Abstract
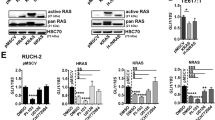
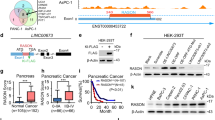
Synergism between the RAS and Hedgehog (HH) pathways has been suggested for carcinogenesis in the pancreas, lung and colon. We investigated the molecular cross-talk between RAS and HH signaling and found that, although mutant RAS induces or enhances SHH expression and favors paracrine HH signaling, it antagonizes autocrine HH signal transduction. Activated RAS can be found in primary cilia, the central organelle of HH signal transduction, but functions in a cilium-independent manner and interferes with Gli2 function and Gli3 processing. In addition, the cell-autonomous negative regulation of HH signal transduction involves the RAS effector molecule dual specificity tyrosine phosphorylated and regulated kinase 1B (DYRK1B). In line with a redirection of autocrine toward paracrine HH signaling by a KRAS-DYRK1B network, we find high levels of GLI1 expression restricted to the stromal compartment and not to SHH-expressing tumor cells in human pancreatic adenocarcinoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Rubin, L.L. & de Sauvage, F.J. Targeting the Hedgehog pathway in cancer. Nat. Rev. Drug Discov. 5, 1026–1033 (2006).
Watkins, D.N. et al. Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 422, 313–317 (2003).
Berman, D.M. et al. Widespread requirement for Hedgehog ligand stimulation in growth of digestive tract tumours. Nature 425, 846–851 (2003).
Thayer, S.P. et al. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 425, 851–856 (2003).
Stecca, B. et al. Melanomas require HEDGEHOG-GLI signaling regulated by interactions between GLI1 and the RAS-MEK/AKT pathways. Proc. Natl. Acad. Sci. USA 104, 5895–5900 (2007).
Malumbres, M. & Barbacid, M. RAS oncogenes: the first 30 years. Nat. Rev. Cancer 3, 459–465 (2003).
Hezel, A.F., Kimmelman, A.C., Stanger, B.Z., Bardeesy, N. & Depinho, R.A. Genetics and biology of pancreatic ductal adenocarcinoma. Genes Dev. 20, 1218–1249 (2006).
Hingorani, S.R. et al. Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell 7, 469–483 (2005).
Feldmann, G. et al. Blockade of hedgehog signaling inhibits pancreatic cancer invasion and metastases: a new paradigm for combination therapy in solid cancers. Cancer Res. 67, 2187–2196 (2007).
Feldmann, G. et al. Hedgehog inhibition prolongs survival in a genetically engineered mouse model of pancreatic cancer. Gut 57, 1420–1430 (2008).
Jones, S. et al. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science 321, 1801–1806 (2008).
Yauch, R.L. et al. A paracrine requirement for hedgehog signalling in cancer. Nature 455, 406–410 (2008).
Habbe, N. et al. Spontaneous induction of murine pancreatic intraepithelial neoplasia (mPanIN) by acinar cell targeting of oncogenic Kras in adult mice. Proc. Natl. Acad. Sci. USA 105, 18913–18918 (2008).
De La O, J.P. et al. Notch and Kras reprogram pancreatic acinar cells to ductal intraepithelial neoplasia. Proc. Natl. Acad. Sci. USA 105, 18907–18912 (2008).
Guerra, C. et al. Chronic pancreatitis is essential for induction of pancreatic ductal adenocarcinoma by K-Ras oncogenes in adult mice. Cancer Cell 11, 291–302 (2007).
Morton, J.P. et al. Sonic hedgehog acts at multiple stages during pancreatic tumorigenesis. Proc. Natl. Acad. Sci. USA 104, 5103–5108 (2007).
Fendrich, V. et al. Hedgehog signaling is required for effective regeneration of exocrine pancreas. Gastroenterology 135, 621–631 (2008).
Schubbert, S., Shannon, K. & Bollag, G. Hyperactive Ras in developmental disorders and cancer. Nat. Rev. Cancer 7, 295–308 (2007).
