Abstract
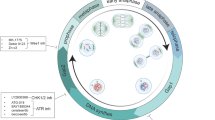
Current theories suggest that mitotic checkpoint proteins are essential for proper cellular response to taxanes, a widely used family of chemotherapeutic compounds. We recently showed that absence or depletion of protein Daxx increases cellular taxol (paclitaxel) resistance—a common trait of patients diagnosed with several malignancies, including breast cancer. Further investigation of Daxx-mediated taxol response revealed that Daxx is important for the proper timing of mitosis progression and cyclin B stability. Daxx interacts with mitotic checkpoint protein RAS-association domain family protein 1 (Rassf1) and partially colocalizes with this protein during mitosis. Rassf1/Daxx depletion or expression of Daxx-binding domain of Rassf1 elevates cyclin B stability and increases taxol resistance in cells and mouse xenograft models. In breast cancer patients, we observed the inverse correlation between Daxx and clinical response to taxane-based chemotherapy. These data suggest that Daxx and Rassf1 define a mitotic stress checkpoint that enables cells to exit mitosis as micronucleated cells (and eventually die) when encountered with specific mitotic stress stimuli, including taxol. Surprisingly, depletion of Daxx or Rassf1 does not change the activity of E3 ubiquitin ligase anaphase promotion complex/C in in vitro settings, suggesting the necessity of mitotic cellular environment for proper activation of this checkpoint. Daxx and Rassf1 may become useful predictive markers for the proper selection of patients for taxane chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Aapro MS . (2001). Neoadjuvant therapy in breast cancer: can we define its role? Oncologist 6 (Suppl 3): 36–39.
Agathanggelou A, Cooper WN, Latif F . (2005). Role of the Ras-association domain family 1 tumor suppressor gene in human cancers. Cancer Res 65: 3497–3508.
Amaar YG, Baylink DJ, Mohan S . (2005). Ras-association domain family 1 protein, RASSF1C, is an IGFBP-5 binding partner and a potential regulator of osteoblast cell proliferation. J Bone Miner Res 20: 1430–1439.
Bonneterre J, Spielman M, Guastalla JP, Marty M, Viens P, Chollet P et al. (1999). Efficacy and safety of docetaxel (Taxotere) in heavily pretreated advanced breast cancer patients: the French compassionate use programme experience. Eur J Cancer 35: 1431–1439.
Brito DA, Rieder CL . (2006). Mitotic checkpoint slippage in humans occurs via cyclin B destruction in the presence of an active checkpoint. Curr Biol 16: 1194–1200.
Carvalho A, Carmena M, Sambade C, Earnshaw WC, Wheatley SP . (2003). Survivin is required for stable checkpoint activation in taxol-treated HeLa cells. J Cell Sci 116: 2987–2998.
Chabalier C, Lamare C, Racca C, Privat M, Valette A, Larminat F . (2006). BRCA1 downregulation leads to premature inactivation of spindle checkpoint and confers paclitaxel resistance. Cell Cycle 5: 1001–1007.
Chang JC, Wooten EC, Tsimelzon A, Hilsenbeck SG, Gutierrez MC, Elledge R et al. (2003). Gene expression profiling for the prediction of therapeutic response to docetaxel in patients with breast cancer. Lancet 362: 362–369.
Chang JC, Wooten EC, Tsimelzon A, Hilsenbeck SG, Gutierrez MC, Tham YL et al. (2005). Patterns of resistance and incomplete response to docetaxel by gene expression profiling in breast cancer patients. J Clin Oncol 23: 1169–1177.
Crown J, O'Leary M, Ooi WS . (2004). Docetaxel and paclitaxel in the treatment of breast cancer: a review of clinical experience. Oncologist 9 (Suppl 2): 24–32.
Dallol A, Agathanggelou A, Fenton SL, Ahmed-Choudhury J, Hesson L, Vos MD et al. (2004). RASSF1A interacts with microtubule-associated proteins and modulates microtubule dynamics. Cancer Res 64: 4112–4116.
