Abstract
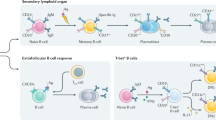
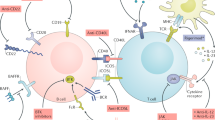
B-cell-directed therapy—the use of agents that eliminate B cells or block cytokines important for B-cell function—is emerging as a promising approach to the treatment of rheumatic disease. Target diseases, including systemic lupus erythematosus (SLE), display diverse patterns of autoantibody production and aberrant activation of B cells. Despite the success of this general approach, the mechanisms by which B-cell-directed therapy ameliorates disease, and the role of autoantibodies as biomarkers of clinical response remain unclear. Importantly, although B-cell-directed therapy can reduce the production of some autoantibodies, the effects can be variable and heterogeneous, probably reflecting the critical (but ill-defined) roles of different B-cell and plasma cell populations in autoantibody production. Future studies during clinical trials of these agents are needed to define which B-cell and autoantibody populations are affected (or ought to be), and to discover informative biomarkers of clinical response that can be used to advance this therapeutic approach.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Dörner, T. & Burmester, G. R. New approaches of B-cell-directed therapy: beyond rituximab. Curr. Opin. Rheumatol. 20, 263–268 (2008).
Cohen, S. B. et al. Rituximab for rheumatoid arthritis refractory to anti-tumor necrosis factor therapy: results of a multicenter, randomized, double-blind, placebo-controlled, phase III trial evaluating primary efficacy and safety at twenty-four weeks. Arthritis Rheum. 54, 2793–2806 (2006).
Stone, J. H. et al. Rituximab versus cyclophosphamide for ANCA-associated vasculitis. N. Engl. J. Med. 363, 221–232 (2010).
Hauser, S. L. et al. B-cell depletion with rituximab in relapsing–remitting multiple sclerosis. N. Engl. J. Med. 358, 676–688 (2008).
Merrill, J. T. et al. Efficacy and safety of rituximab in moderately-to-severely active systemic lupus erythematosus: the randomized, double-blind, phase II/III systemic lupus erythematosus evaluation of rituximab trial. Arthritis Rheum. 62, 222–233 (2010).
Navarra, S. V. et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet 377, 721–731 (2011).
Wallace, D. J. et al. Epratuzumab demonstrates clinically meaningful improvements in patients with moderate to severe systemic lupus erythematosus: results from EMBLEM, a phase IIb study. Presented at the 9th International Congress on Systemic Lupus Erythematosus, Vancouver, Canada, June 24–27 2010.
Dörner, T., Jacobi, A. M. & Lipsky, P. E. B cells in autoimmunity. Arthritis Res Ther 11, 247 (2009).
Hochberg, M. C. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 40, 1725 (1997).
Hahn, B. H. Antibodies to DNA. N. Engl. J. Med. 338, 1359–1368 (1998).
van Venrooij, W. J. et al. Anti-(U1) small nuclear RNA antibodies in anti-small nuclear ribonucleoprotein sera from patients with connective tissue diseases. J. Clin. Invest. 86, 2154–2160 (1990).
Migliorini, P., Baldini, C., Rocchi, V. & Bombardieri, S. Anti-Sm and anti-RNP antibodies. Autoimmunity 38, 47–54 (2005).
Franceschini, F. & Cavazzana, I. Anti-Ro/SSA and La/SSB antibodies. Autoimmunity 38, 55–63 (2005).
Cameron, J. S., Lessof, M. H., Ogg, C. S., Williams, B. D. & Williams, D. G. Disease activity in the nephritis of systemic lupus erythematosus in relation to serum complement concentrations. DNA-binding capacity and precipitating anti-DNA antibody. Clin. Exp. Immunol. 25, 418–427 (1976).
Yung, S. & Chan, T. M. Anti-DNA antibodies in the pathogenesis of lupus nephritis—the emerging mechanisms. Autoimmun. Rev. 7, 317–321 (2008).
Furie, R. et al. Effect of Rituximab (RTX) On Anti-dsDNA and C3 Levels and Relationship to Response: Results From the LUNAR Trial [abstract 271]. Arthritis Rheum. 60 (Suppl.), S99 (2009).
Lovgren, T., Eloranta, M. L., Båve, U., Alm, G. V. & Rönnblom, L. Induction of interferon-α production in plasmacytoid dendritic cells by immune complexes containing nucleic acid released by necrotic or late apoptotic cells and lupus IgG. Arthritis Rheum. 50, 1861–1872 (2004).
Herlands, R. A., Christensen, S. R., Sweet, R. A., Hershberg, U. & Shlomchik, M. J. T cell-independent and toll-like receptor-dependent antigen-driven activation of autoreactive B cells. Immunity 29, 249–260 (2008).
Tian, J. et al. Toll-like receptor 9-dependent activation by DNA-containing immune complexes is mediated by HMGB1 and RAGE. Nat. Immunol. 8, 487–496 (2007).
Guiducci, C. et al. TLR recognition of self nucleic acids hampers glucocorticoid activity in lupus. Nature 465, 937–941 (2010).
