Abstract
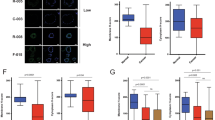
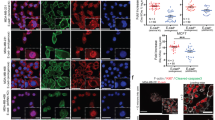
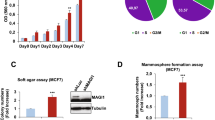
Junctional adhesion molecule-A (JAM-A) is a membranous cell–cell adhesion protein involved in tight-junction formation in epithelial and endothelial cells. Its overexpression in breast tumors has recently been linked with increased risk of metastasis. We sought to identify if JAM-A overexpression was associated with specific subtypes of breast cancer as defined by the expression of human epidermal growth factor receptor-2 (HER2), estrogen receptor (ER) and progesterone receptor. To this end, JAM-A immunohistochemistry was performed in two breast cancer tissue microarrays. In parallel, cross-talk between JAM-A, HER2 and ER was examined in several breast cell lines, using complementary genetic and pharmacological approaches. High JAM-A expression correlated significantly with HER2 protein expression, ER negativity, lower patient age, high-grade breast cancers, and aggressive luminal B, HER2 and basal subtypes of breast cancer. JAM-A and HER2 were co-expressed at high levels in vitro in SKBR3, UACC-812, UACC-893 and MCF7-HER2 cells. Knockdown or functional antagonism of HER2 did not alter JAM-A expression in any cell line tested. Interestingly, however, JAM-A knockdown decreased HER2 and ER-α expression, resulting in reduced levels of phospho-(active) AKT without an effect on the extracellular signal-related kinase phosphorylation. The downstream effects of JAM-A knockdown on HER2 and phospho-AKT were partially reversed upon treatment with the proteasomal inhibitor MG132. We conclude that JAM-A is co-expressed with HER2 and associates with aggressive breast cancer phenotypes. Furthermore, we speculate that JAM-A may regulate HER2 proteasomal degradation and activity, potentially offering a promise as a therapeutic target in HER2-positive breast cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Abbreviations
- HER2:
-
human epidermal growth factor receptor-2
- JAM-A:
-
junctional adhesion molecule-A.
References
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM . GLOBOCAN 2008 v1.2, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 10 [Internet]. International Agency for Research on Cancer, Lyon, France, 2010, (Available from http://globocan.iarc.fr accessed on 4 September 2011).
Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 2001; 98: 10869–10874.
Herschkowitz JI, Simin K, Weigman VJ, Mikaelian I, Usary J, Hu Z et al. Identification of conserved gene expression features between murine mammary carcinoma models and human breast tumors. Genome Biol 2007; 8: R76.
Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA et al. Molecular portraits of human breast tumours. Nature 2000; 406: 747–752.
Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, Conway K et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA 2006; 295: 2492–2502.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL . Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 1987; 235: 177–182.
Baselga J, Swain SM . Novel anticancer targets: revisiting ERBB2 and discovering ERBB3. Nat Rev Cancer 2009; 9: 463–475.
Geyer CE, Forster J, Lindquist D, Chan S, Romieu CG, Pienkowski T et al. Lapatinib plus capecitabine for HER2-positive advanced breast cancer. N Engl J Med 2006; 355: 2733–2743.
Piccart-Gebhart MJ, Procter M, Leyland-Jones B, Goldhirsch A, Untch M, Smith I et al. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med 2005; 353: 1659–1672.
Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE, Davidson NE et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med 2005; 353: 1673–1684.
Osanai M, Murata M, Nishikiori N, Chiba H, Kojima T, Sawada N . Occludin-mediated premature senescence is a fail-safe mechanism against tumorigenesis in breast carcinoma cells. Cancer Sci 2007; 98: 1027–1034.
Hoevel T, Macek R, Swisshelm K, Kubbies M . Reexpression of the TJ protein CLDN1 induces apoptosis in breast tumor spheroids. Int J Cancer 2004; 108: 374–383.
Osanai M, Murata M, Chiba H, Kojima T, Sawada N . Epigenetic silencing of claudin-6 promotes anchorage-independent growth of breast carcinoma cells. Cancer Sci 2007; 98: 1557–1562.
