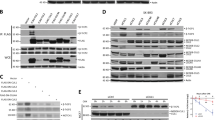
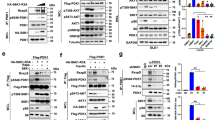
Abstract
The mammalian target of rapamycin (mTOR) is a highly conserved serine–threonine kinase activated in response to growth factors and nutrients. Because of frequent dysregulation of the mTOR signaling pathway in diverse human cancers, this kinase is a key therapeutic target. Redd1 is a negative regulator of mTOR, mediating dissociation of 14-3-3 from tuberous sclerosis complex (TSC)2, which allows formation of a TSC–TSC2 complex. In the present study, we identify TXNIP that inhibits mTOR activity by binding to and stabilizing Redd1 protein. Redd1 and TXNIP expression was induced by a synthetic glucose analog, 2-deoxyglucose (2-DG). Moreover, Redd1 expression in response to 2-DG was regulated by activating transcription factor 4 (ATF4). Overexpression of TXNIP was associated with reduced mTOR activity mediated by an increase in Redd1 level, whereas knockdown of TXNIP using small interfering RNA resulted in recovery of mTOR activity via downregulation of Redd1 during treatment with 2-DG. Interestingly, Redd1 was additionally stabilized via interactions with N-terminal-truncated TXNIP, leading to suppression of mTOR activity. Our results collectively demonstrate that TXNIP stabilizes Redd1 protein induced by ATF4 in response to 2-DG, resulting in potentiation of mTOR suppression. To the best of our knowledge, this is the first study to identify TXNIP as a novel member of the mTOR upstream that acts as a negative regulator in response to stress signals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Brugarolas J, Lei K, Hurley RL, Manning BD, Reiling JH, Hafen E et al. (2004). Regulation of mTOR function in response to hypoxia by REDD1 and the TSC1/TSC2 tumor suppressor complex. Genes Dev 18: 2893–2904.
Chung JW, Jeon JH, Yoon SR, Choi I . (2006). Vitamin D3 upregulated protein 1 (VDUP1) is a regulator for redox signaling and stress-mediated diseases. J Dermatol 33: 662–669.
Dennis PB, Jaeschke A, Saitoh M, Fowler B, Kozma SC, Thomas G . (2001). Mammalian TOR: a homeostatic ATP sensor. Science 294: 1102–1105.
DeYoung MP, Horak P, Sofer A, Sgroi D, Ellisen LW . (2008). Hypoxia regulates TSC1/2-mTOR signaling and tumor suppression through REDD1-mediated 14-3-3 shuttling. Genes Dev 22: 239–251.
Ellisen LW, Ramsayer KD, Johannessen CM, Yang A, Beppu H, Minda K et al. (2002). REDD1, a developmentally regulated transcriptional target of p63 and p53, links p63 to regulation of reactive oxygen species. Mol Cell 10: 995–1005.
Fidler IJ, Radinsky R . (1996). Search for genes that suppress cancer metastasis. J Natl Cancer Inst 88: 1700–1703.
Goldberg SF, Miele ME, Hatta N, Takata M, Paquette-Straub C, Freedman LP et al. (2003). Melanoma metastasis suppression by chromosome 6: evidence for a pathway regulated by CRSP3 and TXNIP. Cancer Res 63: 432–440.
Han SH, Jeon JH, Ju HR, Jung U, Kim KY, Yoo HS et al. (2003). VDUP1 upregulated by TGF-beta1 and 1,25-dihydorxyvitamin D3 inhibits tumor cell growth by blocking cell-cycle progression. Oncogene 22: 4035–4046.
Hardie DG, Hawley SA . (2001). AMP-activated protein kinase: the energy charge hypothesis revisited. Bioessays 23: 1112–1119.
Hawley SA, Davison M, Woods A, Davies SP, Beri RK, Carling D et al. (1996). Characterization of the AMP-activated protein kinase from rat liver and identification of threonine 172 as the major site at which it phosphorylates AMP-activated protein kinase. J Biol Chem 271: 27879–27887.
