Abstract
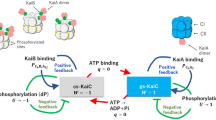
KaiA, KaiB and KaiC constitute the circadian clock machinery in cyanobacteria, and KaiA activates kaiBC expression whereas KaiC represses it. Here we show that KaiA is composed of three functional domains, the N-terminal amplitude-amplifier domain, the central period-adjuster domain and the C-terminal clock-oscillator domain. The C-terminal domain is responsible for dimer formation, binding to KaiC, enhancing KaiC phosphorylation and generating the circadian oscillations. The X-ray crystal structure at a resolution of 1.8 Å of the C-terminal clock-oscillator domain of KaiA from the thermophilic cyanobacterium Thermosynechococcus elongatus BP-1 shows that residue His270, located at the center of a KaiA dimer concavity, is essential to KaiA function. KaiA binding to KaiC probably occurs via the concave surface. On the basis of the structure, we predict the structural roles of the residues that affect circadian oscillations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Dunlap, J.C. Molecular bases for circadian clocks. Cell 96, 271–290 (1999).
Young, M.W. & Kay, S.A. Time zones: a comparative genetics of circadian clocks. Nat. Rev. Genet. 2, 702–715 (2001).
Grobbelaar, N., Huang, T.C., Lin, H.Y. & Chow, T.J. Dinitrogen-fixing endogenous rhythm in Synechococcus RF-1. FEMS Microbiol. Lett. 37, 173–177 (1986).
Mitsui, A. et al. Strategy by which nitrogen-fixing unicellular cyanobacteria grow photoautotrophically. Nature 323, 720–722 (1986).
Kondo, T. et al. Circadian rhythms in prokaryotes: Luciferase as a reporter of circadian gene expression in cyanobacteria. Proc. Natl. Acad. Sci. USA 90, 5672–5676 (1993).
Aoki, S., Kondo, T. & Ishiura, M. Circadian expression of the dnaK gene in the cyanobacterium Synechocystis sp. strain PCC 6803. J. Bacteriol. 177, 5606–5611 (1995).
Chen, Y.B., Dominic, B., Mellon, M.T. & Zehr, J.P. Circadian rhythm of nitrogenase gene expression in the diazotrophic filamentous nonheterocystous cyanobacterium Trichodesmium sp. strain IMS 101. J. Bacteriol. 180, 3598–3605 (1998).
Ishiura, M. et al. Expression of a gene cluster kaiABC as a circadian feedback process in cyanobacteria. Science 281, 1519–1523 (1998).
Iwasaki, H., Taniguchi, Y., Ishiura, M. & Kondo, T. Physical interactions among circadian clock proteins KaiA, KaiB and KaiC in cyanobacteria. EMBO J. 18, 1137–1145 (1999).
Hayashi, F. et al. Stoichiometric interactions between cyanobacterial clock proteins KaiA and KaiC. Biochem. Biophys. Res. Comm. 316, 195–202 (2004).
Nishiwaki, T., Iwasaki, H., Ishiura, M. & Kondo, T. Nucleotide binding and autophosphorylation of the clock protein KaiC as a circadian timing process of cyanobacteria. Proc. Natl. Acad. Sci. USA 97, 495–499 (2000).
Taniguchi, Y. et al. Two KaiA-binding domains of cyanobacterial circadian clock protein KaiC. FEBS Lett. 496, 86–90 (2001).
Nishimura, H. et al. Mutations in KaiA, a clock protein, extend the period of circadian rhythm in the cyanobacterium Synechococcus elongatus PCC 7942. Microbiology 148, 2903–2909 (2002).
Iwasaki, H., Nishiwaki, T., Kitayama, Y., Nakajima, M. & Kondo, T. KaiA-stimulated KaiC phosphorylation in circadian timing loops in cyanobacteria. Proc. Natl. Acad. Sci. USA 99, 15788–15793 (2002).
Yamaoka, T., Satoh, K. & Katoh, S. Photosynthetic activities of a thermophilic blue-green alga. Plant Cell Physiol. 19, 943–954 (1978).
Van Baalen, C. Studies on marine blue-green algae. Bot. Mar. 4, 129–139 (1962).
Rippka, R., Deruelles, J., Waterbury, J.B., Herdman, M. & Stanier, R.Y. Generic assignments, strain histories, and properties of pure cultures of cyanobacteria. J. Gen. Microbiol. 111, 1–61 (1979).
Marquardt, J., Senger, H., Miyashita, H., Miyachi, S. & Morschel, E. Isolation and characterization of biliprotein aggregates from Acaryochloris marina, a Prochloron-like prokaryote containing mainly chlorophyll d. FEBS Lett. 410, 428–432 (1997).
Hirose, M. Energy supply system for the gliding movement of hormogonia of the cyanobacterium Nostoc cycadae. Plant Cell Physiol. 28, 584–597 (1987).
Kaneko, T. et al. Complete genomic sequence of the filamentous nitrogen-fixing cyanobacterium Anabaena sp. strain PCC 7120. DNA Res. 8, 205–213 (2001).
Hendrickson, W.A., Horton, J.R. & LeMaster, D.M. Selenomethionyl proteins produced for analysis by multiwavelength anomalous diffraction (MAD): a vehicle for direct determination of three-dimensional structure. EMBO J. 9, 1665–1672 (1990).
