Key Points
-
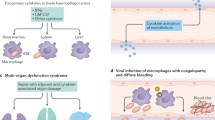
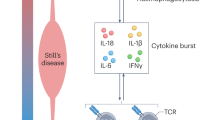
A 'cytokine storm' is the final pathophysiological pathway in macrophage activation syndrome (MAS), and blocking various cytokines could be an attractive therapeutic strategy
-
Standard doses of anti-IL-1 and anti-IL-6 biologic therapies do not have a major effect on MAS rates even if the underlying disease responds well to the treatment
-
Several case reports suggest that anakinra might be effective at least in some patients with systemic juvenile idiophatic arthritis (sJIA)-associated MAS, particularly when used in high doses
-
Findings from several studies support IFN-γ blockade as a novel therapy for haemophagocytic lymphohistiocytosis (HLH); increasing evidence suggests the same approach could be beneficial in MAS presenting as a complication of rheumatic diseases
-
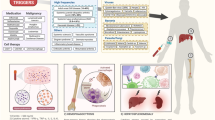
The exact mechanism of predisposition to MAS in sIJA is yet to be defined, but might be independent of underlying sJIA activity and similar to infection-associated secondary HLH
-
Whole-exome/genome sequencing approaches exploring hypomorphic mutations that affect the cytolytic pathway to support this theory might reveal promising therapeutic alternatives
Abstract
Macrophage activation syndrome (MAS) refers to acute overwhelming inflammation caused by a 'cytokine storm'. Although increasingly recognized as a life-threatening complication of various rheumatic diseases, clinically, MAS is strikingly similar to primary and secondary forms of haemophagocytic lymphohistiocytosis (HLH). Not surprisingly, many rheumatologists prefer the term secondary HLH rather than MAS to describe this condition, and efforts to change the nomenclature are in progress. The pathophysiology of MAS remains elusive, but observations in animal models, as well as data on the effects of new anticytokine therapies on rates and clinical presentations of MAS in patients with systemic juvenile idiopathic arthritis (sJIA), provide clues to the understanding of this perplexing clinical phenomenon. In this Review, we explore the latest available evidence and discuss potential diagnostic challenges in the era of increasing use of biologic therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Silverman, E. D., Miller, J. J., Bernstein, B. & Shafai, T. Consumption coagulopathy associated with systemic juvenile rheumatoid arthritis. J. Pediatr. 103, 872–876 (1983).
Hadchouel, M., Prieur, A. M. & Griscelli, C. Acute hemorrhagic, hepatic, and neurologic manifestations in juvenile rheumatoid arthritis: possible relationship to drugs or infection. J. Pediatr. 106, 561–566 (1985).
Mouy, R. et al. Efficacy of cyclosporine A in the treatment of macrophage activation syndrome in juvenile arthritis: report of five cases. J. Pediatr. 129, 750–754 (1996).
Grom, A. A. & Passo, M. Macrophage activation syndrome in systemic juvenile rheumatoid arthritis. J. Pediatr. 129, 630–632 (1996).
Ravelli, A., De Benedetti, F., Viola, S. & Martini, A. Macrophage activation syndrome in systemic juvenile rheumatoid arthritis successfully treated with cyclosporine. J. Pediatr. 128, 275–278 (1996).
Stephan, J. L. et al. Reactive haemophagocytic syndrome in children with inflammatory disorders. A retrospective study of 24 patients. Rheumatology (Oxford) 40, 1285–1292 (2001).
Sawhney, S., Woo, P. & Murray, K. J. Macrophage activation syndrome: a potentially fatal complication of rheumatic disorders. Arch. Dis. Child. 85, 421–426 (2001).
Mellins, E. D., Macubas, C. & Grom, A. A. Pathogenesis of systemic juvenile idiopathic arthritis: some answers, more questions. Nat. Rev. Rheumatol. 7, 416–426 (2011).
Rossi-Semerano, L. & Kone-Paut, I. Is Still's disease an autoinflammatory syndrome? Int. J. Inflamm. 2012, 480373 (2012).
Rigante, D. & Cantarini, L. The systemic onset variant of juvenile idiopathic arthritis needs to be recorded as an autoinnflammatory syndrome: comment on the review by Nigrovic. Arthritis Rheumatol. 66, 2645 (2014).
