Abstract
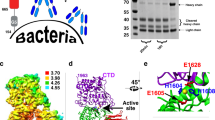
The antigen 85 (ag85) complex, composed of three proteins (ag85A, B and C), is a major protein component of the Mycobacterium tuberculosis cell wall. Each protein possesses a mycolyltransferase activity required for the biogenesis of trehalose dimycolate (cord factor), a dominant structure necessary for maintaining cell wall integrity. The crystal structure of recombinant ag85C from M. tuberculosis, refined to a resolution of 1.5 Å, reveals an α/β-hydrolase polypeptide fold, and a catalytic triad formed by Ser 124, Glu 228 and His 260. ag85C complexed with a covalent inhibitor implicates residues Leu 40 and Met 125 as components of the oxyanion hole. A hydrophobic pocket and tunnel extending 21 Å into the core of the protein indicates the location of a probable trehalose monomycolate binding site. Also, a large region of conserved surface residues among ag85A, B and C is a probable site for the interaction of ag85 proteins with human fibronectin.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fukui, Y., Hirai, T., Uchida, T. & Yoneda, M. Extracellular proteins of tubercle bacilli. IV. Alpha and beta antigens as major extracellular protein products and as cellular components of a strain (H37Rv) of Mycobacterium tuberculosis. Biken J. 8, 189– 199 (1965).
Belisle, J.T. et al. Role of the major antigen of Mycobacterium tuberculosis in cell wall biogenesis. Science 276, 1420–1422 (1997).
Brennan, P.J. & Nikaido, H. The envelope of mycobacteria. Annu. Rev. Biochem. 96, 29–63 (1995).
Jackson, M. et al. Inactivation of the antigen 85C gene profoundly affects the mycolate content and alters the permeability of the Mycobacterium tuberculosis cell envelope. Mol. Microbiol. 31, 1573–1587 (1999).
Robitzek, E.H. & Selikoff, I.J. Hydrazine derivatives of isonicotinic acid (Rimifon, Marsilid) in the treatment of active progressive caseous–pneumonic tuberculosis. Am. Rev. Tuberc. Pulm. Dis. 65, 765 (1952).
Blanchard, J.S. Molecular mechanisms of drug resistance in Mycobacterium tuberculosis. Annu. Rev. Biochem. 65, 215– 239 (1996).
Banerjee, A. et al. inhA, a gene encoding a target for isoniazid and ethionamide in Mycobacterium tuberculosis. Science 263, 227–230 (1994).
Takayama, K. & Kilburn, J.O. Inhibition of synthesis of arabinogalactan in Mycobacterium smegmatis. Antimicrob Agents Chemother. 33, 1493–1499 (1989).
Silva, C.L., Ekizlerian, S.M. & Fazioli, R. Role of cord factor in the modulation of infection caused by mycobacteria. Am. J. Pathol. 118, 238 –247 (1985).
Schrag, J.D. & Cygler, M. Lipases and α/β hydrolase fold. Methods Enzymol. 284, 85– 107 (1997).
Powers, J.C. et al. Proteases—structures, mechanism and inhibitors. Agents Actions Suppl. 42, 3–18 (1993).
Kraut, J. Serine proteases: structure and mechanism of catalysis. Annu. Rev. Biochem. 46, 331–358 ( 1977).
Egloff, M.P. et al. The 2.46 Å resolution structure of the pancreatic lipase–colipase complex inhibited by a C11 alkyl phosphonate. Biochemistry 34, 2751–2762 (1995).
Uppenberg, J. et al. Crystallographic and molecular-modeling studies of lipase B from Candida antarctica reveal a stereospecificity pocket for secondary alcohols. Biochemistry 31, 16838– 16851 (1995).
Bullock, T.L., Breddam, K. & Remington, S.J. Peptide aldehyde complexes with wheat serine carboxypeptidase II: implications for the catalytic mechanism and substrate specificity. J. Mol. Biol. 255, 714–725 (1996).
Ransac, S. et al. Covalent inactivation of lipases. Methods Enzymol. 286, 190–231 ( 1997).
Leuveling Tjeenk, M. et al. Inactivation of Staphylococcus hyicus lipase by hexadecylsulfonyl fluoride: evidence for an active site serine. Protein Eng. 7, 579 (1994).
Maylie, M.F., Charles, M. & Desnuelle P. Action of organophosphates and sulfonyl halides on porcine pancreatic lipase. Biochim. Biophys. Acta 276, 162–175 (1972).
Longhi, S., Czjzek, M., Lamzin, V., Nicolas, A. & Cambillau C. Atomic resolution (1.0 Å) crystal structure of Fusarium solani cutinase: stereochemical analysis. J. Mol. Biol. 268, 779–799 ( 1997).
Martinez, C. Cutinase, a lipolytic enzyme with a preformed oxyanion hole. Biochemistry 33, 83–89 ( 1994).
Abou-Zeid, C. et al. Characterization of fibronectin-binding antigens released by Mycobacterium tuberculosis and Mycobacterium bovis BCG. Infect. Immun. 56, 3046–3051 (1988).
