Abstract
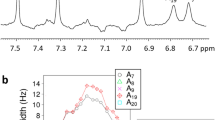
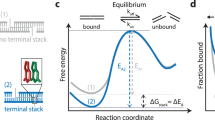
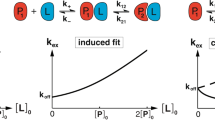
Signal propagation in biological systems occurs through a series of inter- and intramolecular events, the precise pathways of which remain elusive in most cases. With respect to protein–DNA interactions in particular, little is known about the association and dissociation reaction pathways. Here we show that the exchange of amide protons detected by NMR can be used to characterize, at residue level, the mechanism, kinetics and thermodynamics of Lac headpiece (HP) interaction with DNA operators. Specific protein–DNA contacts responsible for the direct readout of the sequence are formed and broken at distinct time scales. Unfolding of the hinge helices triggers protein–DNA dissociation by progressive destabilization of distinct structural units, which is facilitated by the low stability of the protein in the uncomplexed state. Upon DNA binding, a dramatic alteration in the dynamics of the protein is observed, which may be used advantageously by the biological system to switch between functional states. Hydrogen-deuterium exchange can provide an unusually detailed view of the interaction mechanism of a protein–DNA complex and the associated energetics of DNA recognition with residue-level specificity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Freire, E. Proc. Natl. Acad. Sci. USA 96, 10118–10122 (1999).
Dunker, A.K. et al. J. Mol. Graph. Model. 19, 26–59 (2001).
Wright, P.E. & Dyson, H.J. J. Mol. Biol. 293, 321–331 (1999).
Lefstin J.A. & Yamamoto, K.R. Nature 392, 885–888 (1998).
Bell, C.E. & Lewis, M. Curr. Opin. Struct. Biol. 11, 19–25 (2001).
Lewis, M. et al. Science 271, 1247–1254 (1996).
Spronk, C.A.E.M. et al. Structure 7, 1483–1492 (1999).
Spronk, C.A.E.M., Slijper, M., van Boom, J.H., Kaptein, R. & Boelens, R. Nature Struct. Biol. 3, 916–919 (1996).
Bell, C.E. & Lewis, M. Nature Struct. Biol. 7, 209–214 (2000).
Kalodimos, C.G., Folkers, G.E., Boelens, R. & Kaptein, R. Proc. Natl. Acad. Sci. USA 98, 6039–6044 (2001).
Englander, S.W. & Krishna, M.M.G. Nature Struct. Biol. 8, 741–742 (2001).
Englander, S.W. Annu. Rev. Biophys. Biomol. Struct. 29, 213–238 (2000).
Xu, Y., Mayne, L. & Englander, S.W. Nature Struct. Biol. 5, 774–778 (1998).
Chamberlain, A.K., Handel, T.M. & Marqusee, S. Nature Struct. Biol. 3, 782–787 (1996).
Kiefhaber, T. & Baldwin, R.L. Proc. Natl. Acad. Sci. USA 92, 2657–2661 (1995).
Zahn, R., Perrett, S., Stenberg, G. & Fersht, A.R. Science 271, 642–645 (1996).
Parker, M.J., Dempsey, C.E., Hosszu, L.L.P., Waltho, J.P. & Clarke, A.R. Nature Struct. Biol. 5, 194–198 (1998).
Hvidt, A. & Nielsen, S.O. Adv. Protein Chem. 21, 287–386 (1966).
Bai, Y., Milne, J.S., Mayne, L. & Englander, S.W. Proteins 17, 75–86 (1993).
Sivaraman, T., Arrington, C.B. & Robertson, A.D. Nature Struct. Biol. 8, 331–333 (2001).
Sasmor, H.M. & Betz, J.L. Gene 89, 1–6 (1990).
Spolar, R.S. & Record, M.T. Jr. Science 263, 777–784 (1994).
Arrington, C.B. & Robertson, A.D. J. Mol. Biol. 300, 221–232 (2000).
Falcon, C.M. & Matthews, K.S. Biochemistry 39, 11074–11083 (2000).
Wintjens, R. & Rooman, M. J. Mol. Biol. 262, 294–313 (1996).
Ciubotatu, M., Bright, F.V., Ingersoll, C.M. & Koudelka, G.B. J. Mol. Biol. 294, 859–873 (1999).
van Tilborg, M.A. et al. J. Mol. Biol. 301, 947–958 (2000).
Arrington, C.B. & Robertson, A.D. Biochemistry 36, 8686–8691 (1997).
Koradi, R., Billeter, M. & Wüthrich, K. J. Mol. Graph. 14, 52–55 (1996).
Riggs, A D., Bourgeois, S. & Cohn, M. J. Mol. Biol. 53, 401–417 (1970).
Acknowledgements
This work was financially supported by the Netherlands Foundation for Scientific Research (NOW-CW)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalodimos, C., Boelens, R. & Kaptein, R. A residue-specific view of the association and dissociation pathway in protein–DNA recognition. Nat Struct Mol Biol 9, 193–197 (2002). https://doi.org/10.1038/nsb763
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb763