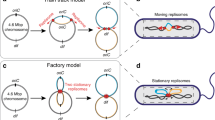
Abstract
In prokaryotes, the transfer of DNA between cellular compartments is essential for the segregation and exchange of genetic material. SpoIIIE and FtsK are AAA+ ATPases responsible for intercompartmental chromosome translocation in bacteria. Despite functional and sequence similarities, these motors were proposed to use drastically different mechanisms: SpoIIIE was suggested to be a unidirectional DNA transporter that exports DNA from the compartment in which it assembles, whereas FtsK was shown to establish translocation directionality by interacting with highly skewed chromosomal sequences. Here we use a combination of single-molecule, bioinformatics and in vivo fluorescence methodologies to study the properties of DNA translocation by SpoIIIE in vitro and in vivo. These data allow us to propose a sequence-directed DNA exporter model that reconciles previously proposed models for SpoIIIE and FtsK, constituting a unified model for directional DNA transport by the SpoIIIE/FtsK family of AAA+ ring ATPases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ryter, A. Morphologic study of the sporulation of Bacillus subtilis. Ann. Inst. Pasteur (Paris) 108, 40–60 (1965).
Thomaides, H.B., Freeman, M., El Karoui, M. & Errington, J. Division site selection protein DivIVA of Bacillus subtilis has a second distinct function in chromosome segregation during sporulation. Genes Dev. 15, 1662–1673 (2001).
Levin, P.A. & Losick, R. Transcription factor Spo0A switches the localization of the cell division protein FtsZ from a medial to a bipolar pattern in Bacillus subtilis. Genes Dev. 10, 478–488 (1996).
Pogliano, J., Sharp, M.D. & Pogliano, K. Partitioning of chromosomal DNA during establishment of cellular asymmetry in Bacillus subtilis. J. Bacteriol. 184, 1743–1749 (2002).
Wu, L.J. & Errington, J. Bacillus subtilis spoIIIE protein required for DNA segregation during asymmetric cell division. Science 264, 572–575 (1994).
Wu, L.J., Lewis, P.J., Allmansberger, R., Hauser, P.M. & Errington, J. A conjugation-like mechanism for prespore chromosome partitioning during sporulation in Bacillus subtilis. Genes Dev. 9, 1316–1326 (1995).
Bath, J., Wu, L.J., Errington, J. & Wang, J.C. Role of Bacillus subtilis SpoIIIE in DNA transport across the mother cell-prespore division septum. Science 290, 995–997 (2000).
Sharpe, M.E. & Errington, J. Postseptational chromosome partitioning in bacteria. Proc. Natl. Acad. Sci. USA 92, 8630–8634 (1995).
Britton, R.A. & Grossman, A.D. Synthetic lethal phenotypes caused by mutations affecting chromosome partitioning in Bacillus subtilis. J. Bacteriol. 181, 5860–5864 (1999).
Lemon, K.P. & Grossman, A.D. Effects of replication termination mutants on chromosome partitioning in Bacillus subtilis. Proc. Natl. Acad. Sci. USA 98, 212–217 (2001).
Liu, G., Draper, G.C. & Donachie, W.D. FtsK is a bifunctional protein involved in cell division and chromosome localization in Escherichia coli. Mol. Microbiol. 29, 893–903 (1998).
Iyer, L.M., Makarova, K.S., Koonin, E.V. & Aravind, L. Comparative genomics of the FtsK-HerA superfamily of pumping ATPases: implications for the origins of chromosome segregation, cell division and viral capsid packaging. Nucleic Acids Res. 32, 5260–5279 (2004).
Capiaux, H., Lesterlin, C., Perals, K., Louarn, J.M. & Cornet, F. A dual role for the FtsK protein in Escherichia coli chromosome segregation. EMBO Rep. 3, 532–536 (2002).
Wu, L.J. & Errington, J. Septal localization of the SpoIIIE chromosome partitioning protein in Bacillus subtilis. EMBO J. 16, 2161–2169 (1997).
Wang, L. & Lutkenhaus, J. FtsK is an essential cell division protein that is localized to the septum and induced as part of the SOS response. Mol. Microbiol. 29, 731–740 (1998).
Yu, X.C., Tran, A.H., Sun, Q. & Margolin, W. Localization of cell division protein FtsK to the Escherichia coli septum and identification of a potential N-terminal targeting domain. J. Bacteriol. 180, 1296–1304 (1998).