Schlesinger L.S. & Horowitz, M.A. Phagocytosis of Mycobacterium leprae by human monocyte-derived macrophages is mediated by complement receptors CR1 (CD35), CR3 (CD11b/CD18), and CR4 (CD11c/CD18) and IFN-γ activation inhibits complement receptor function and phagocytosis of this bacterium. J. Immunol. 147, 1983 –1994 (1991).
Schorey, J.S., Carroll, M.C. & Brown, E.J. A macrophage invasion mechanism of pathogenic mycobacteria . Science 277, 1091–1093 (1997).
Naito, M., Ohara, N., Matsumoto, S. & Yamada, T. The novel fibronectin-binding motif and key residues of mycobacteria. J. Biol. Chem. 273, 2905 (1998).
De Bruyn, J., Bosmans, R., Nyabenda, J. & Van Vooren, J.P. Effect of zinc deficiency on the appearance of two immunodominant protein antigens (32 kDa and 65 kDa) in culture filtrates of mycobacteria. J. Gen. 135, 79–84 ( 1989).
Fukui, Y., Hirai, T., Uchida, T. & Yoneda, M. Extracellular proteins of tubercle bacilli. IV. Alpha and beta antigens as major extracellular protein products and as cellular components of a strain (H37Rv) of Mycobacterium tuberculosis. Biken J. 8, 189– 199 (1965).
Wiker, G.H. & Harboe, M. The antigen 85 complex: a major secretion product of Mycobacterium tuberculosis. Microbiol. Rev. 56, 648–661 (1992).
Roche, P.W., Peake, P.W., Billman-Jacobe, H., Doran, T. & Britton, W.J. T-Cell determinants and antibody binding sites on the major mycobacterial secretory protein MPB59 of Mycobacterium bovis. Infect. Immun. 62, 5319– 5326 (1994).
Horwitz, M.A., Lee, B.W., Dillon, B.J. & Harth, G. Protective immunity against tuberculosis induced by vaccination with major extracellular proteins of Mycobacterium tuberculosis. Proc. Natl. Acad. Sci. USA 92, 1530–1534 ( 1995).
Orme, I.M. Characteristics and specificity of acquired immunologic memory to Mycobacterium tuberculosis infection. J. Immunol. 140, 3589–3593 (1988).
Samanich, K.M. et al. Delineation of human antibody responses to culture filtrate antigens of Mycobacterium tuberculosis. J. Infect. Dis. 178, 1534–1538 ( 1998).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 27, 307–326 ( 1997).
Collaborative Computational Project, Number 4. CCP4 Suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994).
Furey, W. & Swaminathan, S. PHASES. Am. Crystallogr. Assoc. Annu. Mtg. Program. Abstr. 18, 73 (1990).
Brünger, A.T. et al. Crystallography & NMR system (version 0.9): a new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 ( 1998).
de La Fortelle, E. & Bricogne, G. Maximum-likelihood heavy atom parameter refinement for the multiple isomorphous replacement and multiwavelength anomalous diffraction methods. Methods Enzymol. 276, 472–494 ( 1997).
Cowtan, K.D. & P. Main, P. Phase combination and cross validation in iterated density-modification calculations. Acta Crystallogr. D 52, 43–48 ( 1996).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Navaza, J. AMoRe: an Automated Package for Molecular Replacement. Acta Crystallogr. A 50, 157–163 ( 1994).
Evans, S.V. Setor: hardware lighted three-dimensional solid model representations of macromolecules . J. Mol. Graph. 11, 134– 138 (1993).
Guex, N. & Peitsch, M.C. SWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modeling. Electrophoresis 18, 2714–2723 ( 1997).
Amundsen, S. et al. _X-POV-Team POV-Ray: persistence of vision ray-tracer. http://www.povray.org (1997).
Christopher, J.A. SPOCK: the structural properties observation and calculation kit (program manual). (The Center for Macromolecular Design, Texas A&M University, College Station, Texas; 1998).
Higgins, D.G., Bleasby, A.J. & Fuchs, R. CLUSTAL V: improved software for multiple sequence alignment . Comput. Applic. Biosci. 8, 189– 191 (1992).
Acknowledgements
Financial support was provided by the Robert A. Welch Foundation and the NIH. We would like to thank the staff of the SBC-CAT at the APS at Argonne National laboratory, specifically, Frank Rotella for all of his help with data collection.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ronning, D., Klabunde, T., Besra, G. et al. Crystal structure of the secreted form of antigen 85C reveals potential targets for mycobacterial drugs and vaccines. Nat Struct Mol Biol 7, 141–146 (2000). https://doi.org/10.1038/72413
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/72413
This article is cited by
-
Distribution of sigma factors delineates segregation of virulent and avirulent Mycobacterium
Archives of Microbiology (2021)
-
Functional insights from a comparative study on the dynamics of Antigen85 proteins and MPT51 from Mycobacterium tuberculosis
Journal of Molecular Modeling (2015)
-
Mechanism of inhibition of Mycobacterium tuberculosis antigen 85 by ebselen
Nature Communications (2013)
-
Uptake of unnatural trehalose analogs as a reporter for Mycobacterium tuberculosis
Nature Chemical Biology (2011)
-
Antigen 85C-mediated acyl-transfer between synthetic acyl donors and fragments of the arabinan
Glycoconjugate Journal (2009)