De Mulder PH . (1999). The chemotherapy of head and neck cancer. Acta Otorhinolaryngol Belg 53: 247–252.
Escobar-Cabrera E, Lau DK, Giovinazzi S, Ishov AM, McIntosh LP . (2010). Structural characterization of the DAXX N-terminal helical bundle domain and its complex with Rassf1C. Structure 18: 1642–1653.
Estrabaud E, Lassot I, Blot G, Le Rouzic E, Tanchou V, Quemeneur E et al. (2007). RASSF1C, an isoform of the tumor suppressor RASSF1A, promotes the accumulation of beta-catenin by interacting with betaTrCP. Cancer Res 67: 1054–1061.
Hari M, Wang Y, Veeraraghavan S, Cabral F . (2003a). Mutations in alpha- and beta-tubulin that stabilize microtubules and confer resistance to colcemid and vinblastine. Mol Cancer Ther 2: 597–605.
Hari M, Yang H, Zeng C, Canizales M, Cabral F . (2003b). Expression of class III beta-tubulin reduces microtubule assembly and confers resistance to paclitaxel. Cell Motil Cytoskeleton 56: 45–56.
Henderson IC, Berry DA, Demetri GD, Cirrincione CT, Goldstein LJ, Martino S et al. (2003). Improved outcomes from adding sequential Paclitaxel but not from escalating Doxorubicin dose in an adjuvant chemotherapy regimen for patients with node-positive primary breast cancer. J Clin Oncol 21: 976–983.
Ishov AM, Sotnikov AG, Negorev D, Vladimirova OV, Neff N, Kamitani T et al. (1999). PML is critical for ND10 formation and recruits the PML-interacting protein daxx to this nuclear structure when modified by SUMO-1. J Cell Biol 147: 221–234.
Ishov AM, Vladimirova OV, Maul GG . (2004). Heterochromatin and ND10 are cell-cycle regulated and phosphorylation-dependent alternate nuclear sites of the transcription repressor Daxx and SWI/SNF protein ATRX. J Cell Sci 117: 3807–3820.
Iwao-Koizumi K, Matoba R, Ueno N, Kim SJ, Ando A, Miyoshi Y et al. (2005). Prediction of docetaxel response in human breast cancer by gene expression profiling. J Clin Oncol 23: 422–431.
Kitagawa D, Kajiho H, Negishi T, Ura S, Watanabe T, Wada T et al. (2006). Release of RASSF1C from the nucleus by Daxx degradation links DNA damage and SAPK/JNK activation. EMBO J 25: 3286–3297.
Kubota T, Matsuzaki SW, Hoshiya Y, Watanabe M, Kitajima M, Asanuma F et al. (1997). Antitumor activity of paclitaxel against human breast carcinoma xenografts serially transplanted into nude mice. J Surg Oncol 64: 115–121.
Lee EA, Keutmann MK, Dowling ML, Harris E, Chan G, Kao GD . (2004). Inactivation of the mitotic checkpoint as a determinant of the efficacy of microtubule-targeted drugs in killing human cancer cells. Mol Cancer Ther 3: 661–669.
Lens SM, Wolthuis RM, Klompmaker R, Kauw J, Agami R, Brummelkamp T et al. (2003). Survivin is required for a sustained spindle checkpoint arrest in response to lack of tension. EMBO J 22: 2934–2947.
Lindsay CR, Giovinazzi S, Ishov AM . (2009). Daxx is a predominately nuclear protein that does not translocate to the cytoplasm in response to cell stress. Cell Cycle 8: 1544–1551.
Lindsay CR, Morozov VM, Ishov AM . (2008). PML NBs (ND10) and Daxx: from nuclear structure to protein function. Front Biosci 13: 7132–7142.
