Abstract
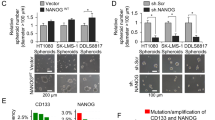
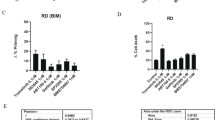
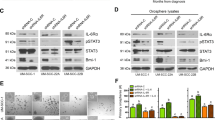
The cellular and molecular mechanisms of tumor progression following chemotherapy are largely unknown. Here, we demonstrate that cisplatin (CDDP) treatment upregulates VEGF and Flt1 expression leading to the survival and expansion of a highly tumorigenic fraction of side-population (SP) cells in osteosarcoma (HOS), neuroblastoma (SK-N-BE2) and rhabdomyosarcoma (RH-4) cell lines. In all three lines, we show that CDDP treatment increases levels of VEGF and Flt1 expression, and induces enhanced clonogenic capacity and increased expression of the ‘stemness’-associated genes Nanog, Bmi-1 and Oct-4 in the SP fraction. In HOS, these changes are associated with the transformation of a non-tumorigenic osteosarcoma SP fraction to a highly tumorigenic phenotype. Inhibition of Flt1 led to complete reduction of tumorigenicity in the HOS SP fraction, and reduction of clonogenic capacity and expression of stemness genes in the SK-N-BE(2) and RH-4 SP fractions. Treatment with U0126, a specific inhibitor of MAPK/ERK1,2 completely downregulates CDDP-induced VEGF and Flt1 expression and induction/expansion of SP fraction in all three cell lines, indicating that these effects are mediated through MAPK/ERK1,2 signaling. In conclusion, we report a novel mechanism of CDDP-induced tumor progression, whereby the activation of VEGF/Flt1 autocrine signaling leads to the survival and expansion of a highly tumorigenic SP fraction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bates RC, Goldsmith JD, Bachelder RE, Brown C, Shibuya M, Oettgen P et al. (2003). Flt-1-dependent survival characterizes the epithelial–mesenchymal transition of colonic organoids. Curr Biol 13: 1721–1727.
Behrends M, Schulz R, Post H, Alexandrov A, Belosjorow S, Michel MC et al. (2000). Inconsistent relation of MAPK activation to infarct size reduction by ischemic preconditioning in pigs. Am J Physiol Heart Circ Physiol 279: H1111–H1119.
Bellamy WT, Richter L, Sirjani D, Roxas C, Glinsmann-Gibson B, Frutiger Y et al. (2001). Vascular endothelial cell growth factor is an autocrine promoter of abnormal localized immature myeloid precursors and leukemia progenitor formation in myelodysplastic syndromes. Blood 97: 1427–1434.
Biswas S, Guix M, Rinehart C, Dugger TC, Chytil A, Moses HL et al. (2007). Inhibition of TGF-beta with neutralizing antibodies prevents radiation-induced acceleration of metastatic cancer progression. J Clin Invest 117: 1305–1313.
Brozovic A, Osmak M . (2007). Activation of mitogen-activated protein kinases by cisplatin and their role in cisplatin-resistance. Cancer Lett 251: 1–16.
Cara S, Tannock IF . (2001). Retreatment of patients with the same chemotherapy: implications for clinical mechanisms of drug resistance. Ann Oncol 12: 23–27.
Cole S, Tannock IF . (2004). Drug Resistance, 4th edn. McGraw-Hill: Toronto, 390 pp.
Cottler-Fox MH, Lapidot T, Petit I, Kollet O, DiPersio JF, Link D et al. (2003). Stem cell mobilization. Hematology Am Soc Hematol Educ Program, 419–437.
Das B, Tsuchida R, Malkin D, Baruchel S, Yeger H . (2007). Hypoxia enhances tumor stemness by increasing the invasive and tumorigenic side-population fraction. Stem Cells (under review).
Das B, Yeger H, Baruchel H, Freedman M, Koren G, Baruchel S . (2003). In vitro cytoprotective activity of squalene on a bone marrow versus neuroblastoma model of cisplatin-induced toxicity. Implications in cancer chemotherapy. Eur J Cancer 39: 2556–2565.