Lanigan F, McKiernan E, Brennan DJ, Hegarty S, Millikan RC, McBryan J et al. Increased claudin-4 expression is associated with poor prognosis and high tumour grade in breast cancer. Int J Cancer 2009; 124: 2088–2097.
Hewitt KJ, Agarwal R, Morin PJ . The claudin gene family: expression in normal and neoplastic tissues. BMC Cancer 2006; 6: 186.
Martin TA, Watkins G, Jiang WG . The Coxsackie-adenovirus receptor has elevated expression in human breast cancer. Clin Exp Med 2005; 5: 122–128.
McSherry EA, McGee SF, Jirstrom K, Doyle EM, Brennan DJ, Landberg G et al. JAM-A expression positively correlates with poor prognosis in breast cancer patients. Int J Cancer 2009; 125: 1343–1351.
Nelson WJ . Adaptation of core mechanisms to generate cell polarity. Nature 2003; 422: 766–774.
Liang TW, DeMarco RA, Mrsny RJ, Gurney A, Gray A, Hooley J et al. Characterization of huJAM: evidence for involvement in cell-cell contact and tight junction regulation. Am J Physiol Cell Physiol 2000; 279: C1733–C1743.
Ebnet K, Suzuki A, Horikoshi Y, Hirose T, Meyer Zu Brickwedde MK, Ohno S et al. The cell polarity protein ASIP/PAR-3 directly associates with junctional adhesion molecule (JAM). EMBO J. 2001; 20: 3738–3748.
Mandell KJ, Babbin BA, Nusrat A, Parkos CA . Junctional adhesion molecule 1 regulates epithelial cell morphology through effects on beta1 integrins and Rap1 activity. J Biol Chem 2005; 280: 11665–11674.
Cooke VG, Naik MU, Naik UP . Fibroblast growth factor-2 failed to induce angiogenesis in junctional adhesion molecule-A-deficient mice. Arterioscler Thromb Vasc Biol 2006; 26: 2005–2011.
Babinska A, Kedees MH, Athar H, Sobocki T, Sobocka MB, Ahmed T et al. Two regions of the human platelet F11-receptor (F11R) are critical for platelet aggregation, potentiation and adhesion. Thromb Haemost 2002; 87: 712–721.
Ostermann G, Weber KS, Zernecke A, Schroder A, Weber C . JAM-1 is a ligand of the beta(2) integrin LFA-1 involved in transendothelial migration of leukocytes. Nat Immunol 2002; 3: 151–158.
Naik MU, Naik TU, Suckow AT, Duncan MK, Naik UP . Attenuation of junctional adhesion molecule-A is a contributing factor for breast cancer cell invasion. Cancer Res 2008; 68: 2194–2203.
Murakami M, Giampietro C, Giannotta M, Corada M, Torselli I, Orsenigo F et al. Abrogation of junctional adhesion molecule-A expression induces cell apoptosis and reduces breast cancer progression. PLoS One 2011; 6: e21242.
Gotte M, Mohr C, Koo CY, Stock C, Vaske AK, Viola M et al. miR-145-dependent targeting of junctional adhesion molecule A and modulation of fascin expression are associated with reduced breast cancer cell motility and invasiveness. Oncogene 2010; 29: 6569–6580.
McSherry EA, Brennan K, Hudson L, Hill AD, Hopkins AM . Breast cancer cell migration is regulated through junctional adhesion molecule-A-mediated activation of Rap1 GTPase. Breast Cancer Res 2011; 13: R31.
Gyorffy B LA, Eklund AC, Denkert C, Budczies J, Li Q, Szallasi Z . An online survival analysis tool to rapidly assess the effect of 22,277 genes on breast cancer prognosis using microarray data of 1809 patients. Breast Cancer Res Treatment 2010; 123: 725–731.
Pegram MD, Finn RS, Arzoo K, Beryt M, Pietras RJ, Slamon DJ . The effect of HER-2/neu overexpression on chemotherapeutic drug sensitivity in human breast and ovarian cancer cells. Oncogene 1997; 15: 537–547.