Massey, T.H., Mercogliano, C.P., Yates, J., Sherratt, D.J. & Lowe, J. Double-stranded DNA translocation: structure and mechanism of hexameric FtsK. Mol. Cell 23, 457–469 (2006).
Bigot, S. et al. KOPS: DNA motifs that control E. coli chromosome segregation by orienting the FtsK translocase. EMBO J. 24, 3770–3780 (2005).
Levy, O. et al. Identification of oligonucleotide sequences that direct the movement of the Escherichia coli FtsK translocase. Proc. Natl. Acad. Sci. USA 102, 17618–17623 (2005).
Pease, P.J. et al. Sequence-directed DNA translocation by purified FtsK. Science 307, 586–590 (2005).
Sharp, M.D. & Pogliano, K. Role of cell-specific SpoIIIE assembly in polarity of DNA transfer. Science 295, 137–139 (2002).
Sharp, M.D. & Pogliano, K. MinCD-dependent regulation of the polarity of SpoIIIE assembly and DNA transfer. EMBO J. 21, 6267–6274 (2002).
Becker, E.C. & Pogliano, K. Cell-specific SpoIIIE assembly and DNA translocation polarity are dictated by chromosome orientation. Mol. Microbiol. 66, 1066–1079 (2007).
Partridge, S.R. & Errington, J. The importance of morphological events and intercellular interactions in the regulation of prespore-specific gene expression during sporulation in Bacillus subtilis. Mol. Microbiol. 8, 945–955 (1993).
Lewis, P.J., Partridge, S.R. & Errington, J. Sigma factors, asymmetry, and the determination of cell fate in Bacillus subtilis. Proc. Natl. Acad. Sci. USA 91, 3849–3853 (1994).
Pogliano, J. et al. A vital stain for studying membrane dynamics in bacteria: a novel mechanism controlling septation during Bacillus subtilis sporulation. Mol. Microbiol. 31, 1149–1159 (1999).
Ptacin, J.L., Nollmann, M., Bustamante, C. & Cozzarelli, N.R. Identification of the FtsK sequence-recognition domain. Nat. Struct. Mol. Biol. 13, 1023–1025 (2006).
Sivanathan, V. et al. The FtsK γ domain directs oriented DNA translocation by interacting with KOPS. Nat. Struct. Mol. Biol. 13, 965–972 (2006).
Bigot, S., Saleh, O.A., Cornet, F., Allemand, J.F. & Barre, F.X. Oriented loading of FtsK on KOPS. Nat. Struct. Mol. Biol. 13, 1026–1028 (2006).
Griffiths, A.A. & Wake, R.G. Search for additional replication terminators in the Bacillus subtilis 168 chromosome. J. Bacteriol. 179, 3358–3361 (1997).
Sciochetti, S.A., Piggot, P.J. & Blakely, G.W. Identification and characterization of the dif site from Bacillus subtilis. J. Bacteriol. 183, 1058–1068 (2001).
Becker, E. et al. DNA segregation by the bacterial actin AlfA during Bacillus subtilis growth and development. EMBO J. 25, 5919–5931 (2006).
Hendrickson, H. & Lawrence, J.G. Selection for chromosome architecture in bacteria. J. Mol. Evol. 62, 615–629 (2006).
Wu, L.J. & Errington, J. A large dispersed chromosomal region required for chromosome segregation in sporulating cells of Bacillus subtilis. EMBO J. 21, 4001–4011 (2002).
Kohler, J.J. & Schepartz, A. Kinetic studies of FosJunDNA complex formation: DNA binding prior to dimerization. Biochemistry 40, 130–142 (2001).
Burton, B.M., Marquis, K.A., Sullivan, N.L., Rapoport, T.A. & Rudner, D.Z. The ATPase SpoIIIE transports DNA across fused septal membranes during sporulation in Bacillus subtilis. Cell 131, 1301–1312 (2007).
Pettis, G.S. & Cohen, S.N. Unraveling the essential role in conjugation of the Tra protein of Streptomyces lividans plasmid pIJ101. Antonie Van Leeuwenhoek 79, 247–250 (2001).
Bigot, S., Sivanathan, V., Possoz, C., Barre, F.X. & Cornet, F. FtsK, a literate chromosome segregation machine. Mol. Microbiol. 64, 1434–1441 (2007).
Sharpe, M.E. & Errington, J. The Bacillus subtilis soj-spo0J locus is required for a centromere-like function involved in prespore chromosome partitioning. Mol. Microbiol. 21, 501–509 (1996).
Wu, L.J. & Errington, J. RacA and the Soj-Spo0J system combine to effect polar chromosome segregation in sporulating Bacillus subtilis. Mol. Microbiol. 49, 1463–1475 (2003).
Ben-Yehuda, S., Rudner, D.Z. & Losick, R. RacA, a bacterial protein that anchors chromosomes to the cell poles. Science 299, 532–536 (2003).
Ben-Yehuda, S. et al. Defining a centromere-like element in Bacillus subtilis by identifying the binding sites for the chromosome-anchoring protein RacA. Mol. Cell 17, 773–782 (2005).
Rost, B., Yachdav, G. & Liu, J. The PredictProtein server. Nucleic Acids Res. 32, W321–W326 (2004).
McClelland, S.E., Dryden, D.T. & Szczelkun, M.D. Continuous assays for DNA translocation using fluorescent triplex dissociation: application to type I restriction endonucleases. J. Mol. Biol. 348, 895–915 (2005).
Firman, K. & Szczelkun, M.D. Measuring motion on DNA by the type I restriction endonuclease EcoR124I using triplex displacement. EMBO J. 19, 2094–2102 (2000).
Strick, T.R., Allemand, J.F., Bensimon, D. & Croquette, V. Behavior of supercoiled DNA. Biophys. J. 74, 2016–2028 (1998).
Perez, A.R., Abanes-De Mello, A. & Pogliano, K. SpoIIB localizes to active sites of septal biogenesis and spatially regulates septal thinning during engulfment in Bacillus subtilis. J. Bacteriol. 182, 1096–1108 (2000).
Sterlini, J.M. & Mandelstam, J. Commitment to sporulation in Bacillus subtilis and its relationship to development of actinomycin resistance. Biochem. J. 113, 29–37 (1969).
Hoch, J.A. Genetic analysis in Bacillus subtilis. Methods Enzymol. 204, 305–320 (1991).
Acknowledgements
The authors would like to dedicate this work to our friend and colleague Nicholas R. Cozzarelli, who passed away during completion of this research. We thank J. Berger for his continuous advice during this research and N. Crisona for critical reading. This work was supported by National Institutes of Health Grants GM31655 (to N.R.C.) and the Human Frontier Science Program (M.N.).
Author information
Authors and Affiliations
Contributions
J.L.P. and M.N. planned, collected and interpreted single-molecule, biochemical and fluorescence data; E.C.B. performed time-lapse fluorescence studies of SpoIIIE-SK chimera and compartment-specific GFP-tagging experiments; J.L.P., M.N., E.C.B., K.P. and C.B. wrote the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6, Supplementary Tables 1–3, Supplementary Results and Supplementary Methods (PDF 1665 kb)
Supplementary Video 1
DNA is forward translocated into forespore compartment in all events tracked (N=28) and all cells observed (N=243). (MOV 3687 kb)
Supplementary Video 2
Frequently, DNA is initially translocated into the forespore followed by a pause in translocation and subsequent reversal of DNA translocation direction that pumps the chromosome out of the forespore compartment (N=140 out of 349 cells). (MOV 3251 kb)
Rights and permissions
About this article
Cite this article
Ptacin, J., Nollmann, M., Becker, E. et al. Sequence-directed DNA export guides chromosome translocation during sporulation in Bacillus subtilis. Nat Struct Mol Biol 15, 485–493 (2008). https://doi.org/10.1038/nsmb.1412
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1412
This article is cited by
-
vB_BcM_Sam46 and vB_BcM_Sam112, members of a new bacteriophage genus with unusual small terminase structure
Scientific Reports (2021)
-
Replication termination without a replication fork trap
Scientific Reports (2019)
-
Sequence-dependent catalytic regulation of the SpoIIIE motor activity ensures directionality of DNA translocation
Scientific Reports (2018)
-
The fluorescence properties and binding mechanism of SYTOX green, a bright, low photo-damage DNA intercalating agent
European Biophysics Journal (2015)
-
SpoIIIE mechanism of directional translocation involves target search coupled to sequence‐dependent motor stimulation
EMBO reports (2013)