Das B, Yeger H, Tsuchida R, Torkin R, Gee MF, Thorner PS et al. (2005). A hypoxia-driven vascular endothelial growth factor/Flt1 autocrine loop interacts with hypoxia-inducible factor-1alpha through mitogen-activated protein kinase/extracellular signal-regulated kinase 1/2 pathway in neuroblastoma. Cancer Res 65: 7267–7275.
Dias S, Hattori K, Zhu Z, Heissig B, Choy M, Lane W et al. (2000). Autocrine stimulation of VEGFR-2 activates human leukemic cell growth and migration. J Clin Invest 106: 511–521.
Ferrara N, Gerber HP, LeCouter J . (2003). The biology of VEGF and its receptors. Nat Med 9: 669–676.
Folkman J . (1971). Tumor angiogenesis: therapeutic implications. N Engl J Med 285: 1182–1186.
Fukumura D, Xavier R, Sugiura T, Chen Y, Park EC, Lu N et al. (1998). Tumor induction of VEGF promoter activity in stromal cells. Cell 94: 715–725.
Gasparini G, Harris AL . (1995). Clinical importance of the determination of tumor angiogenesis in breast carcinoma: much more than a new prognostic tool. J Clin Oncol 13: 765–782.
Gee MF, Tsuchida R, Eichler-Jonsson C, Das B, Baruchel S, Malkin D . (2005). Vascular endothelial growth factor acts in an autocrine manner in rhabdomyosarcoma cell lines and can be inhibited with all-trans-retinoic acid. Oncogene 24: 8025–8037.
Hirschmann-Jax C, Foster AE, Wulf GG, Goodell MA, Brenner MK . (2005). A distinct ‘side population’ of cells in human tumor cells: implications for tumor biology and therapy. Cell Cycle 4: 203–205.
Hirschmann-Jax C, Foster AE, Wulf GG, Nuchtern JG, Jax TW, Gobel U et al. (2004). A distinct ‘side population’ of cells with high drug efflux capacity in human tumor cells. Proc Natl Acad Sci USA 101: 14228–14233.
Ho MM, Ng AV, Lam S, Hung JY . (2007). Side population in human lung cancer cell lines and tumors is enriched with stem-like cancer cells. Cancer Res 67: 4827–4833.
Kim JJ, Tannock IF . (2005). Repopulation of cancer cells during therapy: an important cause of treatment failure. Nat Rev Cancer 5: 516–525.
Kondo T, Setoguchi T, Taga T . (2004). Persistence of a small subpopulation of cancer stem-like cells in the C6 glioma cell line. Proc Natl Acad Sci USA 101: 781–786.
Lin X, Costa M . (1994). Transformation of human osteoblasts to anchorage-independent growth by insoluble nickel particles. Environ Health Perspect 102 (Suppl 3): 289–292.
McAllister RM, Gardner MB, Greene AE, Bradt C, Nichols WW, Landing BH . (1971). Cultivation in vitro of cells derived from a human osteosarcoma. Cancer 27: 397–402.
Mercurio AM, Bachelder RE, Bates RC, Chung J . (2004). Autocrine signaling in carcinoma: VEGF and the alpha6beta4 integrin. Semin Cancer Biol 14: 115–122.
Miller AC, Blakely WF, Livengood D, Whittaker T, Xu J, Ejnik JW et al. (1998). Transformation of human osteoblast cells to the tumorigenic phenotype by depleted uranium-uranyl chloride. Environ Health Perspect 106: 465–471.
Miller AC, Mog S, McKinney L, Luo L, Allen J, Xu J et al. (2001). Neoplastic transformation of human osteoblast cells to the tumorigenic phenotype by heavy metal-tungsten alloy particles: induction of genotoxic effects. Carcinogenesis 22: 115–125.
Miura K, Uniyal S, Leabu M, Oravecz T, Chakrabarti S, Morris VL et al. (2005). Chemokine receptor CXCR4-beta1 integrin axis mediates tumorigenesis of osteosarcoma HOS cells. Biochem Cell Biol 83: 36–48.
Montanaro F, Liadaki K, Schienda J, Flint A, Gussoni E, Kunkel LM . (2004). Demystifying SP cell purification: viability, yield, and phenotype are defined by isolation parameters. Exp Cell Res 298: 144–154.
