Key Points
-
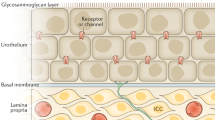
Mitomycin C (MMC) is a chemotherapeutic agent commonly used for intravesical treatment of non-muscle-invasive bladder cancer (NMIBC)
-
Physiological and pharmacological approaches, such as emptying of the bladder and urine alkalization, can be taken to optimize the pharmacokinetics and, therefore, increase the efficacy of MMC therapy
-
Newer approaches such as electromotive drug administration, chemohyperthermia, maintenance MMC-based regimens and combination of MMC with other intravesical agents have been shown to increase efficacy of therapy using this agent
-
Combining MMC with novel agents targeting treatment resistance mechanisms, such as fibroblast growth factor signalling, histone deacetylases and heat shock proteins, might improve outcomes in patients with NMIBC
-
Further study of mechanisms of resistance to MMC is a promising avenue of investigation that might lead to major advances in intravesical therapy
-
As we move towards precision medicine, the goal should be to pre-select patients who will benefit from MMC therapy and avoid or alter the treatment of predicted nonresponders
Abstract
Nearly three-quarters of all newly diagnosed urothelial cancers are non-muscle-invasive bladder cancers (NMIBCs). Although bladder-preserving surgery can be used to treat NMIBC, the rate of recurrence remains high. Intravesical chemotherapy has been shown to reduce the rate of NMIBC recurrence, and mitomycin C (MMC) has become the most commonly used intravesical cytotoxic agent. Despite the popularity of this agent in the treatment of NMIBCs, many questions regarding the optimal approach to MMC therapy remain unanswered. Strategies to enhance delivery of MMC have been well studied and multiple measures are recommended for implementation in routine clinical practice. In addition, less widely investigated techniques, such as hyperthermia and electromotive drug administration, have been shown to increase the efficacy of MMC therapy. Nevertheless, even when the current 'optimal' approaches to MMC administration are used, a large proportion of NMIBCs recur. This apparent treatment resistance might be overcome by combination of MMC with other agents that have different mechanisms of action and are unlikely to have cross-resistance. Study of the mechanisms of resistance is, therefore, important to identify key pathways underlying this phenomenon, which could be rationally targeted using specific combinations of drugs. Knowledge of these mechanisms might also reveal markers of responsiveness to therapy that could be used for patient selection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Tolley, D. A. et al. The effect of intravesical mitomycin C on recurrence of newly diagnosed superficial bladder cancer: a further report with 7 years of follow up. J. Urol. 155, 1233–1238 (1996).
Sylvester, R. J., Oosterlinck, W. & van der Meijden, A. P. A single immediate postoperative instillation of chemotherapy decreases the risk of recurrence in patients with stage Ta–T1 bladder cancer: a meta-analysis of published results of randomized clinical trials. J. Urol. 171, 2186–2190, quiz 2435 (2004).
Dalton, J. T., Wientjes, M. G., Badalament, R. A., Drago, J. R. & Au, J. L. Pharmacokinetics of intravesical mitomycin C in superficial bladder cancer patients. Cancer Res. 51, 5144–5152 (1991).
Maeda, T., Kikuchi, E., Matsumoto, K., Miyajima, A. & Oya, M. Urinary pH is highly associated with tumor recurrence during intravesical mitomycin C therapy for nonmuscle invasive bladder tumor. J. Urol. 185, 802–806 (2011).
Ersoy, H. et al. Single early instillation of mitomycin C and urinary alkalinization in low-risk non-muscle-invasive bladder cancer: a preliminary study. Drug Des. Devel. Ther. 7, 1–6 (2013).
Wientjes, M. G., Badalament, R. A. & Au, J. L. Use of pharmacologic data and computer simulations to design an efficacy trial of intravesical mitomycin C therapy for superficial bladder cancer. Cancer Chemother. Pharmacol. 32, 255–262 (1993).