Tew, G. W. et al. Baseline autoantibody profiles predict normalization of complement and anti-dsDNA autoantibody levels following rituximab treatment in systemic lupus erythematosus. Lupus 19, 146–157 (2010).
McCarty, G. A., Rice, J. R., Bembe, M. L. & Pisetsky, D. S. Independent expression of autoantibodies in systemic lupus erythematosus. J. Rheumatol. 9, 691–695 (1982).
Kavanaugh, A. F. & Solomon, D. H. Guidelines for immunologic laboratory testing in the rheumatic diseases: anti-DNA antibody tests. Arthritis Rheum. 47, 546–555 (2002).
Fragoso-Loyo, H. et al. Serum and cerebrospinal fluid autoantibodies in patients with neuropsychiatric lupus erythematosus. Implications for diagnosis and pathogenesis. PLoS ONE 3, e3347 (2008).
Agarwal, S., Harper, J. & Kiely, P. D. Concentration of antibodies to extractable nuclear antigens and disease activity in systemic lupus erythematosus. Lupus 18, 407–412 (2009).
Arbuckle, M. R. et al. Development of autoantibodies before the clinical onset of systemic lupus erythematosus. N. Engl. J. Med. 349, 1526–1533 (2003).
Grammer, A. C. et al. Abnormal germinal center reactions in systemic lupus erythematosus demonstrated by blockade of CD154-CD40 interactions. J. Clin. Invest. 112, 1506–1520 (2003).
Hoyer, B. F., Manz, R. A., Radbruch, A. & Hiepe, F. Long-lived plasma cells and their contribution to autoimmunity. Ann. NY Acad. Sci. 1050, 124–133 (2005).
Neubert, K. et al. The proteasome inhibitor bortezomib depletes plasma cells and protects mice with lupus-like disease from nephritis. Nat. Med. 14, 748–755 (2008).
Halicka, H. D., Bedner, E. & Darzynkiewicz, Z. Segregation of RNA and separate packaging of DNA and RNA in apoptotic bodies during apoptosis. Exp. Cell Res. 260, 248–256 (2000).
Pisetsky, D. S., McCarty, G. A. & Peters, D. V. Mechanisms of autoantibody production in autoimmune MRL mice. J. Exp. Med. 152, 1302–1310 (1980).
Eisenberg, R. A., Craven, S. Y., Warren, R. W. & Cohen, P. L. Stochastic control of anti-Sm autoantibodies in MRL/Mp-lpr/lpr mice. J. Clin. Invest. 80, 691–697 (1987).
Pisetsky, D., Caster, S., Roths, J. B. & Murphy, E. Lpr gene control of the anti-DNA antibody response. J. Immunol. 128, 2322–2325 (1982).
Pawar, R. D. et al. Inhibition of Toll-like receptor-7 (TLR-7) or TLR-7 plus TLR-9 attenuates glomerulonephritis and lung injury in experimental lupus. J. Am. Soc. Nephrol. 18, 1721–1731 (2007).
Nickerson, K. M. et al. TLR9 regulates TLR7- and MyD88-dependent autoantibody production and disease in a murine model of lupus. J. Immunol. 184, 1840–1848 (2010).
Witte, T. et al. IgM anti-dsDNA antibodies in systemic lupus erythematosus: negative association with nephritis. SLE Study Group. Rheumatol. Int. 18, 85–91 (1998).
Hoyer, B. F. et al. Short-lived plasmablasts and long-lived plasma cells contribute to chronic humoral autoimmunity in NZB/W mice. J. Exp. Med. 199, 1577–1584 (2004).
Bingham, C. O. 3rd et al. Immunization responses in rheumatoid arthritis patients treated with rituximab: results from a controlled clinical trial. Arthritis Rheum. 62, 64–74 (2010).
Jacobi, A. M. et al. Effect of long-term belimumab treatment on B cells in systemic lupus erythematosus: extension of a phase II, double-blind, placebo-controlled, dose-ranging study. Arthritis Rheum. 62, 201–210 (2010).
Hiepe, F. et al. Long-lived autoreactive plasma cells drive persistent autoimmune inflammation. Nat. Rev. Rheumatol. 7, 170–178 (2011).
Hartung, H. P. & Kieseier, B. C. Atacicept: targeting B cells in multiple sclerosis. Ther. Adv. Neurol. Disord. 3, 205–216 (2010).
Acknowledgements
The authors' research activities are supported by a Veterans Administration Merit Review grant and NIH grants AI083923, AI082302 and T32-AI07217-28. P. E. Lipsky and A. C. Grammer were formerly in the autoimmunity branch of the Intramural Research Program, NIAMS, NIH, Bethseda, MD, USA.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to all aspects of the preparation of this manuscript.
Corresponding author
Supplementary information
Supplementary Table 1
(DOC 52 kb)
Rights and permissions
About this article
Cite this article
Pisetsky, D., Grammer, A., Ning, T. et al. Are autoantibodies the targets of B-cell-directed therapy?. Nat Rev Rheumatol 7, 551–556 (2011). https://doi.org/10.1038/nrrheum.2011.108
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2011.108