Patrawala L, Calhoun T, Schneider-Broussard R, Zhou J, Claypool K, Tang DG . (2005). Side population is enriched in tumorigenic, stem-like cancer cells, whereas ABCG2+ and ABCG2− cancer cells are similarly tumorigenic. Cancer Res 65: 6207–6219.
Patrawala L, Calhoun-Davis T, Schneider-Broussard R, Tang DG . (2007). Hierarchical organization of prostate cancer cells in xenograft tumors: the CD44+{alpha}2{beta}1+ cell population is enriched in tumor-initiating cells. Cancer Res 67: 6796–6805.
Qi L, Robinson WA, Brady BM, Glode LM . (2003). Migration and invasion of human prostate cancer cells is related to expression of VEGF and its receptors. Anticancer Res 23: 3917–3922.
Ranganathan AC, Adam AP, Aguirre-Ghiso JA . (2006). Opposing roles of mitogenic and stress signaling pathways in the induction of cancer dormancy. Cell Cycle 5: 1799–1807.
Richman CM, Weiner RS, Yankee RA . (1976). Increase in circulating stem cells following chemotherapy in man. Blood 47: 1031–1039.
Salnikow K, An WG, Melillo G, Blagosklonny MV, Costa M . (1999). Nickel-induced transformation shifts the balance between HIF-1 and p53 transcription factors. Carcinogenesis 20: 1819–1823.
Setoguchi T, Taga T, Kondo T . (2004). Cancer stem cells persist in many cancer cell lines. Cell Cycle 3: 414–415.
Soker S, Kaefer M, Johnson M, Klagsbrun M, Atala A, Freeman MR . (2001). Vascular endothelial growth factor-mediated autocrine stimulation of prostate tumor cells coincides with progression to a malignant phenotype. Am J Pathol 159: 651–659.
Steiner HH, Karcher S, Mueller MM, Nalbantis E, Kunze S, Herold-Mende C . (2004). Autocrine pathways of the vascular endothelial growth factor (VEGF) in glioblastoma multiforme: clinical relevance of radiation-induced increase of VEGF levels. J Neurooncol 66: 129–138.
Strizzi L, Catalano A, Vianale G, Orecchia S, Casalini A, Tassi G et al. (2001). Vascular endothelial growth factor is an autocrine growth factor in human malignant mesothelioma. J Pathol 193: 468–475.
Woessmann W, Chen X, Borkhardt A . (2002). Ras-mediated activation of ERK by cisplatin induces cell death independently of p53 in osteosarcoma and neuroblastoma cell lines. Cancer Chemother Pharmacol 50: 397–404.
Acknowledgements
This work is supported by the National Cancer Institute of Canada, the Andrew Mizzoni Cancer Research Fund and the Harry and Hannah Fisher Research Fund. RT and BD are supported in part by awards of the Hospital for Sick Children's Research Training Centre and the National Cancer Institute of Canada Fellowship with funds from the Terry Fox Foundation. We thank Dr Meredith Irwin for critical review of the manuscript; Sherry Zhao, Shamim Lotif, Micky Tsui, Reza Mokhtari, Michael Ho, Suzanne McGovern and Ana Novokmet for technical assistance; and the staff of the animal facility of the Hospital for Sick Children for their technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Tsuchida, R., Das, B., Yeger, H. et al. Cisplatin treatment increases survival and expansion of a highly tumorigenic side-population fraction by upregulating VEGF/Flt1 autocrine signaling. Oncogene 27, 3923–3934 (2008). https://doi.org/10.1038/onc.2008.38
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.38
Keywords
This article is cited by
-
The effect of thymoquinone and propranolol combination on epidermoid laryngeal carcinoma cell
European Archives of Oto-Rhino-Laryngology (2023)
-
The correlation between LDH serum levels and clinical outcome in advanced biliary tract cancer patients treated with first line chemotherapy
Scientific Reports (2016)
-
Perhexiline maleate enhances antitumor efficacy of cisplatin in neuroblastoma by inducing over-expression of NDM29 ncRNA
Scientific Reports (2015)
-
BMP4/Thrombospondin-1 loop paracrinically inhibits tumor angiogenesis and suppresses the growth of solid tumors
Oncogene (2014)
-
Isolation and identification of cancer stem cells from a side population of a human hepatoblastoma cell line, HuH-6 clone-5
Pediatric Surgery International (2011)