Au, J. L. et al. Methods to improve efficacy of intravesical mitomycin C: results of a randomized phase III trial. J. Natl Cancer Inst. 93, 597–604 (2001).
van der Heijden, A. G., Verhaegh, G., Jansen, C. F., Schalken, J. A. & Witjes, J. A. Effect of hyperthermia on the cytotoxicity of 4 chemotherapeutic agents currently used for the treatment of transitional cell carcinoma of the bladder: an in vitro study. J. Urol. 173, 1375–1380 (2005).
Paroni, R. et al. Effect of local hyperthermia of the bladder on mitomycin C pharmacokinetics during intravesical chemotherapy for the treatment of superficial transitional cell carcinoma. Br. J. Clin. Pharmacol. 52, 273–278 (2001).
Synergo®—For the treatment of Non-muscle Invasive Bladder Cancer. The technology [online], (2014).
Colombo, R. et al. Multicentric study comparing intravesical chemotherapy alone and with local microwave hyperthermia for prophylaxis of recurrence of superficial transitional cell carcinoma. J. Clin. Oncol. 21, 4270–4276 (2003).
Colombo, R., Salonia, A., Leib, Z., Pavone-Macaluso, M. & Engelstein, D. Long-term outcomes of a randomized controlled trial comparing thermochemotherapy with mitomycin-C alone as adjuvant treatment for non-muscle-invasive bladder cancer (NMIBC). BJU Int. 107, 912–918 (2011).
Lammers, R. J. et al. The role of a combined regimen with intravesical chemotherapy and hyperthermia in the management of non-muscle-invasive bladder cancer: a systematic review. Eur. Urol. 60, 81–93 (2011).
ISRCTN Register. HYMN: a trial comparing hyperthermia and mitomycin chemotherapy with a second BCG treatment, or other standard treatment, for bladder cancer that has come back [online], (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2010).
Di Stasi, S. M. et al. Electromotive versus passive diffusion of mitomycin C into human bladder wall: concentration-depth profiles studies. Cancer Res. 59, 4912–4918 (1999).
Di Stasi, S. M. et al. Intravesical electromotive mitomycin C versus passive transport mitomycin C for high risk superficial bladder cancer: a prospective randomized study. J. Urol. 170, 777–782 (2003).
Di Stasi, S. M. et al. Sequential BCG and electromotive mitomycin versus BCG alone for high-risk superficial bladder cancer: a randomised controlled trial. Lancet Oncol. 7, 43–51 (2006).
Kaasinen, E. et al. Alternating mitomycin C and BCG instillations versus BCG alone in treatment of carcinoma in situ of the urinary bladder: a nordic study. Eur. Urol. 43, 637–645 (2003).
Rintala, E., Jauhiainen, K., Kaasinen, E., Nurmi, M. & Alfthan, O. Alternating mitomycin C and bacillus Calmette-Guerin instillation prophylaxis for recurrent papillary (stages Ta to T1) superficial bladder cancer. J. Urol. 156, 56–59; discussion 59–60 (1996).
Di Stasi, S. M. et al. Electromotive instillation of mitomycin immediately before transurethral resection for patients with primary urothelial non-muscle invasive bladder cancer: a randomised controlled trial. Lancet Oncol. 12, 871–879 (2011).
Colombo, R. et al. Neoadjuvant combined microwave induced local hyperthermia and topical chemotherapy versus chemotherapy alone for superficial bladder cancer. J. Urol. 155, 1227–1232 (1996).
Colombo, R. et al. Neoadjuvant short-term intensive intravesical mitomycin C regimen compared with weekly schedule for low-grade recurrent non-muscle-invasive bladder cancer: preliminary results of a randomised phase 2 study. Eur. Urol. 62, 797–802 (2012).
Dominguez-Escrig, J. L., Kelly, J. D., Neal, D. E., King, S. M. & Davies, B. R. Evaluation of the therapeutic potential of the epidermal growth factor receptor tyrosine kinase inhibitor gefitinib in preclinical models of bladder cancer. Clin. Cancer Res. 10, 4874–4884 (2004).