Lindsay CR, Scholz A, Morozov VM, Ishov AM . (2007). Daxx shortens mitotic arrest caused by paclitaxel. Cell Cycle 6: 1200–1204.
Liu L, Baier K, Dammann R, Pfeifer GP . (2007). The tumor suppressor RASSF1A does not interact with Cdc20, an activator of the anaphase-promoting complex. Cell Cycle 6: 1663–1665.
Liu L, Tommasi S, Lee DH, Dammann R, Pfeifer GP . (2003). Control of microtubule stability by the RASSF1A tumor suppressor. Oncogene 22: 8125–8136.
Mantel C, Guo Y, Lee MR, Han MK, Rhorabough S, Kim KS et al. (2008). Cells enter a unique intermediate 4N stage, not 4N-G1, after aborted mitosis. Cell Cycle 7: 484–492.
Mauriac L, Debled M, MacGrogan G . (2005). When will more useful predictive factors be ready for use? Breast 14: 617–623.
Meraldi P, Draviam VM, Sorger PK . (2004). Timing and checkpoints in the regulation of mitotic progression. Dev Cell 7: 45–60.
Michaelson JS . (2000). The Daxx enigma. Apoptosis 5: 217–220.
Michaelson JS, Bader D, Kuo F, Kozak C, Leder P . (1999a). Loss of Daxx, a promiscuously interacting protein, results in extensive apoptosis in early mouse development. Genes Dev 13: 1918–1923.
Michaelson JS, Bader D, Kuo F, Kozak C, Leder P . (1999b). Loss of daxx, a promiscuously interacting protein, results in extensive apoptosis in early mouse development [In Process Citation]. Genes Dev 13: 1918–1923.
Miyoshi Y, Kim SJ, Akazawa K, Kamigaki S, Ueda S, Yanagisawa T et al. (2004). Down-regulation of intratumoral aromatase messenger RNA levels by docetaxel in human breast cancers. Clin Cancer Res 10: 8163–8169.
Morozov VM, Massoll NA, Vladimirova OV, Maul GG, Ishov AM . (2008). Regulation of c-met expression by transcription repressor Daxx. Oncogene 27: 2177–2186.
Niikura Y, Dixit A, Scott R, Perkins G, Kitagawa K . (2007). BUB1 mediation of caspase-independent mitotic death determines cell fate. J Cell Biol 178: 283–296.
Nilsson J, Yekezare M, Minshull J, Pines J . (2008). The APC/C maintains the spindle assembly checkpoint by targeting Cdc20 for destruction. Nat Cell Biol 10: 1411–1420.
O'Shaughnessy J . (2005). Extending survival with chemotherapy in metastatic breast cancer. Oncologist 10 (Suppl 3): 20–29.
Ravdin P, Erban J, Overmoyer B . (2003). Phase III comparison of docetaxel and paclitaxel in patients with metastatic breast cancer (abstract). Eur J Cancer (Suppl 1): 32.
Rong R, Jiang LY, Sheikh MS, Huang Y . (2007). Mitotic kinase Aurora-A phosphorylates RASSF1A and modulates RASSF1A-mediated microtubule interaction and M-phase cell cycle regulation. Oncogene 26: 7700–7708.
Rong R, Jin W, Zhang J, Sheikh MS, Huang Y . (2004). Tumor suppressor RASSF1A is a microtubule-binding protein that stabilizes microtubules and induces G2/M arrest. Oncogene 23: 8216–8230.
Sablina AA, Budanov AV, Ilyinskaya GV, Agapova LS, Kravchenko JE, Chumakov PM . (2005). The antioxidant function of the p53 tumor suppressor. Nat Med 11: 1306–1313.
Saffert RT, Kalejta RF . (2008). Promyelocytic leukemia-nuclear body proteins: herpesvirus enemies, accomplices, or both? Fut Virol 3: 265–277.