Quint, K. et al. The expression pattern of PDX-1, SHH, Patched and Gli-1 is associated with pathological and clinical features in human pancreatic cancer. Pancreatology 9, 116–126 (2009).
Taipale, J. et al. Effects of oncogenic mutations in Smoothened and Patched can be reversed by cyclopamine. Nature 406, 1005–1009 (2000).
Chen, J.K., Taipale, J., Young, K.E., Maiti, T. & Beachy, P.A. Small molecule modulation of Smoothened activity. Proc. Natl. Acad. Sci. USA 99, 14071–14076 (2002).
Guerra, C. et al. Tumor induction by an endogenous K-ras oncogene is highly dependent on cellular context. Cancer Cell 4, 111–120 (2003).
Wakabayashi, Y., Mao, J.H., Brown, K., Girardi, M. & Balmain, A. Promotion of Hras-induced squamous carcinomas by a polymorphic variant of the Patched gene in FVB mice. Nature 445, 761–765 (2007).
Svard, J. et al. Genetic elimination of Suppressor of fused reveals an essential repressor function in the mammalian Hedgehog signaling pathway. Dev. Cell 10, 187–197 (2006).
Haycraft, C.J. et al. Gli2 and Gli3 localize to cilia and require the intraflagellar transport protein polaris for processing and function. PLoS Genet. 1, e53 (2005).
Schneider, L. et al. PDGFRalphaalpha signaling is regulated through the primary cilium in fibroblasts. Curr. Biol. 15, 1861–1866 (2005).
Lauth, M., Bergstrom, A. & Toftgard, R. Phorbol esters inhibit the Hedgehog signalling pathway downstream of Suppressor of Fused, but upstream of Gli. Oncogene 26, 5163–5168 (2007).
Jin, K., Park, S., Ewton, D.Z. & Friedman, E. The survival kinase Mirk/Dyrk1B is a downstream effector of oncogenic K-ras in pancreatic cancer. Cancer Res. 67, 7247–7255 (2007).
Mao, J. et al. Regulation of Gli1 transcriptional activity in the nucleus by Dyrk1. J. Biol. Chem. 277, 35156–35161 (2002).
Varjosalo, M. et al. Application of active and kinase-deficient kinome collection for identification of kinases regulating hedgehog signaling. Cell 133, 537–548 (2008).
Heidenblad, M. et al. Genome-wide array-based comparative genomic hybridization reveals multiple amplification targets and novel homozygous deletions in pancreatic carcinoma cell lines. Cancer Res. 64, 3052–3059 (2004).
Seeley, E.S., Carriere, C., Goetze, T., Longnecker, D.S. & Korc, M. Pancreatic cancer and precursor pancreatic intraepithelial neoplasia lesions are devoid of primary cilia. Cancer Res. 69, 422–430 (2009).
Huangfu, D. et al. Hedgehog signalling in the mouse requires intraflagellar transport proteins. Nature 426, 83–87 (2003).
Nolan-Stevaux, O. et al. GLI1 is regulated through Smoothened-independent mechanisms in neoplastic pancreatic ducts and mediates PDAC cell survival and transformation. Genes Dev. 23, 24–36 (2009).
Dennler, S. et al. Induction of sonic hedgehog mediators by transforming growth factor-beta: Smad3-dependent activation of Gli2 and Gli1 expression in vitro and in vivo. Cancer Res. 67, 6981–6986 (2007).
Steg, A. et al. Hedgehog pathway expression in heterogeneous pancreatic adenocarcinoma: implications for the molecular analysis of clinically available biopsies. Diagn. Mol. Pathol. 16, 229–237 (2007).
Nielsen, S.K. et al. Characterization of primary cilia and Hedgehog signaling during development of the human pancreas and in human pancreatic duct cancer cell lines. Dev. Dyn. 237, 2039–2052 (2008).
