Abstract
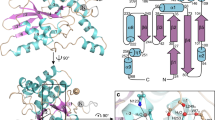
Members of the inhibitor of apoptosis (IAP) family of proteins are able to inhibit cell death following viral infection, during development or in cell lines in vitro. All IAP proteins bear one or more baculoviral IAP repeats (BIRs). Here we describe the solution structure of the third BIR domain from the mammalian IAP homolog B (MIHB /c- IAP-1). The BIR domain has a novel fold that is stabilized by zinc tetrahedrally coordinated by one histidine and three cysteine residues. The structure consists of a series of short α-helices and turns with the zinc packed in an unusually hydrophobic environment created by residues that are highly conserved among all BIRs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Crook, N.E., Clem, R.J. & Miller, L.K. J. Virol. 67, 2168– 2174 (1993).
Duckett, C.S. et al. EMBO J. 15, 2685–2694 (1996).
Hay, B.A., Wassarman, D.A. & Rubin, G.M. Cell 83, 1253– 1262 (1995).
Liston, P. et al. Nature 379, 349–353 (1996).
Uren, A.G., Pakusch, M., Hawkins, C.J., Puls, K.L. & Vaux, D.L. Proc. Natl. Acad. Sci. USA 93, 4974–4978 (1996).
Uren, A.G., Coulson, E.J. & Vaux, D.L. Trends Biochem. Sci. 23, 159 –162 (1998).
Clem, R.J. & Miller, L.K. Mol. Cell. Biol. 14 , 5212–5222 (1994).
Hawkins, C.J., Uren, A.G., Hacker, G., Medcalf, R.L. & Vaux, D.L. Proc. Natl. Acad. Sci. USA 93, 13786–13790 (1996).
Vucic, D., Kaiser, W.J., Harvey, A.J. & Miller, L.K. Proc. Natl. Acad. Sci. USA 94, 10183– 10188 (1997).
Ambrosini, G., Adida, C. & Altieri, D.C. Nature Med. 3, 917– 921 (1997).
Roy, N., Deveraux, Q.L., Takahashi, R., Salvesen, G.S. & Reed, J.C. EMBO J. 16, 6914–6925 (1997).
Deveraux, Q.L., Takahashi, R., Salvesen, G.S. & Reed, J.C. Nature 388, 300–304 ( 1997).
Manji, G.A., Hozak, R.R., Lacount, D.J. & Friesen, P.D. J. Virol. 71, 4509–4516 (1997).
Deveraux, Q.L. et al. EMBO J. 17, 2215– 2223 (1998).
Rothe, M., Pan, M.G., Henzel, W.J., Ayres, T.M. & Goeddel, D.V. Cell 83, 1243– 1252 (1995).
Vucic, D., Kaiser, W.J. & Miller, L.K. J. Biol. Chem. 273, 33915– 33921 (1998).
Birnbaum, M.J., Clem, R.J. & Miller, L.K. J. Virol. 68, 2521– 2528 (1994).
Mackay, J.P. & Crossley, M. Trends Biochem. Sci. 23, 1–4 (1998).
Berg, J.M. & Shi, Y.G. Science 271, 1081–1085 (1996).
Holm, L. & Sander, C. J. Mol. Biol. 233, 123–138 (1993).
Karlin, S. & Zhu, Z.Y. Proc. Natl. Acad. Sci. USA 94, 14231–14236 (1997).
Vucic, D., Kaiser, W.J. & Miller, L.K. Mol. Cell. Biol. 18, 3300– 3309 (1998).
McCarthy, J.V. & Dixit, V.M. J. Biol. Chem. 273, 24009–24015 ( 1998).
Eberstadt, M. et al. Nature 392, 941–945 (1998).
Chou, J.J., Matsuo, H., Duan, H. & Wagner, G. Cell 94, 171–180 (1998).
Huang, B.H., Eberstadt, M., Olejniczak, E.T., Meadows, R.P. & Fesik, S.W. Nature 384, 638–641 (1996).
Clore, G.M. & Gronenborn, A.M. Nature Struct. Biol. (Suppl.) 4, 849–853 ( 1997).
Luginbühl, P., Szyperski, T. & Wüthrich, K. J. Magn. Reson. Series B 109, 229–233 (1995).
Ludvigsen, S. & Poulsen, F.M. J. Biomol. NMR 2, 227–233 (1992).
Güntert, P., Mumenthaler, C. & Wüthrich, K. J. Mol. Biol. 273, 283– 298 (1997).
Brünger, A.T. X–PLOR version 3.851. A system for X–ray crystallography and NMR. (Yale University Press, New Haven, Connecticut; 1996 ).
Hinds, M.G., Maurer, T., Zhang, J.G., Nicola, N.A. & Norton, R.S. J. Biol. Chem. 273, 13738– 13745 (1998).
Mackerell, A.D. et al. J. Phys. Chem. B 102, 3586– 3616 (1998).
Laskowski, R.A., Rullmann, J.A.C., MacArthur, M.W., Kaptein, R. & Thornton, J.M. J. Biomol. NMR 8, 477–486 (1996).
Koradi, R., Billeter, M. & Wüthrich, K. J. Mol. Graphics 14, 51– 55 (1996).
Acknowledgements
We wish to thank L. Miller for providing a preprint of a mutagenesis paper. This work was supported in part by a grant from the Australian Government (National Health and Medical Research Council) to D.L.V., the CRC for Cellular Growth Factors and grants to C.L.D from FRST (New Zealand) and the Marsden Fund (New Zealand).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hinds, M., Norton, R., Vaux, D. et al. Solution structure of a baculoviral inhibitor of apoptosis (IAP) repeat . Nat Struct Mol Biol 6, 648–651 (1999). https://doi.org/10.1038/10701
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/10701
This article is cited by
-
XIAP as a multifaceted molecule in Cellular Signaling
Apoptosis (2022)
-
Genome sequence of Perigonia lusca single nucleopolyhedrovirus: insights into the evolution of a nucleotide metabolism enzyme in the family Baculoviridae
Scientific Reports (2016)
-
X-linked Inhibitor of Apoptosis Protein negatively regulates neuronal differentiation through interaction with cRAF and Trk
Scientific Reports (2013)
-
CLL cells are resistant to smac mimetics because of an inability to form a ripoptosome complex
Cell Death & Disease (2013)
-
Targeting IAP proteins for therapeutic intervention in cancer
Nature Reviews Drug Discovery (2012)