Vakonakis, I. et al. NMR structure of the KaiC-interacting C-terminal domain of KaiA, a circadian clock protein: Implications for KaiA-KaiC interaction. Proc. Natl. Acad. Sci. USA 101, 1479–1484 (2004).
Ye, S., Vakonakis, I., Ioerger, T.R., LiWang, A.C. & Sacchettini, J.C. Crystal structure of circadian clock protein KaiA from Synechococcus elongatus. J. Biol. Chem. 279, 20511–20518 (2004).
Onai, K., Morishita, M., Itoh, S., Okamoto, K., & Ishiura, M. Circadian rhythms in the thermophilic cyanobacterium Thermosynechococcus elongatus: Compensation of period length over wide temperature range. J. Bacteriol. (in the press).
Williams, S.B., Vakonakis, I., Golden, S.S. & LiWang, A.C. Structure and function from the circadian clock protein KaiA of Synechococcus elongatus: A potential clock input mechanism. Proc. Natl. Acad. Sci. USA 99, 15357–15362 (2002).
Hayashi, F. et al. ATP-induced hexameric ring structure of the cyanobacterial circadian clock protein KaiC. Genes Cells 8, 287–296 (2003).
Onai, K., Morishita, M., Kaneko, T., Tabata, S. & Ishiura, M. Natural transformation of the thermophilic cyanobacterium Thermosynechococcus elongatus BP-1: A simple and efficient method for gene transfer. Mol. Genet. Genom. 271, 50–59 (2004).
Pflugrath, J.W. The finer things in X-ray diffraction data collection. Acta Crystallogr. D 55, 1718–1725 (1999).
Terwilliger, T.C. & Berendzen, J. Automated structure solution for MIR and MAD. Acta Crystallogr. D 55, 849–861 (1999).
Perrakis, A., Morris, R. & Lamzin, V.S. Automated protein model building combined with iterative structure refinement. Nature Struct. Biol. 6, 458–463 (1999).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997).
Roussel, A. & Cambillau, C. Turbo-Frodo Manual (AFMB-CNRS, Marseille, France, 1996).
Thompson, J.D., Higgins, D.G. & Gibson, T.J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positions-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680 (1994).
Kraulis, P.J. MOLSCRIPT: a program to produce both detailed and schematic plots of protein structures. J. Appl. Crystallogr. 24, 946–950 (1991).
Merritt, E.A. & Bacon, D.J. Raster3D: photorealistic molecular graphics. Methods Enzymol. 277, 505–524 (1997).
Acknowledgements
We thank J.-R. Shen and Y. Inoue (RIKEN Harima Institute) for T. vulcanus, M. Ikeuchi for T. elongatus, S. Itoh (Nagoya University) for Synechococcus 7002 and A. marina, M. Hirose (Wakayama University) for N. cycadae, and C.P. Wolk (Michigan State University) for Anabaena. We thank K. Onai and M. Morishita for their help in rhythm analysis. We thank M. Yamamoto (RIKEN Harima Institute) for X-ray diffraction data collection on the RIKEN beamline BL26B1, B2 at SPring-8. We thank M. Bloom (SciWrite Biomedical Writing & Editing Services) for professional editing. This research was supported by grants from the Japanese Ministry of Education, Science and Culture (MEXT), the Naito Foundation, PROBRAIN, the Future of Japan Society for the Promotion of Science, the Japan Space Forum, the Aichi Science and Technology Foundation, and the National Project on Protein Structural and Functional Analyses by MEXT to M.I. The Division of Biological Science, Graduate School of Science, Nagoya University, was supported by a 21st Century Center of Excellence grant from MEXT.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Alignment of the deduced amino acid sequences of KaiAs from 11 strains of cyanobacteria. (PDF 35 kb)
Supplementary Fig. 2
Rescue of circadian bioluminescence rhythms in a Synechococcus kaiA-null mutant strain by transferring the T. elongatus kaiA gene into the strain. (PDF 30 kb)
Supplementary Fig. 3
Density map (stereo view) showing the atomic arrangement in the region of the functionally essential residue His270. (PDF 53 kb)
Supplementary Fig. 4
The biophysical properties of T. elongatus KaiA(174–283) WT (the C-terminal clock-oscillator domain) and T. elongatus KaiA(174–283) H270A. (PDF 31 kb)
Supplementary Fig. 5
Differences in the in vivo rhythm assay procedures used between a previous report13 and this study. (PDF 34 kb)
Supplementary Fig. 6
Superposition of the crystal structure of KaiA(174–283) with ThKaiA180C NMR solution structure22. (PDF 57 kb)
Rights and permissions
About this article
Cite this article
Uzumaki, T., Fujita, M., Nakatsu, T. et al. Crystal structure of the C-terminal clock-oscillator domain of the cyanobacterial KaiA protein. Nat Struct Mol Biol 11, 623–631 (2004). https://doi.org/10.1038/nsmb781
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb781
This article is cited by
-
Circadian oscillator proteins across the kingdoms of life: structural aspects
BMC Biology (2019)
-
Minimal tool set for a prokaryotic circadian clock
BMC Evolutionary Biology (2017)
-
Structural and dynamic aspects of protein clocks: how can they be so slow and stable?
Cellular and Molecular Life Sciences (2012)
-
Thermodynamically Induced Conformational Changes of the Cyanobacterial Circadian Clock Protein KaiB
Applied Magnetic Resonance (2011)
-
A sequestration feedback determines dynamics and temperature entrainment of the KaiABC circadian clock
Molecular Systems Biology (2010)