Pritchett, T. R., Wang, J. K. & Jones, P. A. Mesenchymal-epithelial interactions between normal and transformed human bladder cells. Cancer Res. 49, 2750–2754 (1989).
Bohle, A. & Bock, P. R. Intravesical bacille Calmette-Guerin versus mitomycin C in superficial bladder cancer: formal meta-analysis of comparative studies on tumor progression. Urology 63, 682–686; discussion 686–687 (2004).
Malmstrom, P. U. et al. An individual patient data meta-analysis of the long-term outcome of randomised studies comparing intravesical mitomycin C versus bacillus Calmette-Guerin for non-muscle-invasive bladder cancer. Eur. Urol. 56, 247–256 (2009).
Dalbagni, G. Is intravesical bacillus Calmette-Guerin better than mitomycin for intermediate-risk bladder cancer? Eur. Urol. 56, 257–258; discussion 258–259 (2009).
Oddens, J. et al. Final results of an EORTC-GU cancers group randomized study of maintenance bacillus Calmette-Guerin in intermediate- and high-risk Ta, T1 papillary carcinoma of the urinary bladder: one-third dose versus full dose and 1 year versus 3 years of maintenance. Eur. Urol. 63, 462–472 (2013).
Lamm, D. L. et al. Maintenance bacillus Calmette-Guerin immunotherapy for recurrent TA, T1 and carcinoma in situ transitional cell carcinoma of the bladder: a randomized Southwest Oncology Group Study. J. Urol. 163, 1124–1129 (2000).
Krege, S., Giani, G., Meyer, R., Otto, T. & Rubben, H. A randomized multicenter trial of adjuvant therapy in superficial bladder cancer: transurethral resection only versus transurethral resection plus mitomycin C versus transurethral resection plus bacillus Calmette-Guerin. J. Urol. 156, 962–966 (1996).
Malmstrom, P. U. et al. 5-year followup of a randomized prospective study comparing mitomycin C and bacillus Calmette-Guerin in patients with superficial bladder carcinoma. J. Urol. 161, 1124–1127 (1999).
Gardmark, T., Jahnson, S., Wahlquist, R., Wijkstrom, H. & Malmstrom, P. U. Analysis of progression and survival after 10 years of a randomized prospective study comparing mitomycin-C and bacillus Calmette-Guerin in patients with high-risk bladder cancer. BJU Int. 99, 817–820 (2007).
Friedrich, M. G., Pichlmeier, U., Schwaibold, H., Conrad, S. & Huland, H. Long-term intravesical adjuvant chemotherapy further reduces recurrence rate compared with short-term intravesical chemotherapy and short-term therapy with Bacillus Calmette-Guerin (BCG) in patients with non-muscle-invasive bladder carcinoma. Eur. Urol. 52, 1123–1129 (2007).
Bouffioux, C. et al. Intravesical adjuvant chemotherapy for superficial transitional cell bladder carcinoma: results of 2 European Organization for Research and Treatment of Cancer randomized trials with mitomycin C and doxorubicin comparing early versus delayed instillations and short-term versus long-term treatment. J. Urol. 153, 934–941 (1995).
Badalato, G. M., Hruby, G., Razmjoo, M. & McKiernan, J. M. Maximizing intravesical therapy options: is there an advantage to the administration of perioperative mitomycin C prior to an induction course of BCG? Can. J. Urol. 18, 5890–5895 (2011).
Rajala, P. et al. Cytostatic effect of different strains of Bacillus Calmette-Guerin on human bladder cancer cells in vitro alone and in combination with mitomycin C and interferon-alpha. Urol. Res. 20, 215–217 (1992).
Matsushima, M. et al. Enhanced antitumor effect of combination intravesical mitomycin C and bacillus Calmette-Guerin therapy in an orthotopic bladder cancer model. Oncol. Lett. 2, 13–19 (2011).
