Abstract
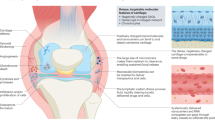
Gene transfer technologies enable the controlled, targeted and sustained expression of gene products at precise anatomical locations, such as the joint. In this way, they offer the potential for more-effective, less-expensive treatments of joint diseases with fewer extra-articular adverse effects. A large body of preclinical data confirms the utility of intra-articular gene therapy in animal models of rheumatoid arthritis and osteoarthritis. However, relatively few clinical trials have been conducted, only one of which has completed phase II. This article summarizes the status in 2010 of the clinical development of gene therapy for arthritis, identifies certain constraints to progress and suggests possible solutions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Bandara, G. et al. Gene transfer to synoviocytes: prospects for gene treatment of arthritis. DNA Cell Biol. 11, 227–231 (1992).
Mease, P. J. et al. Safety, tolerability, and clinical outcomes after intraarticular injection of a recombinant adeno-associated vector containing a tumor necrosis factor antagonist gene: results of a phase 1/2 study. J. Rheumatol. 37, 692–703 (2010).
Traister, R. S. & Hirsch, R. Gene therapy for arthritis. Mod. Rheumatol. 18, 2–14 (2008).
Ghivizzani, S. C. et al. Perspectives on the use of gene therapy for chronic joint diseases. Curr. Gene Ther. 8, 273–286 (2008).
Gouze, E. et al. Transgene persistence and cell turnover in the diarthrodial joint: implications for gene therapy of chronic joint diseases. Mol. Ther. 15, 1114–1120 (2007).
Madry, H., Cucchiarini, M., Terwilliger, E. F. & Trippel, S. B. Recombinant adeno-associated virus vectors efficiently and persistently transduce chondrocytes in normal and osteoarthritic human articular cartilage. Hum. Gene Ther. 14, 393–402 (2003).
Zhang, H., Gao, G., Clayburne, G. & Schumacher, H. R. Elimination of rheumatoid synovium in situ using a Fas ligand 'gene scalpel'. Arthritis Res. Ther. 7, R1235–R1243 (2005).
Goossens, P. H. et al. Feasibility of adenovirus-mediated nonsurgical synovectomy in collagen-induced arthritis-affected rhesus monkeys. Hum. Gene Ther. 10, 1139–1149 (1999).
Sant, S. M. et al. Molecular lysis of synovial lining cells by in vivo herpes simplex virus-thymidine kinase gene transfer. Hum. Gene Ther. 9, 2735–2743 (1998).
Miagkov, A. V., Varley, A. W., Munford, R. S. & Makarov, S. S. Endogenous regulation of a therapeutic transgene restores homeostasis in arthritic joints. J. