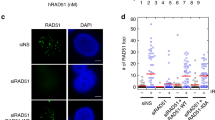
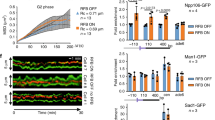
Abstract
In higher eukaryotes, the dynamics of replisome components during fork collapse and restart are poorly understood. Here we have reconstituted replication fork collapse and restart by inducing single-strand DNA lesions that create a double-strand break in one of the replicated sister chromatids after fork passage. We found that, upon fork collapse, the active CDC45–MCM–GINS (CMG) helicase complex loses its GINS subunit. A functional replisome is restored by the reloading of GINS and polymerase ɛ onto DNA in a fashion that is dependent on RAD51 and MRE11 but independent of replication origin assembly and firing. PCNA mutant alleles defective in break-induced replication (BIR) are unable to support restoration of replisome integrity. These results show that, in higher eukaryotes, replisomes are partially dismantled after fork collapse and fully re-established by a recombination-mediated process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Michel, B., Grompone, G., Flores, M.J. & Bidnenko, V. Multiple pathways process stalled replication forks. Proc. Natl. Acad. Sci. USA 101, 12783–12788 (2004).
Santocanale, C., Sharma, K. & Diffley, J.F. Activation of dormant origins of DNA replication in budding yeast. Genes Dev. 13, 2360–2364 (1999).
Woodward, A.M. et al. Excess Mcm2–7 license dormant origins of replication that can be used under conditions of replicative stress. J. Cell Biol. 173, 673–683 (2006).
Petermann, E. & Helleday, T. Pathways of mammalian replication fork restart. Nat. Rev. Mol. Cell Biol. 11, 683–687 (2010).
Chang, D.J. & Cimprich, K.A. DNA damage tolerance: when it's OK to make mistakes. Nat. Chem. Biol. 5, 82–90 (2009).
Andersen, P.L., Xu, F. & Xiao, W. Eukaryotic DNA damage tolerance and translesion synthesis through covalent modifications of PCNA. Cell Res. 18, 162–173 (2008).
Daigaku, Y., Davies, A.A. & Ulrich, H.D. Ubiquitin-dependent DNA damage bypass is separable from genome replication. Nature 465, 951–955 (2010).
Karras, G.I. & Jentsch, S. The RAD6 DNA damage tolerance pathway operates uncoupled from the replication fork and is functional beyond S phase. Cell 141, 255–267 (2010).
Lambert, S. et al. Homologous recombination restarts blocked replication forks at the expense of genome rearrangements by template exchange. Mol. Cell 39, 346–359 (2010).
Moriel-Carretero, M. & Aguilera, A. A postincision-deficient TFIIH causes replication fork breakage and uncovers alternative Rad51- or Pol32-mediated restart mechanisms. Mol. Cell 37, 690–701 (2010).
Petermann, E., Orta, M.L., Issaeva, N., Schultz, N. & Helleday, T. Hydroxyurea-stalled replication forks become progressively inactivated and require two different RAD51-mediated pathways for restart and repair. Mol. Cell 37, 492–502 (2010).
Huertas, P. DNA resection in eukaryotes: deciding how to fix the break. Nat. Struct. Mol. Biol. 17, 11–16 (2010).
Llorente, B., Smith, C.E. & Symington, L.S. Break-induced replication: what is it and what is it for? Cell Cycle 7, 859–864 (2008).
Lydeard, J.R. et al. Break-induced replication requires all essential DNA replication factors except those specific for pre-RC assembly. Genes Dev. 24, 1133–1144 (2010).
Gambus, A. et al. GINS maintains association of Cdc45 with MCM in replisome progression complexes at eukaryotic DNA replication forks. Nat. Cell Biol. 8, 358–366 (2006).
Tanaka, S. & Araki, H. Regulation of the initiation step of DNA replication by cyclin-dependent kinases. Chromosoma 119, 565–574 (2010).
Moyer, S.E., Lewis, P.W. & Botchan, M.R. Isolation of the Cdc45/Mcm2–7/GINS (CMG) complex, a candidate for the eukaryotic DNA replication fork helicase. Proc. Natl. Acad. Sci. USA 103, 10236–10241 (2006).
Errico, A. & Costanzo, V. Differences in the DNA replication of unicellular eukaryotes and metazoans: known unknowns. EMBO Rep. 11, 270–278 (2010).