Witjes, J. A., Caris, C. T., Mungan, N. A., Debruyne, F. M. & Witjes, W. P. Results of a randomized phase III trial of sequential intravesical therapy with mitomycin C and bacillus Calmette-Guerin versus mitomycin C alone in patients with superficial bladder cancer. J. Urol. 160, 1668–1671; discussion 1671–1672 (1998).
Oosterlinck, W. et al. Sequential intravesical chemoimmunotherapy with mitomycin C and bacillus Calmette-Guerin and with bacillus Calmette-Guerin alone in patients with carcinoma in situ of the urinary bladder: results of an EORTC genito-urinary group randomized phase 2 trial (30993). Eur. Urol. 59, 438–446 (2011).
Rintala, E. et al. Alternating mitomycin C and bacillus Calmette-Guerin instillation therapy for carcinoma in situ of the bladder. J. Urol. 154, 2050–2053 (1995).
Delto, J. C., Kobayashi, T., Benson, M., McKiernan, J. & Abate-Shen, C. Preclinical analyses of intravesical chemotherapy for prevention of bladder cancer progression. Oncotarget 4, 269–276 (2013).
Maymi, J. L., Saltsgaver, N. & O'Donnell, M. A. Intravesical sequential gemcitabine- mitomycin chemotherapy as salvage treatment for patients with refractory superficial bladder cancer [abstract 840]. J. Urol. 175 (Suppl.), 271 (2006).
Lightfoot, A. J. et al. Multi-institutional analysis of sequential intravesical gemcitabine and mitomycin C chemotherapy for non-muscle invasive bladder cancer. Urol. Oncol. 32, 35. e15–e19 (2014).
Breyer, B. N., Whitson, J. M., Carroll, P. R. & Konety, B. R. Sequential intravesical gemcitabine and mitomycin C chemotherapy regimen in patients with non-muscle invasive bladder cancer. Urol. Oncol. 28, 510–514 (2010).
Chen, C. H. et al. A cocktail regimen of intravesical mitomycin-C, doxorubicin, and cisplatin (MDP) for non-muscle-invasive bladder cancer. Urol. Oncol. 30, 421–427 (2012).
Mullerad, M. et al. Herpes simplex virus based gene therapy enhances the efficacy of mitomycin C for the treatment of human bladder transitional cell carcinoma. J. Urol. 174, 741–746 (2005).
Deb, A. A. et al. Potentiation of mitomycin C tumoricidal activity for transitional cell carcinoma by histone deacetylase inhibitors in vitro. J. Urol. 186, 2426–2433 (2011).
Villalona-Calero, M. A. et al. Phase I study of low-dose suramin as a chemosensitizer in patients with advanced non-small cell lung cancer. Clin. Cancer Res. 9, 3303–3311 (2003).
Xin, Y. et al. Low dose suramin as a chemosensitizer of bladder cancer to mitomycin C. J. Urol. 174, 322–327 (2005).
Lamont, F. R. et al. Small molecule FGF receptor inhibitors block FGFR-dependent urothelial carcinoma growth in vitro and in vivo. Br. J. Cancer 104, 75–82 (2011).
Gust, K. M. et al. Fibroblast growth factor receptor 3 is a rational therapeutic target in bladder cancer. Mol. Cancer Ther. 12, 1245–1254 (2013).
Hartl, F. U. & Hayer-Hartl, M. Converging concepts of protein folding in vitro and in vivo. Nat. Struct. Mol. Biol. 16, 574–581 (2009).
Calderwood, S. K., Khaleque, M. A., Sawyer, D. B. & Ciocca, D. R. Heat shock proteins in cancer: chaperones of tumorigenesis. Trends Biochem. Sci. 31, 164–172 (2006).
Parsell, D. A. & Lindquist, S. The function of heat-shock proteins in stress tolerance: degradation and reactivation of damaged proteins. Annu. Rev. Genet. 27, 437–496 (1993).
Zoubeidi, A. & Gleave, M. Small heat shock proteins in cancer therapy and prognosis. Int. J. Biochem. Cell Biol. 44, 1646–1656 (2012).