Pascual, V. et al. Role of interleukin-1 (IL-1) in the pathogenesis of systemic onset juvenile idiopathic arthritis and clinical response to IL-1 blockade. J. Exp. Med. 201, 1479–1486 (2005).
Quartier, P. et al. A multicentre, randomised, double-blind, placebo-controlled trial with the interleukin-1 receptor antagonist anakinra in patients with systemic-onset juvenile idiopathic arthritis. Ann. Rheum. Dis. 70, 747–754 (2011).
Ruperto, N. et al. Two randomized trials of canakinumabin systemic juvenile idiopathic arthritis. N. Engl. J. Med. 367, 2396–2406 (2012).
De Benedetti, F. & Martini, A. Is systemic juvenile rheumatoid arthritis an IL-6 mediated disease? J. Rheumatol. 25, 203–207 (1998).
Yokota, S. et al. Efficacy and safety of tocilizumab in patients with systemic-onset juvenile idiopathic arthritis: a randomised, double-blind, placebo-controlled, withdrawal phase III trial. Lancet 371, 998–1006 (2008).
De Benedetti, F. et al. Randomized trial of tocilizumab in systemic juvenile idiopathic arthritis. N. Engl. J. Med. 367, 2385–2395 (2012).
Billiau, A. D. et al. Macrophage activation syndrome: characteristic findings on liver biopsy illustrating the key role of activated, IFN-γ-producing lymphocytes and IL-6- and TNF-α-producing macrophages. Blood 105, 1648–1651 (2005).
Prahalad, S., Bove, K. E., Dickens, D., Lovell, D. J. & Grom, A. A. Successful use of etanercept in the treatment of macrophage activation syndrome. J. Rheumatol. 28, 2120–2124 (2001).
Favara, B. E. et al. Contemporary classification of histiocytic disorders. Med. Pediatr. Oncol. 199, 157–166 (1997).
Jordan, M. B. et al. How I treat hemophagocytic lymphohistiocytosis. Blood 118, 4041–4052 (2011).
Henter, J. I. et al. HLH-2004: diagnostic and therapeutic guidelines for hemophagocytic lymphohistiocytosis. Pediatr. Blood Cancer 48, 124–131 (2007).
Stepp, S. E. et al. Perforin gene defects in familial hemophagocytic lymphohistiocytosis. Science 286, 1957–1959 (1999).
Feldmann, J. et al. Munc13-4 is essential for cytolytic granules fusion and is mutated in a form of familial hemophagocytic lymphohistiocytosis (FHL3). Cell 115, 461–473 (2003).
zur Stadt, U. et al. Linkage of familial hemophagocytic lymphohistiocytosis (FHL) type-4 to chromosome 6q24 and identification of mutations in syntaxin 11. Hum. Mol. Genet. 14, 827–834 (2005).
zur Stadt, U. et al. Familial hemophagocytic lymphohistiocytosis type 5 (FHL-5) is caused by mutations in Munc18-2 and impaired binding to Syntaxin 11. Am. J. Hum. Genet. 85, 482–492 (2009).
Chandrakasan, S. & Filipovich, A. H. Hemophagocytic lymphohistiocytosis: advances in pathophysiology, diagnosis, and treatment. J. Pediatr. 163, 1253–1259 (2013).
Menasche, G. et al. Mutations in RAB27A cause Griscelli syndrome associated with haemophagocytic syndrome. Nat. Genet. 25, 173–176 (2000).
Barbosa, M. D. et al. Identification of the homologous beige and Chediak–Higashi syndrome genes. Nature 382, 262–265 (1996).
Jenkins, M. R. et al. Failed CTL/NK cell killing and cytokine hypersecretion are directly linked through prolonged synapse time. J. Exp. Med. 212, 307–317 (2015).
Menasche, G., Feldmann, J., Fischer, A. & de Saint Basile, G. Primary hemophagocytic syndromes point to a direct link between lymphocyte cytotoxicity and homeostasis. Immunol. Rev. 203, 165–179 (2005).
Kagi, D., Odermatt, B. & Mak, T. W. Homeostatic regulation of CD8+ T cells by perforin. Eur. J. Immunol. 29, 3262–3272 (1999).
Lykens, J. E., Terrell, C. E., Zoller, E. E., Risma, K. & Jordan, M. B. Perforin is a critical physiologic regulator of T-cell activation. Blood 118, 618–626 (2011).