Horak P, Crawford AR, Vadysirisack DD, Nash ZM, DeYoung MP, Sgroi D et al. (2010). Negative feedback control of HIF-1 through REDD1-regulated ROS suppresses tumorigenesis. Proc Natl Acad Sci USA 107: 4675–4680.
Inoki K, Zhu T, Guan KL . (2003). TSC2 mediates cellular energy response to control cell growth and survival. Cell 115: 577–590.
Jin HO, Seo SK, Woo SH, Choe TB, Hong SI, Kim JI et al. (2009a). Nuclear protein 1 induced by ATF4 in response to various stressors acts as a positive regulator on the transcriptional activation of ATF4. IUBMB Life 61: 1153–1158.
Jin HO, Seo SK, Woo SH, Kim ES, Lee HC, Yoo DH et al. (2009b). Activating transcription factor 4 and CCAAT/enhancer-binding protein-beta negatively regulate the mammalian target of rapamycin via Redd1 expression in response to oxidative and endoplasmic reticulum stress. Free Radic Biol Med 46: 1158–1167.
Junn E, Han SH, Im JY, Yang Y, Cho EW, Um HD et al. (2000). Vitamin D3 up-regulated protein 1 mediates oxidative stress via suppressing the thioredoxin function. J Immunol 164: 6287–6295.
Kang HT, Hwang ES . (2006). 2-Deoxyglucose: an anticancer and antiviral therapeutic, but not any more a low glucose mimetic. Life Sci 78: 1392–1399.
Katiyar S, Liu E, Knutzen CA, Lang ES, Lombardo CR, Sankar S et al. (2009). REDD1, an inhibitor of mTOR signalling, is regulated by the CUL4A-DDB1 ubiquitin ligase. EMBO Rep 10: 866–872.
Kim KY, Shin SM, Kim JK, Paik SG, Yang Y, Choi I . (2004). Heat shock factor regulates VDUP1 gene expression. Biochem Biophys Res Commun 315: 369–375.
Little E, Ramakrishnan M, Roy B, Gazit G, Lee AS . (1994). The glucose-regulated proteins (GRP78 and GRP94): functions, gene regulation, and applications. Crit Rev Eukaryot Gene Expr 4: 1–18.
Nakamura H, Masutani H, Yodoi J . (2006). Extracellular thioredoxin and thioredoxin-binding protein 2 in control of cancer. Semin Cancer Biol 16: 444–451.
Nishinaka Y, Masutani H, Oka S, Matsuo Y, Yamaguchi Y, Nishio K et al. (2004). Importin alpha1 (Rch1) mediates nuclear translocation of thioredoxin-binding protein-2/vitamin D(3)-up-regulated protein 1. J Biol Chem 279: 37559–37565.
Oka S, Liu W, Masutani H, Hirata H, Shinkai Y, Yamada S et al. (2006). Impaired fatty acid utilization in thioredoxin binding-2 (TBP-2)-deficient mice: a unique animal model of Reye syndrome. FASEB J 20: 121–123.
Peterson TR, Laplante M, Thoreen CC, Sancak Y, Kang SA, Kuehl WM et al. (2009). DEPTOR is an mTOR inhibitor frequently overexpressed in multiple myeloma cells and required for their survival. Cell 137: 873–886.
Saxena G, Chen J, Shalev A . (2010). Intracellular shuttling and mitochondrial function of thioredoxin-interacting protein. J Biol Chem 285: 3997–4005.
Schmelzle T, Hall MN . (2000). TOR, a central controller of cell growth. Cell 103: 253–262.
Schneider A, Younis RH, Gutkind JS . (2008). Hypoxia-induced energy stress inhibits the mTOR pathway by activating an AMPK/REDD1 signaling axis in head and neck squamous cell carcinoma. Neoplasia 10: 1295–1302.
Schulze PC, De Keulenaer GW, Yoshioka J, Kassik KA, Lee RT . (2002). Vitamin D3-upregulated protein-1 (VDUP-1) regulates redox-dependent vascular smooth muscle cell proliferation through interaction with thioredoxin. Circ Res 91: 689–695.
