Abstract
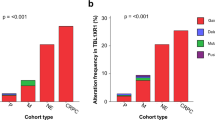
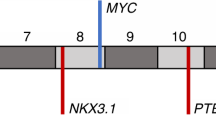
A clearer definition of the molecular determinants that drive the development and progression of prostate cancer (PCa) is urgently needed. Efforts to map recurrent somatic deletions in the tumor genome, especially homozygous deletions (HODs), have provided important positional information in the search for cancer-causing genes. Analyzing HODs in the tumors of 244 patients from two independent cohorts and 22 PCa xenografts using high-resolution single-nucleotide polymorphism arrays, herein we report the identification of CHD1, a chromatin remodeler, as one of the most frequently homozygously deleted genes in PCa, second only to PTEN in this regard. The HODs observed in CHD1, including deletions affecting only internal exons of CHD1, were found to completely extinguish the expression of mRNA of this gene in PCa xenografts. Loss of this chromatin remodeler in clinical specimens is significantly associated with an increased number of additional chromosomal deletions, both hemi- and homozygous, especially on 2q, 5q and 6q. Together with the deletions observed in HEK293 cells stably transfected with CHD1 small hairpin RNA, these data suggest a causal relationship. Downregulation of Chd1 in mouse prostate epithelial cells caused dramatic morphological changes indicative of increased invasiveness, but did not result in transformation. Indicating a new role of CHD1, these findings collectively suggest that distinct CHD1-associated alterations of genomic structure evolve during and are required for the development of PCa.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Anderson K, Lutz C, van Delft FW, Bateman CM, Guo Y, Colman SM et al. (2011). Genetic variegation of clonal architecture and propagating cells in leukaemia. Nature 469: 356–361.
Avvakumov N, Nourani A, Côté J . (2011). Histone chaperones: modulators of chromatin marks. Mol Cell 41: 502–514.
Barclay WW, Axanova LS, Chen W, Romero L, Maund SL, Soker S et al. (2008). Characterization of adult prostatic progenitor/stem cells exhibiting self-renewal and multilineage differentiation. Stem Cells 26: 600–610.
Barclay WW, Cramer SD . (2005). Culture of mouse prostatic epithelial cells from genetically engineered mice. Prostate 63: 291–298.
Berger MF, Lawrence MS, Demichelis F, Drier Y, Cibulskis K, Sivachenko AY et al. (2011). The genomic complexity of primary human prostate cancer. Nature 470: 214–220.
Beroukhim R, Getz G, Nghiemphu L, Barretina J, Hsueh T, Linhart D et al. (2007). Assessing the significance of chromosomal aberrations in cancer: methodology and application to glioma. Proc Natl Acad Sci USA 104: 20007–20012.
Beroukhim R, Mermel CH, Porter D, Wei G, Raychaudhuri S, Donovan J et al. (2010). The landscape of somatic copy-number alteration across human cancers. Nature 463: 899–905.
Bettermann K, Vucur M, Haybaeck J, Koppe C, Janssen J, Heymann F et al. (2010). TAK1 suppresses a NEMO-dependent but NF-kappaB-independent pathway to liver cancer. Cancer Cell 17: 481–496.
Bignell GR, Greenman CD, Davies H, Butler AP, Edkins S, Andrews JM et al. (2010). Signatures of mutation and selection in the cancer genome. Nature 463: 893–898.
Cao P, Deng Z, Wan M, Huang W, Cramer SD, Xu J et al. (2010). MicroRNA-101 negatively regulates Ezh2 and its expression is modulated by androgen receptor and HIF-1alpha/HIF-1beta. Mol Cancer 9: 108.
Carver BS, Tran J, Gopalan A, Chen Z, Shaikh S, Carracedo A et al. (2009). Aberrant ERG expression cooperates with loss of PTEN to promote cancer progression in the prostate. Nat Genet 41: 619–624.