Coffey, A. J. et al. Host response to EBV infection in X-linked lymphoproliferative disease results from mutations in an SH2-domain encoding gene. Nat. Genet. 20, 129–135 (1998).
Marsh, R. A. et al. XIAP deficiency: a unique primary immunodeficiency best classified as X-linked familial hemophagocytic lymphohistiocytosis and not as X-linked lymphoproliferative disease. Blood 116, 1079–1082 (2010).
Kogawa, K. et al. Perforin expression in cytotoxic lymphocytes from patients with hemophagocytic lymphohistiocytosis and their family members. Blood 99, 61–66 (2002).
Zhang, K. J. et al. Hypomorphic mutations in PRF1, MUNC13-4, and STXBP2 are associated with adult-onset familial HLH. Blood 118, 5794–5798 (2011).
Canna, S. W. et al. An activating NLRC4 inflammasome mutation causes autoinflammation with recurrent macrophage activation syndrome. Nat. Genet. 46, 1140–1146 (2014).
Romberg, N. et al. Mutation of NLRC4 causes a syndrome of enterocolitis and autoinflammation. Nat. Genet. 46, 1135–1139 (2014).
Grom, A. A. et al. Natural killer cell dysfunction in patients with systemic-onset juvenile rheumatoid arthritis and macrophage activation syndrome. J. Pediatr. 142, 292–296 (2003).
Cifaldi, L. et al. Inhibition of natural killer cell cytotoxicity by interleukin-6: implications for the pathogenesis of macrophage activation syndrome. Arthritis Rheum. 67, 3037–3046 (2003).
Kaufman, K. M. et al. Whole exome sequencing reveals overlap between macrophage activation syndrome in systemic juvenile idiopathic arthritis and familial hemophagocytic lymphohistiocytosis. Arthritis Rheum. 66, 3486–3495 (2014).
Bracaglia, C. et al. Mutations of familial hemophagocytic lymphohistiocytosis related genes and abnormalities of cytotoxicity function tests in patients with macrophage activation syndrome (MAS) occurring in systemic juvenile idiopathic arthritis. Pediatr. Rheumatol. Online J. 12 (Suppl. 1), 53 (2014).
Vastert, S. J. et al. Mutations in the perforin gene can be linked to macrophage activation syndrome in patients with systemic onset juvenile idiopathic arthritis. Rheumatology (Oxford) 49, 441–449 (2010).
Zhang, M. et al. Genetic defects in cytolysis in macrophage activation syndrome. Curr. Rheumatol. Rep. 16, 439–447 (2014).
Jordan, M. B., Hildeman, D., Kappler, J. & Marrack, P. An animal model of hemophagocytic lymphohistiocytosis (HLH): CD8+ T cells and interferon gamma are essential for the disorder. Blood 104, 735–743 (2004).
Pachlopnik Schmid, J. et al. Neutralization of IFNγ defeats haemophagocytosis in LCMV-infected perforin- and Rab27a-deficient mice. EMBO Mol. Med. 1, 112–124 (2009).
Behrens, E. M. et al. Repeated TLR9 stimulation results in macrophage activation syndrome-like disease in mice. J. Clin. Invest. 121, 2264–2277 (2011).
Canna, S. W. et al. Interferon-γ mediates anemia but is dispensable for fulminant toll-like receptor 9-induced macrophage activation syndrome and hemophagocytosis in mice. Arthritis Rheum. 65, 1764–1775 (2013).
De Min, C. et al. Interferon gamma (IFNg) is the driving mediator of secondary hemophagocytic lymphohistiocytosis (sHLH) in TLR9-mediated pathogenesis in mice and is correlated to disease parameters in children [abstract 3076]. Arthritis Rheumatol. 67 (Suppl. 10) (2015).
Fall, N. et al. Gene expression profiling of peripheral blood from patients with untreated new-onset systemic juvenile idiopathic arthritis reveals molecular heterogeneity that may predict macrophage activation syndrome. Arthritis Rheum. 56, 3793–3804 (2007).
Strippoli, R. et al. Amplification of the response to toll-like receptor ligands by prolonged exposure to interleukin-6 in mice: implication for the pathogenesis of macrophage activation syndrome. Arthritis Rheum. 64, 1680–1688 (2012).
Put, K. et al. Cytokines in systemic juvenile idiopathic arthritis and haemophagocytic lymphohistiocytosis: tipping the balance between interleukin-18 and interferon-γ. Rheumatology (Oxford) 54, 1507–1517 (2015).