Galvin, K.E., Ye, H., Erstad, D.J., Feddersen, R. & Wetmore, C. Gli1 induces G2/M arrest and apoptosis in hippocampal but not tumor-derived neural stem cells. Stem Cells 26, 1027–1036 (2008).
Lauth, M. & Toftgard, R. Non-canonical activation of GLI transcription factors: implications for targeted anti-cancer therapy. Cell Cycle 6, 2458–2463 (2007).
Kasai, K., Inaguma, S., Yoneyama, A., Yoshikawa, K. & Ikeda, H. SCL/TAL1 interrupting locus derepresses GLI1 from the negative control of suppressor-of-fused in pancreatic cancer cell. Cancer Res. 68, 7723–7729 (2008).
Ji, Z., Mei, F.C., Xie, J. & Cheng, X. Oncogenic KRAS activates hedgehog signaling pathway in pancreatic cancer cells. J. Biol. Chem. 282, 14048–14055 (2007).
Tian, H. et al. Hedgehog signaling is restricted to the stromal compartment during pancreatic carcinogenesis. Proc. Natl. Acad. Sci. USA 106, 4254–4259 (2009).
Prasad, N.B. et al. Gene expression profiles in pancreatic intraepithelial neoplasia reflect the effects of Hedgehog signaling on pancreatic ductal epithelial cells. Cancer Res. 65, 1619–1626 (2005).
Lauth, M., Bergstrom, A., Shimokawa, T. & Toftgard, R. Inhibition of GLI-mediated transcription and tumor cell growth by small-molecule antagonists. Proc. Natl. Acad. Sci. USA 104, 8455–8460 (2007).
Nakashima, H. et al. Nuclear factor-κB contributes to hedgehog signaling pathway activation through sonic hedgehog induction in pancreatic cancer. Cancer Res. 66, 7041–7049 (2006).
Shimokawa, T. et al. Distinct roles of first exon variants of the tumor-suppressor Patched1 in Hedgehog signaling. Oncogene 26, 4889–4896 (2007).
Ikram, M.S. et al. GLI2 is expressed in normal human epidermis and BCC and induces GLI1 expression by binding to its promoter. J. Invest. Dermatol. 122, 1503–1509 (2004).
Acknowledgements
We are indebted to R. Marais (ICR, London), K. Okazaki (Kurume Univ.), M. Muenke (US National Institutes of Health), C. Counter (Duke Univ.), J. Bos (Univ. Utrecht), P. Beachy (Stanford), F. Aberger (Univ. Salzburg), J. Taipale (Karolinska Institutet), M. Löhr (Karolinska Institutet) and J. Bergman (Karolinska Institutet) for kind provision of reagents, plasmids and cell lines, F. de Sauvage and S. Scales at Genentech for the provision of the anti-HH antibody, S. Teglund and V. Jaks for critical comments on the manuscript and E. Tuksammel for technical help and animal caretaking. We are grateful for grant support from the Swedish Cancer Society, the Wenner-Gren-Foundation, the Lars Hiertas Minne Foundation and the Karolinska Institutet to M.L. and from the Swedish Cancer Society, the Swedish Research Council, the US National Institutes of Health National Cancer Institute (Mouse Models of Human Cancers Consortium Grant UO1 CA105491) and Swiss Bridge to R.T. T.S. is a Marie Curie International Incoming Fellow.
Author information
Authors and Affiliations
Contributions
M.L., C.G., M.B. and R.T. designed experiments; M.L., Å.B., T.S., U.T., Q.J., V.F. and C.G. performed experiments; M.L. and R.T. interpreted data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Methods (PDF 1472 kb)
Rights and permissions
About this article
Cite this article
Lauth, M., Bergström, Å., Shimokawa, T. et al. DYRK1B-dependent autocrine-to-paracrine shift of Hedgehog signaling by mutant RAS. Nat Struct Mol Biol 17, 718–725 (2010). https://doi.org/10.1038/nsmb.1833
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1833