Schulze PC, Yoshioka J, Takahashi T, He Z, King GL, Lee RT . (2004). Hyperglycemia promotes oxidative stress through inhibition of thioredoxin function by thioredoxin-interacting protein. J Biol Chem 279: 30369–30374.
Shaw RJ, Lamia KA, Vasquez D, Koo SH, Bardeesy N, Depinho RA et al. (2005). The kinase LKB1 mediates glucose homeostasis in liver and therapeutic effects of metformin. Science 310: 1642–1646.
Sheth SS, Bodnar JS, Ghazalpour A, Thipphavong CK, Tsutsumi S, Tward AD et al. (2006). Hepatocellular carcinoma in Txnip-deficient mice. Oncogene 25: 3528–3536.
Shor B, Gibbons JJ, Abraham RT, Yu K . (2009). Targeting mTOR globally in cancer: thinking beyond rapamycin. Cell Cycle 8: 3831–3837.
Sofer A, Lei K, Johannessen CM, Ellisen LW . (2005). Regulation of mTOR and cell growth in response to energy stress by REDD1. Mol Cell Biol 25: 5834–5845.
Stein SC, Woods A, Jones NA, Davison MD, Carling D . (2000). The regulation of AMP-activated protein kinase by phosphorylation. Biochem J 345: 437–443.
Stoltzman CA, Peterson CW, Breen KT, Muoio DM, Billin AN, Ayer DE . (2008). Glucose sensing by MondoA:Mlx complexes: a role for hexokinases and direct regulation of thioredoxin-interacting protein expression. Proc Natl AcadSci USA 105: 6912–6917.
Sudarsanam S, Johnson DE . (2010). Functional consequences of mTOR inhibition. Curr Opin Drug Discov Devel 13: 31–40.
Towler MC, Hardie DG . (2007). AMP-activated protein kinase in metabolic control and insulin signaling. Circ Res 100: 328–341.
Vander Haar E, Lee SI, Bandhakavi S, Griffin TJ, Kim DH . (2007). Insulin signalling to mTOR mediated by the Akt/PKB substrate PRAS40. Nat Cell Biol 9: 316–323.
Wang H, Kubica N, Ellisen LW, Jefferson LS, Kimball SR . (2006a). Dexamethasone represses signaling through the mammalian target of rapamycin in muscle cells by enhancing expression of REDD1. J Biol Chem 281: 39128–39134.
Wang Z, Rong YP, Malone MH, Davis MC, Zhong F, Distelhorst CW . (2006b). Thioredoxin-interacting protein (txnip) is a glucocorticoid-regulated primary response gene involved in mediating glucocorticoid-induced apoptosis. Oncogene 25: 1903–1913.
Whitney ML, Jefferson LS, Kimball SR . (2009). ATF4 is necessary and sufficient for ER stress-induced upregulation of REDD1 expression. Biochem Biophys Res Commun 379: 451–455.
Acknowledgements
This work was supported by the National Nuclear R&D Program and the Basic Science Research Program and the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2010-0009503) in Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Jin, HO., Seo, SK., Kim, YS. et al. TXNIP potentiates Redd1-induced mTOR suppression through stabilization of Redd1. Oncogene 30, 3792–3801 (2011). https://doi.org/10.1038/onc.2011.102
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.102
Keywords
This article is cited by
-
The stress-responsive protein REDD1 and its pathophysiological functions
Experimental & Molecular Medicine (2023)
-
The m6A demethylase ALKBH5-mediated upregulation of DDIT4-AS1 maintains pancreatic cancer stemness and suppresses chemosensitivity by activating the mTOR pathway
Molecular Cancer (2022)
-
C-terminal truncated HBx initiates hepatocarcinogenesis by downregulating TXNIP and reprogramming glucose metabolism
Oncogene (2021)
-
Inhibition of SIRT1/2 upregulates HSPA5 acetylation and induces pro-survival autophagy via ATF4-DDIT4-mTORC1 axis in human lung cancer cells
Apoptosis (2019)
-
BRCA1 deficiency sensitizes breast cancer cells to bromodomain and extra-terminal domain (BET) inhibition
Oncogene (2018)