Cengiz B, Gunduz M, Nagatsuka H, Beder L, Gunduz E, Tamamura R et al. (2007). Fine deletion mapping of chromosome 2q21-37 shows three preferentially deleted regions in oral cancer. Oral Oncol 43: 241–247.
Dalgliesh GL, Furge K, Greenman C, Chen L, Bignell G, Butler A et al. (2010). Systematic sequencing of renal carcinoma reveals inactivation of histone modifying genes. Nature 463: 360–363.
Deng Z, Wan M, Cao P, Rao A, Cramer SD, Sui G . (2009). Yin Yang 1 regulates the transcriptional activity of androgen receptor. Oncogene 28: 3746–3757.
Deng Z, Wan M, Sui G . (2007). PIASy-mediated sumoylation of Yin Yang 1 depends on their interaction but not the RING finger. Mol Cell Biol 27: 3780–3792.
Ding L, Getz G, Wheeler DA, Mardis ER, McLellan MD, Cibulskis K et al. (2008). Somatic mutations affect key pathways in lung adenocarcinoma. Nature 455: 1069–1075.
Ding Z, Wu CJ, Chu GC, Xiao Y, Ho D, Zhang J et al. (2011). SMAD4-dependent barrier constrains prostate cancer growth and metastatic progression. Nature 466: 869–873.
Gaspar-Maia A, Alajem A, Polesso F, Sridharan R, Mason MJ, Heidersbach A et al. (2009). Chd1 regulates open chromatin and pluripotency of embryonic stem cells. Nature 460: 863–868.
Jones S, Wang TL, Shih IeM, Mao TL, Nakayama K, Roden R et al. (2010). Frequent mutations of chromatin remodeling gene ARID1A in ovarian clear cell carcinoma. Science 330: 228–231.
Kan Z, Jaiswal BS, Stinson J, Janakiraman V, Bhatt D, Stern HM et al. (2010). Diverse somatic mutation patterns and pathway alterations in human cancers. Nature 466: 869–873.
King JC, Xu J, Wongvipat J, Hieronymus H, Carver BS, Leung DH et al. (2009). Cooperativity of TMPRSS2-ERG with PI3-kinase pathway activation in prostate oncogenesis. Nat Genet 41: 524–526.
Kohno T, Otsuka A, Girard L, Sato M, Iwakawa R, Ogiwara H et al. (2010). A catalog of genes homozygously deleted in human lung cancer and the candidacy of PTPRD as a tumor suppressor gene. Genes Chromosomes Cancer 49: 342–352.
Konev AY, Tribus M, Park SY, Podhraski V, Lim CY, Emelyanov AV et al. (2007). CHD1 motor protein is required for deposition of histone variant H3.3 into chromatin in vivo. Science 317: 1087–1090.
Liu W, Chang BL, Cramer S, Koty PP, Li T, Sun J et al. (2007). Deletion of a small consensus region at 6q15, including the MAP3K7 gene, is significantly associated with high-grade prostate cancers. Clin Cancer Res 13: 5028–5033.
Liu W, Laitinen S, Khan S, Vihinen M, Kowalski J, Yu G et al. (2009). Copy number analysis indicates monoclonal origin of lethal metastatic prostate cancer. Nat Med 15: 559–565.
Lu YJ, Wu CS, Li HP, Liu HP, Lu CY, Leu YW et al. (2010). Aberrant methylation impairs low density lipoprotein receptor-related protein 1B tumor suppressor function in gastric cancer. Genes Chromosomes Cancer 49: 412–424.
McDaniel IE, Lee JM, Berger MS, Hanagami CK, Armstrong JA . (2008). Investigations of CHD1 function in transcription and development of Drosophila melanogaster. Genetics 178: 583–587.
Medina PP, Romero OA, Kohno T, Montuenga LM, Pio R, Yokota J et al. (2008). Frequent BRG1/SMARCA4-inactivating mutations in human lung cancer cell lines. Hum Mutat 29: 617–622.