De Haro, L.P. et al. Metnase promotes restart and repair of stalled and collapsed replication forks. Nucleic Acids Res. 38, 5681–5691 (2010).
Hashimoto, Y., Chaudhuri, A.R., Lopes, M. & Costanzo, V. Rad51 protects nascent DNA from Mre11-dependent degradation and promotes continuous DNA synthesis. Nat. Struct. Mol. Biol. 17, 1305–1311 (2010).
Trenz, K., Errico, A. & Costanzo, V. Plx1 is required for chromosomal DNA replication under stressful conditions. EMBO J. 27, 876–885 (2008).
Krokan, H., Wist, E. & Krokan, R.H. Aphidicolin inhibits DNA synthesis by DNA polymerase alpha and isolated nuclei by a similar mechanism. Nucleic Acids Res. 9, 4709–4719 (1981).
Sutherland, G.R. The role of nucleotides in human fragile site expression. Mutat. Res. 200, 207–213 (1988).
Balestrini, A., Cosentino, C., Errico, A., Garner, E. & Costanzo, V. GEMC1 is a TopBP1-interacting protein required for chromosomal DNA replication. Nat. Cell Biol. 12, 484–491 (2010).
Chong, J.P., Mahbubani, H.M., Khoo, C.Y. & Blow, J.J. Purification of an MCM-containing complex as a component of the DNA replication licensing system. Nature 375, 418–421 (1995).
McGarry, T.J. & Kirschner, M.W. Geminin, an inhibitor of DNA replication, is degraded during mitosis. Cell 93, 1043–1053 (1998).
Costanzo, V. et al. Reconstitution of an ATM-dependent checkpoint that inhibits chromosomal DNA replication following DNA damage. Mol. Cell 6, 649–659 (2000).
Falck, J., Petrini, J.H., Williams, B.R., Lukas, J. & Bartek, J. The DNA damage-dependent intra-S phase checkpoint is regulated by parallel pathways. Nat. Genet. 30, 290–294 (2002).
Edwards, M.C. et al. MCM2–7 complexes bind chromatin in a distributed pattern surrounding the origin recognition complex in Xenopus egg extracts. J. Biol. Chem. 277, 33049–33057 (2002).
Pacek, M., Tutter, A.V., Kubota, Y., Takisawa, H. & Walter, J.C. Localization of MCM2–7, Cdc45, and GINS to the site of DNA unwinding during eukaryotic DNA replication. Mol. Cell 21, 581–587 (2006).
Mimura, S. & Takisawa, H. Xenopus Cdc45-dependent loading of DNA polymerase alpha onto chromatin under the control of S-phase Cdk. EMBO J. 17, 5699–5707 (1998).
Errico, A. et al. Tipin/Tim1/And1 protein complex promotes Pol alpha chromatin binding and sister chromatid cohesion. EMBO J. 28, 3681–3692 (2009).
Matsuoka, S. et al. ATM and ATR substrate analysis reveals extensive protein networks responsive to DNA damage. Science 316, 1160–1166 (2007).
Sclafani, R.A. & Holzen, T.M. Cell cycle regulation of DNA replication. Annu. Rev. Genet. 41, 237–280 (2007).
Meijer, L. et al. Biochemical and cellular effects of roscovitine, a potent and selective inhibitor of the cyclin-dependent kinases cdc2, cdk2 and cdk5. Eur. J. Biochem. 243, 527–536 (1997).
Trenz, K., Smith, E., Smith, S. & Costanzo, V. ATM and ATR promote Mre11 dependent restart of collapsed replication forks and prevent accumulation of DNA breaks. EMBO J. 25, 1764–1774 (2006).
Bryant, H.E. et al. PARP is activated at stalled forks to mediate Mre11-dependent replication restart and recombination. EMBO J. 28, 2601–2615 (2009).
Ira, G. & Haber, J.E. Characterization of RAD51-independent break-induced replication that acts preferentially with short homologous sequences. Mol. Cell. Biol. 22, 6384–6392 (2002).
Davis, A.P. & Symington, L.S. RAD51-dependent break-induced replication in yeast. Mol. Cell. Biol. 24, 2344–2351 (2004).
Malkova, A. et al. RAD51-independent break-induced replication to repair a broken chromosome depends on a distant enhancer site. Genes Dev. 15, 1055–1060 (2001).
Signon, L., Malkova, A., Naylor, M.L., Klein, H. & Haber, J.E. Genetic requirements for RAD51- and RAD54-independent break-induced replication repair of a chromosomal double-strand break. Mol. Cell. Biol. 21, 2048–2056 (2001).