Lebret, T. et al. Heat shock proteins HSP27, HSP60, HSP70, and HSP90: expression in bladder carcinoma. Cancer 98, 970–977 (2003).
Kassem, H., Sangar, V., Cowan, R., Clarke, N. & Margison, G. P. A potential role of heat shock proteins and nicotinamide N-methyl transferase in predicting response to radiation in bladder cancer. Int. J. Cancer 101, 454–460 (2002).
Kamada, M. et al. HSP27 knockdown using nucleotide-based therapies inhibit tumor growth and enhance chemotherapy in human bladder cancer cells. Mol. Cancer Ther. 6, 299–308 (2007).
Richards, E., Begum, T. & Masters, J. Thermotolerance and sensitivity of human cancer cells to cisplatin and doxorubicin. Int. J. Oncol. 8, 1265–1271 (1996).
Behnsawy, H. M., Miyake, H., Kusuda, Y. & Fujisawa, M. Small interfering RNA targeting heat shock protein 70 enhances chemosensitivity in human bladder cancer cells. Urol. Oncol. 31, 843–848 (2013).
He, L. F. et al. Enhanced sensitivity to mitomycin C by abating heat shock protein 70 expression in human bladder cancer cell line of BIU-87. Chin. Med. J. (Engl.) 118, 1965–1972 (2005).
Hadaschik, B. A. et al. Intravesically administered antisense oligonucleotides targeting heat-shock protein-27 inhibit the growth of non-muscle-invasive bladder cancer. BJU Int. 102, 610–616 (2008).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
Endicott, J. A. & Ling, V. The biochemistry of P-glycoprotein-mediated multidrug resistance. Annu. Rev. Biochem. 58, 137–171 (1989).
Schuldes, H., Dolderer, J. H., Schoch, C., Bickeboller, R. & Woodcock, B. G. Cytostatic sensitivity and MDR in bladder carcinoma cells: implications for tumor therapy. Int. J. Clin. Pharmacol. Ther. 38, 204–208 (2000).
Tasaki, Y. et al. Reversal by a dihydropyridine derivative of non-P-glycoprotein-mediated multidrug resistance in etoposide-resistant human prostatic cancer cell line. J. Urol. 154, 1210–1216 (1995).
Shinohara, N., Liebert, M., Wedemeyer, G., Chang, J. H. & Grossman, H. B. Evaluation of multiple drug resistance in human bladder cancer cell lines. J. Urol. 150, 505–509 (1993).
Park, J. et al. P-glycoprotein expression in bladder cancer. J. Urol. 151, 43–46 (1994).
Naito, S. et al. Correlation between the expression of P-glycoprotein and multidrug-resistant phenotype in transitional cell carcinoma of the urinary tract. Eur. Urol. 22, 158–162 (1992).
Hasegawa, S. et al. Expression of multidrug resistance-associated protein (MRP), MDR1 and DNA topoisomerase II in human multidrug-resistant bladder cancer cell lines. Br. J. Cancer 71, 907–913 (1995).
Nakagawa, M. et al. Clinical significance of multi-drug resistance associated protein and P-glycoprotein in patients with bladder cancer. J. Urol. 157, 1260–1264; discussion 1264–1265 (1997).
Pu, Y. S. et al. Expression of MDR-1 gene in transitional cell carcinoma and its correlation with chemotherapy response. J. Urol. 156, 271–275 (1996).
Gontero, P. et al. Ex vivo chemosensitivity to mitomycin C in bladder cancer and its relationship with P-glycoprotein and apoptotic factors. Anticancer Res. 22, 4073–4080 (2002).
Frisch, S. M. & Francis, H. Disruption of epithelial cell-matrix interactions induces apoptosis. J. Cell Biol. 124, 619–626 (1994).
Frisch, S. M. & Ruoslahti, E. Integrins and anoikis. Curr. Opin. Cell Biol. 9, 701–706 (1997).