Bracaglia, C. et al. Interferon-gamma in macrophage activation syndrome associated with systemic juvenile idiopathic arthritis: high levels in patients and a role in a murine MAS model. Presented at the 21st European Paediatric Rheumatology Congress, Belgrade (2014).
Ibarra, M. F. et al. Serum neopterin levels as a diagnostic marker of hemophagocytic lymphohistiocytosis syndrome. Clin. Vaccine Immunol. 18, 609–614 (2011).
Bleesing, J. et al. The diagnostic significance of soluble CD163 and soluble interleukin-2 receptor α-chain in macrophage activation syndrome and untreated new-onset systemic juvenile idiopathic arthritis. Arthritis Rheum. 56, 965–971 (2007).
Henter, J. I. et al. Hypercytokinemia in familial hemophagocytic lymphohistiocytosis. Blood 78, 2918–2922 (1991).
Henter, J. I. et al. Elevated circulating levels of interleukin-1 receptor antagonist but not IL-1 agonists in hemophagocytic lymphohistiocytosis. Med. Pediatr. Oncol. 27, 21–25 (1996).
Sumegi, J. et al. Gene expression profiling of peripheral blood mononuclear cells from children with active hemophagocytic lymphohistiocytosis. Blood 117, e151–e160 (2011).
Miettunen, P. M. et al. Successful treatment of severe paediatric rheumatic disease-associated macrophage activation syndrome with interleukin-1 inhibition following conventional immunosuppressive therapy: case series with 12 patients. Rheumatology (Oxford) 50, 417–419 (2011).
Durand, M., Troyanov, Y., Laflamme, P. & Gregoire, G. Macrophage activation syndrome treated with anakinra. J. Rheumatol. 37, 879–880 (2010).
Nigrovic, P. A. et al. Anakinra as first-line disease-modifying therapy in systemic juvenile idiopathic arthritis: report of forty-six patients from an international multicenter series. Arthritis Rheum. 63, 545–555 (2011).
Zeft, A. et al. Anakinra for systemic juvenile arthritis: the Rocky Mountain experience. J. Clin. Rheumatol. 15, 161–164 (2009).
Grom, A. A. et al. Rate and clinical presentation of macrophage activation syndrome in patients with systemic juvenile idiopathic arthritis treated with canakinumab. Arthritis Rheumatol. 68, 218–228 (2016).
Ilowite, N. T. et al. The RAndomized Placebo Phase Study Of Rilonacept in the Treatment of systemic juvenile idiopathic arthritis (RAPPORT). Arthritis Rheumatol. 66, 2570–2579 (2014).
Ravelli, A. et al. Macrophage activation syndrome in patients with systemic juvenile idiopathic arthritis treated with tocilizumab [abstract]. Arthritis Rheum. 66 (Suppl.), S83–S84 (2014).
Yokota, S. et al. Macrophage activation syndrome in patients with systemic juvenile idiopathic arthritis under treatment with tocilizumab. J. Rheumatol. 42, 712–722 (2015).
Shimizu, M. et al. Tocilizumab masks the clinical symptoms of systemic juvenile idiopathic arthritis-associated macrophage activation syndrome: the diagnostic significance of interleukin-18 and interleukin-6. Cytokine 58, 287–294 (2012).
Kobayashi, M. et al. Benefit and a possible risk of tocilizumab therapy for adult-onset Still's disease accompanied by macrophage-activation syndrome. Mod. Rheumatol. 21, 92–96 (2011).
Shimizu, M. et al. Distinct cytokine profiles of systemic-onset juvenile idiopathic arthritis-associated macrophage activation syndrome with particular emphasis on the role of interleukin-18 in its pathogenesis. Rheumatology (Oxford) 49, 1645–1653 (2010).
Maeno, N. et al. Increased interleukin-18 expression in bone marrow of a patient with systemic juvenile idiopathic arthritis and unrecognized macrophage-activation syndrome. Arthritis Rheum. 50, 935–1938 (2004).
Kawashima, M. et al. Levels of interleukin-18 and its binding inhibitors in the blood circulation of patients with adult-onset Still's disease. Arthritis Rheum. 44, 550–560 (2001).
Novick, D. et al. High circulating levels of free interleukin-18 in patients with active SLE in the presence of elevated levels of interleukin-18 binding protein. Cytokine 48, 103–104 (2009).
