Abstract
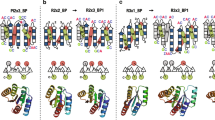
Two new NMR structures of WW domains, the mouse formin binding protein and a putative 84.5 kDa protein from Saccharomyces cerevisiae, show that this domain, only 35 amino acids in length, defines the smallest monomeric triple-stranded antiparallel β-sheet protein domain that is stable in the absence of disulfide bonds, tightly bound ions or ligands. The structural roles of conserved residues have been studied using site-directed mutagenesis of both wild type domains. Crucial interactions responsible for the stability of the WW structure have been identified. Based on a network of highly conserved long range interactions across the β-sheet structure that supports the WW fold and on a systematic analysis of conserved residues in the WW family, we have designed a folded prototype WW sequence.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bork, P., Schultz, J. & Ponting, C.P. Trends Biochem. Sci. 22, 296–298 (1997).
Doolittle, R. Annu. Rev. Biochem. 64, 287–314 (1995).
Pawson, T. Nature 373, 573–580 (1995).
Chen, H.I. & Sudol, M. Proc. Natl. Acad. Sci. USA 92, 7819–7823 (1995).
Sudol, M. et al. J. Biol. Chem. 270, 14733–14741 (1995).
Sudol, M., Chen, H.I., Bougeret, C., Einbond, A. & Boork, P. FEBS Lett. 369, 67–71 (1995).
Macias, M.J. et al. Nature 382, 646–649 (1996).
Ranganathan, R., Lu, K.P., Hunter, T. & Noel, J.P. Cell 89, 875–886 (1997).
Chan, D.C., Bedford, M.T. & Leder, P. EMBO J. 15, 1045–1054 (1996).
Thompson, J.D., Higgins, D.G. & Gibson, T.J. Nucleic Acids Res. 22, 4673–4680 (1994).
Fasman, G.D. Circular dichroism and the conformational analysis of biomolecules. (Plenum Press, New York and London; 1996).
Viguera, A.R., Martinez, J.C., Filimonov, V.V., Mateo, P.L. & Serrano, L. Biochemistry 33, 2142–2150 (1994).
Koepf, E.K., Petrassi, H.M., Sudol, M. & Kelly, J.W. Protein Sci. 8, 841–853 (1999).
Gronenborn, A.M. & Clore, G.M. Science 263, 536 (1994).
Dill, K., Fiebig, K.M. & Chan, H.S. Proc. Natl. Acad. Sci. USA 90, 1942–1946 (1993).
Jeener, J., Meier, B.H., Bachmann, P. & Ernst, R.R. J. Chem. Phys. 71, 4546–4553 (1979).
Braunschweiler, L. & Ernst, R.R. J. Magn. Reson. 53, 521–529 (1983).
Wüthrich, K. NMR of proteins and nucleic acids (Wiley Press, New York; 1986).
Eccles, C., Güntert, P., Billeter, M. & Wüthrich, K. J. Biomol. NMR 1, 111–130 (1991).
Nilges, M. Curr. Opin. Struct. Biol. 6, 617–23 (1996).
Nilges, M., Macias, M.J., O'Donoghue, S.I. & Oschkinat, H. J. Mol. Biol. 269, 408–422 (1997).
Laskowski, R.A., Rullman, J.A., MacArthur, M.W., Kaptein, R. & Thornton, J.M. J. Biomol. NMR. 8, 477–486 (1996).
Vriend, G. J. Mol. Graph. 8, 52–56 (1990).
Koradi, R., Billeter, M. & Wüthrich, K. J. Mol. Graph. 14, 51–55 (1996).
Koradi, R., Billeter, M. & Wüthrich, K. J. Mol. Graph. 14, 29–32 (1996).
Acknowledgements
We thank P. Chambon and the IGBMC-LGME-U.184-ULP for the mouse embryonic day 10 cDNA library, J. Ashurst and S. Moulton for sample preparation, M. Hyvönen for the pGAT2 expression vector and Bruker Analytik GMBH Karlsruhe for measurement time on the DRX-800 MHz spectrometer. The authors are very grateful to J. Castresana, H. Domingues, J. Dixon and R. Wade for useful comments. C.C. was supported by a grant from the Spanish Ministerio de Educacion y Cultura.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Macias, M., Gervais, V., Civera, C. et al. Structural analysis of WW domains and design of a WW prototype. Nat Struct Mol Biol 7, 375–379 (2000). https://doi.org/10.1038/75144
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/75144
This article is cited by
-
SMURF1, a promoter of tumor cell progression?
Cancer Gene Therapy (2021)
-
How Useful can the Voigt Profile be in Protein Folding Processes?
The Protein Journal (2021)
-
Changes in the folding landscape of the WW domain provide a molecular mechanism for an inherited genetic syndrome
Scientific Reports (2016)
-
A Study of the Influence of Charged Residues on β-Hairpin Formation by Nuclear Magnetic Resonance and Molecular Dynamics
The Protein Journal (2014)
-
Phospho-Ser/Thr-binding domains: navigating the cell cycle and DNA damage response
Nature Reviews Molecular Cell Biology (2013)