Salomoni P, Khelifi AF . (2006). Daxx: death or survival protein? Trends Cell Biol 16: 97–104.
Scolnick DM, Halazonetis TD . (2000). Chfr defines a mitotic stress checkpoint that delays entry into metaphase. Nature 406: 430–435.
Song MS, Song SJ, Ayad NG, Chang JS, Lee JH, Hong HK et al. (2004). The tumour suppressor RASSF1A regulates mitosis by inhibiting the APC–Cdc20 complex. Nat Cell Biol 6: 129–137.
Song MS, Song SJ, Kim SY, Oh HJ, Lim DS . (2008). The tumour suppressor RASSF1A promotes MDM2 self-ubiquitination by disrupting the MDM2–DAXX–HAUSP complex. EMBO J 27: 1863–1874.
Sudo T, Nitta M, Saya H, Ueno NT . (2004). Dependence of paclitaxel sensitivity on a functional spindle assembly checkpoint. Cancer Res 64: 2502–2508.
Summers MK, Pan B, Mukhyala K, Jackson PK . (2008). The unique N terminus of the UbcH10 E2 enzyme controls the threshold for APC activation and enhances checkpoint regulation of the APC. Mol Cell 31: 544–556.
Swanton C, Marani M, Pardo O, Warne PH, Kelly G, Sahai E et al. (2007). Regulators of mitotic arrest and ceramide metabolism are determinants of sensitivity to paclitaxel and other chemotherapeutic drugs. Cancer Cell 11: 498–512.
Wang Y, Cabral F . (2005). Paclitaxel resistance in cells with reduced beta-tubulin. Biochim Biophys Acta 1744: 245–255.
Wassmann K, Benezra R . (2001). Mitotic checkpoints: from yeast to cancer. Curr Opin Genet Dev 11: 83–90.
Wysong DR, Chakravarty A, Hoar K, Ecsedy JA . (2009). The inhibition of Aurora A abrogates the mitotic delay induced by microtubule perturbing agents. Cell Cycle 8: 876–888.
Xia G, Luo X, Habu T, Rizo J, Matsumoto T, Yu H . (2004). Conformation-specific binding of p31(comet) antagonizes the function of Mad2 in the spindle checkpoint. EMBO J 23: 3133–3143.
Acknowledgements
We thank Dr Gerd Pfeifer, Beckman Research Institute, for the generous gift of anti-Rassf1 antibodies and Dr Frank Rauscher, The Wistar Institute, for HP1 antibodies. This work was supported by NIH/NCI R01 CA127378-01A1 for CRL, SG, VMM and AMI, by the Canadian Cancer Society (017308) for EE and LPM. NMR spectroscopy support was provided by the Canadian Institutes for Health Research (CIHR), the Canadian Foundation for Innovation (CFI), the British Columbia Knowledge Development Fund (BCKDF), the UBC Blusson Fund and the Michael Smith Foundation for Health Research (MSFHR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Giovinazzi, S., Lindsay, C., Morozov, V. et al. Regulation of mitosis and taxane response by Daxx and Rassf1. Oncogene 31, 13–26 (2012). https://doi.org/10.1038/onc.2011.211
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.211
Keywords
This article is cited by
-
Impact of DAXX and ATRX expression on telomere length and prognosis of breast cancer patients
Journal of the Egyptian National Cancer Institute (2020)
-
The MRVI1-AS1/ATF3 signaling loop sensitizes nasopharyngeal cancer cells to paclitaxel by regulating the Hippo–TAZ pathway
Oncogene (2019)
-
Age-dependent differential expression of death-associated protein 6 (Daxx) in various peripheral tissues and different brain regions of C57BL/6 male mice
Biogerontology (2016)
-
USP7 and Daxx regulate mitosis progression and taxane sensitivity by affecting stability of Aurora-A kinase
Cell Death & Differentiation (2013)
-
Germinal center kinases in immune regulation
Cellular & Molecular Immunology (2012)