Sethi, T. et al. Extracellular matrix proteins protect small cell lung cancer cells against apoptosis: a mechanism for small cell lung cancer growth and drug resistance in vivo. Nat. Med. 5, 662–668 (1999).
Aoudjit, F. & Vuori, K. Integrin signaling inhibits paclitaxel-induced apoptosis in breast cancer cells. Oncogene 20, 4995–5004 (2001).
Damiano, J. S., Cress, A. E., Hazlehurst, L. A., Shtil, A. A. & Dalton, W. S. Cell adhesion mediated drug resistance (CAM-DR): role of integrins and resistance to apoptosis in human myeloma cell lines. Blood 93, 1658–1667 (1999).
Pan, C. W. et al. Cell adhesion to fibronectin induces mitomycin C resistance in bladder cancer cells. BJU Int. 104, 1774–1779 (2009).
Zhang, C. J. et al. Engagement of integrin-β1 induces resistance of bladder cancer cells to mitomycin-C. Urology 79, 638–643 (2012).
Tannock, I. F., Lee, C. M., Tunggal, J. K., Cowan, D. S. & Egorin, M. J. Limited penetration of anticancer drugs through tumor tissue: a potential cause of resistance of solid tumors to chemotherapy. Clin. Cancer Res. 8, 878–884 (2002).
Burgues, J. P. et al. A chemosensitivity test for superficial bladder cancer based on three-dimensional culture of tumour spheroids. Eur. Urol. 51, 962–969; discussion 969–970 (2007).
Gazzaniga, P. et al. A chemosensitivity test to individualize intravesical treatment for non-muscle-invasive bladder cancer. BJU Int. 104, 184–188 (2009).
Lee, J. K. et al. A strategy for predicting the chemosensitivity of human cancers and its application to drug discovery. Proc. Natl Acad. Sci. USA 104, 13086–13091 (2007).
Smith, S. C., Baras, A. S., Lee, J. K. & Theodorescu, D. The COXEN principle: translating signatures of in vitro chemosensitivity into tools for clinical outcome prediction and drug discovery in cancer. Cancer Res. 70, 1753–1758 (2010).
Smith, S. C. et al. Use of yeast chemigenomics and COXEN informatics in preclinical evaluation of anticancer agents. Neoplasia 13, 72–80 (2011).
Shen, K. et al. A systematic evaluation of multi-gene predictors for the pathological response of breast cancer patients to chemotherapy. PLoS ONE 7, e49529 (2012).
Ferriss, J. S. et al. Multi-gene expression predictors of single drug responses to adjuvant chemotherapy in ovarian carcinoma: predicting platinum resistance. PLoS ONE 7, e30550 (2012).
Author information
Authors and Affiliations
Contributions
H.Z., J.A. and J.I. researched the data for the article, H.Z., A.S. and P.B. made substantial contributions to discussion of content, and all authors contributed substantially to writing and review/editing of the manuscript before publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Zargar, H., Aning, J., Ischia, J. et al. Optimizing intravesical mitomycin C therapy in non-muscle-invasive bladder cancer. Nat Rev Urol 11, 220–230 (2014). https://doi.org/10.1038/nrurol.2014.52
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2014.52
This article is cited by
-
High and selective cytotoxicity of ex vivo expanded allogeneic human natural killer cells from peripheral blood against bladder cancer: implications for natural killer cell instillation after transurethral resection of bladder tumor
Journal of Experimental & Clinical Cancer Research (2024)
-
Device-assisted intravesical chemotherapy versus bacillus Calmette–Guerin for intermediate or high-risk non-muscle invasive bladder cancer: a systematic reviewer and meta-analysis
International Urology and Nephrology (2023)
-
Adjuvant therapies for non-muscle-invasive bladder cancer: advances during BCG shortage
World Journal of Urology (2022)
-
Solubilization and Stability of Mitomycin C Solutions Prepared for Intravesical Administration
Drugs in R&D (2017)
-
The interplay of extracellular matrix and microbiome in urothelial bladder cancer
Nature Reviews Urology (2016)