generated mice deficient for flotillin-2 and also found a reduction of flotillin-1 protein levels and complete absence of flotillin-specific membrane microdomains in these mice. To examine the role of Flot2 in mammary tumorigenesis and lung metastasis, we used an in vivo molecular genetics approach, crossing a well-characterized transgenic mouse model of breast cancer, the MMTV-PyMT (mouse mammary tumor virus-polyoma middle T antigen) mouse, with gene-targeted Flot2−/− mice. Flotillin-2 deficiency lead to a striking reduction in the number of lung metastasis observed, but had no influence on primary tumor formation in this model. Our results indicate, using a novel in vivo animal model approach, that Flot2 is an important regulator of mammary tumor-derived lung metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Boyle P, Levin B, International Agency for Research on Cancer. World Health Organization World Cancer Report 2008. International Agency for Research on Cancer; Distributed by WHO Press, Lyon Geneva, 2008, 510pp.
Simons K, Toomre D . Lipid rafts and signal transduction. Nat Rev Mol Cell Biol 2000; 1: 31–39.
Le Moyec L, Tatoud R, Eugene M, Gauville C, Primot I, Charlemagne D et al. Cell and membrane lipid analysis by proton magnetic resonance spectroscopy in five breast cancer cell lines. Br J Cancer 1992; 66: 623–628.
Nohara K, Wang F, Spiegel S . Glycosphingolipid composition of MDA-MB-231 and MCF-7 human breast cancer cell lines. Breast Cancer Res Treat 1998; 48: 149–157.
Rothberg KG, Heuser JE, Donzell WC, Ying YS, Glenney JR, Anderson RG . Caveolin, a protein component of caveolae membrane coats. Cell 1992; 68: 673–682.
Yamada E . The fine structure of the renal glomerulus of the mouse. J Biophys Biochem Cytol 1955; 1: 551–566.
Schulte T, Paschke KA, Laessing U, Lottspeich F, Stuermer CA . Reggie-1 and reggie-2, two cell surface proteins expressed by retinal ganglion cells during axon regeneration. Development 1997; 124: 577–587.
Bickel PE, Scherer PE, Schnitzer JE, Oh P, Lisanti MP, Lodish HF . Flotillin and epidermal surface antigen define a new family of caveolae-associated integral membrane proteins. J Biol Chem 1997; 272: 13793–13802.
Neumann-Giesen C, Fernow I, Amaddii M, Tikkanen R . Role of EGF-induced tyrosine phosphorylation of reggie-1/flotillin-2 in cell spreading and signaling to the actin cytoskeleton. J Cell Sci 2007; 120 (Pt 3): 395–406.
Stuermer CA, Langhorst MF, Wiechers MF, Legler DF, Von Hanwehr SH, Guse AH et al. PrPc capping in T cells promotes its association with the lipid raft proteins reggie-1 and reggie-2 and leads to signal transduction. FASEB J 2004; 18: 1731–1733.
de Laurentiis A, Donovan L, Arcaro A . Lipid rafts and caveolae in signaling by growth factor receptors. Open Biochem J 2007; 1: 12–32.
Pike LJ . Lipid rafts: bringing order to chaos. J Lipid Res 2003; 44: 655–667.
Buccione R, Caldieri G, Ayala I . Invadopodia: specialized tumor cell structures for the focal degradation of the extracellular matrix. Cancer Metastasis Rev 2009; 28: 137–149.
Yamaguchi H, Takeo Y, Yoshida S, Kouchi Z, Nakamura Y, Fukami K . Lipid rafts and caveolin-1 are required for invadopodia formation and extracellular matrix degradation by human breast cancer cells. Cancer Res 2009; 69: 8594–8602.
Lin C, Wu Z, Lin X, Yu C, Shi T, Zeng Y et al. Knockdown of FLOT1 impairs cell proliferation and tumorigenicity in breast cancer through upregulation of FOXO3a. Clin Cancer Res 2011; 17: 3089–3099.
Hazarika P, McCarty MF, Prieto VG, George S, Babu D, Koul D et al. Up-regulation of Flotillin-2 is associated with melanoma progression and modulates expression of the thrombin receptor protease activated receptor 1. Cancer Res 2004; 64: 7361–7369.