Nancarrow DJ, Handoko HY, Smithers BM, Gotley DC, Drew PA, Watson DI et al. (2008). Genome-wide copy number analysis in esophageal adenocarcinoma using high-density single-nucleotide polymorphism arrays. Cancer Res 68: 4163–4172.
Rahrmann EP, Collier LS, Knutson TP, Doyal ME, Kuslak SL, Green LE et al. (2009). Identification of PDE4D as a proliferation promoting factor in prostate cancer using a Sleeping Beauty transposon-based somatic mutagenesis screen. Cancer Res 69: 4388–4397.
Robbins CM, Tembe WA, Baker A, Sinari S, Moses TY, Beckstrom-Sternberg S et al. (2011). Copy number and targeted mutational analysis reveals novel somatic events in metastatic prostate tumors. Genome Res 21: 47–55.
Siegel R, Ward E, Brawley O, Jemal A . (2011). Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin 61: 212–236.
Sims 3rd RJ, Millhouse S, Chen CF, Lewis BA, Erdjument-Bromage H, Tempst P et al. (2007). Recognition of trimethylated histone H3 lysine 4 facilitates the recruitment of transcription postinitiation factors and pre-mRNA splicing. Mol Cell 28: 665–676.
Sims 3rd RJ, Reinberg D . (2009). Stem cells: escaping fates with open states. Nature 460: 802–803.
Stephens PJ, Greenman CD, Fu B, Yang F, Bignell GR, Mudie LJ et al. (2011). Massive genomic rearrangement acquired in a single catastrophic event during cancer development. Cell 144: 27–40.
Stokes DG, Perry RP . (1995). DNA-binding and chromatin localization properties of CHD1. Mol Cell Biol 15: 2745–2753.
Sui G, Shi Y . (2005). Gene silencing by a DNA vector-based RNAi technology. Methods Mol Biol 309: 205–218.
Sui G, Soohoo C, Affar el B, Gay F, Shi Y, Forrester WC et al. (2002). A DNA vector-based RNAi technology to suppress gene expression in mammalian cells. Proc Natl Acad Sci USA 99: 5515–5520.
Taylor BS, Schultz N, Hieronymus H, Gopalan A, Xiao Y, Carver BS et al. (2010). Integrative genomic profiling of human prostate cancer. Cancer Cell 18: 11–22.
Weissman B, Knudsen KE . (2009). Hijacking the chromatin remodeling machinery: impact of SWI/SNF perturbations in cancer. Cancer Res 69: 8223–8230 Review.
Yin D, Ogawa S, Kawamata N, Tunici P, Finocchiaro G, Eoli M et al. (2009). High-resolution genomic copy number profiling of glioblastoma multiforme by single nucleotide polymorphism DNA microarray. Mol Cancer Res 7: 665–677.
Acknowledgements
The study is partially supported by the National Institutes of Health Grants CA135008 and CA133066 (to W Liu and WB Isaacs), CA119069 (to J Xu), CA131338 and 133009 (to SL Zheng and WB Isaacs). We thank Tamara Adams for editing the manuscript. The support of William T Gerrard, Mario Duhon, Jennifer and John Chalsty and P Kevin Jaffe (to WBI) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, W., Lindberg, J., Sui, G. et al. Identification of novel CHD1-associated collaborative alterations of genomic structure and functional assessment of CHD1 in prostate cancer. Oncogene 31, 3939–3948 (2012). https://doi.org/10.1038/onc.2011.554
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.554
Keywords
This article is cited by
-
Resistance to the Androgen Receptor Centred Therapies: Biology and Management
SN Comprehensive Clinical Medicine (2021)
-
Chromoanagenesis: cataclysms behind complex chromosomal rearrangements
Molecular Cytogenetics (2019)
-
Molecular Subtypes of Prostate Cancer
Current Oncology Reports (2018)
-
Sequencing of prostate cancers identifies new cancer genes, routes of progression and drug targets
Nature Genetics (2018)
-
Drug discovery in advanced prostate cancer: translating biology into therapy
Nature Reviews Drug Discovery (2016)