Scaltriti M, Verma C, Guzman M, Jimenez J, Parra JL, Pedersen K et al. Lapatinib, a HER2 tyrosine kinase inhibitor, induces stabilization and accumulation of HER2 and potentiates trastuzumab-dependent cell cytotoxicity. Oncogene 2009; 28: 803–814.
Chakraborty AK, Liang K, DiGiovanna MP . Co-targeting insulin-like growth factor I receptor and HER2: dramatic effects of HER2 inhibitors on nonoverexpressing breast cancer. Cancer Res 2008; 68: 1538–1545.
Bazzoni G, Tonetti P, Manzi L, Cera MR, Balconi G, Dejana E . Expression of junctional adhesion molecule-A prevents spontaneous and random motility. J Cell Sci 2005; 118 (Pt 3): 623–632.
Severson EA, Lee WY, Capaldo CT, Nusrat A, Parkos CA . Junctional adhesion molecule A interacts with Afadin and PDZ-GEF2 to activate Rap1A, regulate beta1 integrin levels, and enhance cell migration. Mol Biol Cell 2009; 20: 1916–1925.
Jang JY, Jeon YK, Kim CW . Degradation of HER2/neu by ANT2 shRNA suppresses migration and invasiveness of breast cancer cells. BMC Cancer 2010; 10: 391.
Zimmermann S, Moelling K . Phosphorylation and regulation of Raf by Akt (protein kinase B). Science 1999; 286: 1741–1744.
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999; 96: 857–868.
Clark AS, West K, Streicher S, Dennis PA . Constitutive and inducible Akt activity promotes resistance to chemotherapy, trastuzumab, or tamoxifen in breast cancer cells. Mol Cancer Ther 2002; 1: 707–717.
Kim D, Kim S, Koh H, Yoon SO, Chung AS, Cho KS et al. Akt/PKB promotes cancer cell invasion via increased motility and metalloproteinase production. FASEB J. 2001; 15: 1953–1962.
Braniste V, Leveque M, Buisson-Brenac C, Bueno L, Fioramonti J, Houdeau E . Oestradiol decreases colonic permeability through oestrogen receptor beta-mediated up-regulation of occludin and junctional adhesion molecule-A in epithelial cells. J Physiol 2009; 587 (Pt 13): 3317–3328.
Acknowledgements
We are grateful for funding from Science Foundation Ireland (2008/RFP/NSC1427 to AMH), to Dr Tony O'Grady and Trudi Roche for their assistance with obtaining tissue microarray clinical data, and to Dr Marie McIlroy for statistical advice. Herceptin was a kind gift from the St James' Hospital Pharmacy, Dublin, Ireland; LCC1 cells were a kind gift from Professor Robert Clarke, Georgetown University, USA; and MCF7-HER2 cells were a kind gift from Professor Dennis Slamon, University College Los Angeles, USA and Dr Norma O'Donovan, Dublin City University, Ireland.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Brennan, K., McSherry, E., Hudson, L. et al. Junctional adhesion molecule-A is co-expressed with HER2 in breast tumors and acts as a novel regulator of HER2 protein degradation and signaling. Oncogene 32, 2799–2804 (2013). https://doi.org/10.1038/onc.2012.276
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.276
Keywords
This article is cited by
-
Triple negative breast cancer metastasis is hindered by a peptide antagonist of F11R/JAM‑A protein
Cancer Cell International (2023)
-
The F11 Receptor (F11R)/Junctional Adhesion Molecule-A (JAM-A) (F11R/JAM-A) in cancer progression
Molecular and Cellular Biochemistry (2022)
-
Cleavage of the extracellular domain of junctional adhesion molecule-A is associated with resistance to anti-HER2 therapies in breast cancer settings
Breast Cancer Research (2018)
-
The Contribution of Ig-Superfamily and MARVEL D Tight Junction Proteins to Cancer Pathobiology
Current Pathobiology Reports (2016)
-
Functions of novel Junctional Adhesion Molecule-A (JAM-A) inhibitor in breast cancer cells
BMC Proceedings (2015)