Rickman DS, Millon R, De Reynies A, Thomas E, Wasylyk C, Muller D et al. Prediction of future metastasis and molecular characterization of head and neck squamous-cell carcinoma based on transcriptome and genome analysis by microarrays. Oncogene 2008; 27: 6607–6622.
Guy CT, Cardiff RD, Muller WJ . Induction of mammary tumors by expression of polyomavirus middle T oncogene: a transgenic mouse model for metastatic disease. Mol Cell Biol 1992; 12: 954–961.
Saeed AI, Bhagabati NK, Braisted JC, Liang W, Sharov V, Howe EA et al. TM4 microarray software suite. Methods Enzymol 2006; 411: 134–193.
Saeed AI, Sharov V, White J, Li J, Liang W, Bhagabati N et al. TM4: a free, open-source system for microarray data management and analysis. BioTechniques 2003; 34: 374–378.
Ludwig A, Otto GP, Riento K, Hams E, Fallon PG, Nichols BJ . Flotillin microdomains interact with the cortical cytoskeleton to control uropod formation and neutrophil recruitment. J Cell Biol 2010; 191: 771–781.
Lin EY, Jones JG, Li P, Zhu L, Whitney KD, Muller WJ et al. Progression to malignancy in the polyoma middle T oncoprotein mouse breast cancer model provides a reliable model for human diseases. Am J Pathol 2003; 163: 2113–2126.
Qiu TH, Chandramouli GV, Hunter KW, Alkharouf NW, Green JE, Liu ET . Global expression profiling identifies signatures of tumor virulence in MMTV-PyMT-transgenic mice: correlation to human disease. Cancer Res 2004; 64: 5973–5981.
Maglione JE, Moghanaki D, Young LJ, Manner CK, Ellies LG, Joseph SO et al. Transgenic polyoma middle-T mice model premalignant mammary disease. Cancer Res 2001; 61: 8298–8305.
de Leeuw RJ, Davies JJ, Rosenwald A, Bebb G, Gascoyne RD, Dyer MJ et al. Comprehensive whole genome array CGH profiling of mantle cell lymphoma model genomes. Hum Mol Genet 2004; 13: 1827–1837.
Ishkanian AS, Malloff CA, Watson SK, DeLeeuw RJ, Chi B, Coe BP et al. A tiling resolution DNA microarray with complete coverage of the human genome. Nat Genet 2004; 36: 299–303.
Watson SK, deLeeuw RJ, Ishkanian AS, Malloff CA, Lam WL . Methods for high throughput validation of amplified fragment pools of BAC DNA for constructing high resolution CGH arrays. BMC Genomics 2004; 5: 6.
Arpaia E, Blaser H, Quintela-Fandino M, Duncan G, Leong HS, Ablack A et al. The interaction between caveolin-1 and Rho-GTPases promotes metastasis by controlling the expression of alpha5-integrin and the activation of Src, Ras and Erk. Oncogene 2012; 31: 884–896.
Acknowledgements
We thank Lily Zhou for expert technical assistance, and Dr Mary Saunders for insightful scientific editing. This work was supported by grant no. 179815 from the Canadian Institutes of Health Research to TWM. This research was funded in part by the Ontario Ministry of Health and Long Term Care. The views expressed do not necessarily reflect those of the OMOHLTC. ES is in part supported by Ontario Research Fund (GL2-01-030) to IJ, and IJ was in part supported by Canada Research Chair Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Berger, T., Ueda, T., Arpaia, E. et al. Flotillin-2 deficiency leads to reduced lung metastases in a mouse breast cancer model. Oncogene 32, 4989–4994 (2013). https://doi.org/10.1038/onc.2012.499
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.499
Keywords
This article is cited by
-
Sphingomyelin synthase 2 promotes the stemness of breast cancer cells via modulating NF-κB signaling pathway
Journal of Cancer Research and Clinical Oncology (2024)
-
Flotillins affect LPS-induced TLR4 signaling by modulating the trafficking and abundance of CD14
Cellular and Molecular Life Sciences (2024)
-
Flotillin membrane domains in cancer
Cancer and Metastasis Reviews (2020)
-
Insufficient radiofrequency ablation promotes the metastasis of residual hepatocellular carcinoma cells via upregulating flotillin proteins
Journal of Cancer Research and Clinical Oncology (2019)
-
A mobile endocytic network connects clathrin-independent receptor endocytosis to recycling and promotes T cell activation
Nature Communications (2018)