Kraus, E., Leung, W.Y. & Haber, J.E. Break-induced replication: a review and an example in budding yeast. Proc. Natl. Acad. Sci. USA 98, 8255–8262 (2001).
Li, S.J. & Hochstrasser, M. The Ulp1 SUMO isopeptidase: distinct domains required for viability, nuclear envelope localization, and substrate specificity. J. Cell Biol. 160, 1069–1081 (2003).
Kubota, Y. et al. A novel ring-like complex of Xenopus proteins essential for the initiation of DNA replication. Genes Dev. 17, 1141–1152 (2003).
Takayama, Y. et al. GINS, a novel multiprotein complex required for chromosomal DNA replication in budding yeast. Genes Dev. 17, 1153–1165 (2003).
Costa, A. et al. The structural basis for MCM2–7 helicase activation by GINS and Cdc45. Nat. Struct. Mol. Biol. 18, 471–477 (2011).
Bruck, I. & Kaplan, D.L. GINS and Sld3 compete with one another for Mcm2–7 and Cdc45 binding. J. Biol. Chem. 286, 14157–14167 (2011).
Bjergbaek, L., Cobb, J.A., Tsai-Pflugfelder, M. & Gasser, S.M. Mechanistically distinct roles for Sgs1p in checkpoint activation and replication fork maintenance. EMBO J. 24, 405–417 (2005).
Cobb, J.A. et al. Replisome instability, fork collapse, and gross chromosomal rearrangements arise synergistically from Mec1 kinase and RecQ helicase mutations. Genes Dev. 19, 3055–3069 (2005).
Tittel-Elmer, M., Alabert, C., Pasero, P. & Cobb, J.A. The MRX complex stabilizes the replisome independently of the S phase checkpoint during replication stress. EMBO J. 28, 1142–1156 (2009).
Muramatsu, S., Hirai, K., Tak, Y.S., Kamimura, Y. & Araki, H. CDK-dependent complex formation between replication proteins Dpb11, Sld2, Pol ɛ, and GINS in budding yeast. Genes Dev. 24, 602–612 (2010).
Kawamoto, T. et al. Dual roles for DNA polymerase eta in homologous DNA recombination and translesion DNA synthesis. Mol. Cell 20, 793–799 (2005).
McIlwraith, M.J. et al. Human DNA polymerase eta promotes DNA synthesis from strand invasion intermediates of homologous recombination. Mol. Cell 20, 783–792 (2005).
Stephens, P.J. et al. Complex landscapes of somatic rearrangement in human breast cancer genomes. Nature 462, 1005–1010 (2009).
Buis, J. et al. Mre11 nuclease activity has essential roles in DNA repair and genomic stability distinct from ATM activation. Cell 135, 85–96 (2008).
Acknowledgements
We are grateful to J. Haber and J. Lydeard for sharing unpublished results on PCNA BIR–defective mutants. We thank members of the genome stability lab for their insightful comments. We thank H. Mahbubani and J. Gannon for technical support with X. laevis. This work was funded by Cancer Research UK. V.C. is also supported by the European Research Council (ERC) startup grant (206281), the Lister Institute of Preventive Medicine and the European Molecular Biology Organization (EMBO) Young Investigator Program (YIP).
Author information
Authors and Affiliations
Contributions
Y.H. and F.P. performed experiments. Y.H. and V.C. analyzed the results and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 3459 kb)
Rights and permissions
About this article
Cite this article
Hashimoto, Y., Puddu, F. & Costanzo, V. RAD51- and MRE11-dependent reassembly of uncoupled CMG helicase complex at collapsed replication forks. Nat Struct Mol Biol 19, 17–24 (2012). https://doi.org/10.1038/nsmb.2177
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2177
This article is cited by
-
The RAD51 recombinase protects mitotic chromatin in human cells
Nature Communications (2021)
-
Acute hydroxyurea-induced replication blockade results in replisome components disengagement from nascent DNA without causing fork collapse
Cellular and Molecular Life Sciences (2020)
-
Replication fork reversal triggers fork degradation in BRCA2-defective cells
Nature Communications (2017)
-
The end-joining factor Ku acts in the end-resection of double strand break-free arrested replication forks
Nature Communications (2017)
-
Replication stress: getting back on track
Nature Structural & Molecular Biology (2016)