Favilli, F. et al. IL-18 activity in systemic lupus erythematosus. Ann. NY Acad. Sci. 1173, 301–309 (2009).
Mazodier, K. et al. Severe imbalance of IL-18/IL-18BP in patients with secondary hemophagocytic syndrome. Blood 106, 3483–3489 (2005).
Dinarello, C. A. Interleukin-18 and the pathogenesis of inflammatory diseases. Semin. Nephrol. 27, 98–114 (2007).
Chiossone, L. et al. Protection from inflammatory organ damage in a murine model of hemophagocytic lymphohistiocytosis using treatment with IL-18 binding protein. Front. Immunol. 3, 239–249 (2012).
Takada, H. et al. Increased serum levels of interferon-γ-inducible protein 10 and monokine induced by gamma interferon in patients with haemophagocytic lymphohistiocytosis. Clin. Exp. Immunol. 133, 448–453 (2003).
US National Library of Science. ClinicalTrials.gov [online], (2015).
Sikora, K. A., Fall, N., Thornton, S. & Grom, A. A. The limited role of interferon-γ in systemic juvenile idiopathic arthritis cannot be explained by cellular hyporesponsiveness. Arthritis Rheum. 3799–3808 (2012).
Ogilvie, E. M. et al. Specific gene expression profiles in systemic juvenile idiopathic arthritis. Arthritis Rheum. 56, 1954–1965 (2007).
Ravelli, A. et al. Preliminary diagnostic guidelines for macrophage activation syndrome complicating systemic juvenile idiopathic arthritis. J. Pediatr. 146, 598–604 (2005).
Davi, S. et al. An international consensus survey of diagnostic criteria for macrophage activation syndrome in systemic juvenile idiopathic arthritis. J. Rheumatol. 38, 764–768 (2011).
Ravelli, A. et al. Development and initial validation of classification criteria for macrophage activation syndrome complicating systemic juvenile idiopathic arthritis. Arthritis Rheumatol. http://dx.doi.org/10.1002/art.39332 (2015).
Writing Group of the Histiocyte Society. Histiocytosis syndromes in children. Lancet 1, 208–209 (1987).
Athreya, B. H. Is macrophage activation syndrome a new entity? Clin. Exp. Rheumatol. 20, 121–123 (2002).
Ramana, A. V. & Baildam, E. M. Macrophage activation syndrome is hemophagocytic lymphohistiocytosis — need for the right terminology. J. Rheumatol. 29, 1105 (2002).
Acknowledgements
A.A.G. is supported by NIH grants NIAMS R01-AR059049 and NIH P01-AR048929.
Author information
Authors and Affiliations
Contributions
A.A.G. contributed to researching data, discussions of content and writing of the article. A.C.H. contributed to discussions of content, review and editing of the manuscript before submission. F.D.B. contributed to discussions of content, review and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
A.A.G declares that he has served as a consultant and speaker for Novartis and Roche and worked in collaboration with NovImmune. F.D.B declares that he has received unrestricted research grants from Pfizer, AbbVie, Novartis, NovImmune, Roche, and SOBI, and travel support from Roche. A.C.H. declares no competing interests.
Supplementary information
Supplementary information S1 (table)
Adjudication criteria for MAS cases (PDF 84 kb)
Rights and permissions
About this article
Cite this article
Grom, A., Horne, A. & De Benedetti, F. Macrophage activation syndrome in the era of biologic therapy. Nat Rev Rheumatol 12, 259–268 (2016). https://doi.org/10.1038/nrrheum.2015.179
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2015.179
This article is cited by
-
Proceedings from the 4th NextGen Therapies for SJIA and MAS virtual symposium held February 13–14, 2022
Pediatric Rheumatology (2024)
-
Neutrophil extracellular trap-induced intermediate monocytes trigger macrophage activation syndrome in adult-onset Still’s disease
BMC Medicine (2023)
-
Hepatosplenic T-cell lymphoma with hemophagocytic lymphohistiocytosis, revealed by fever of unknown origin
memo - Magazine of European Medical Oncology (2023)
-
Pyroptosis: the potential eye of the storm in adult-onset Still’s disease
Inflammopharmacology (2023)
-
Clinical Presentation and Treatment of Juvenile Idiopathic Arthritis Combined with Lung Disease: A Narrative Review
Rheumatology and Therapy (2023)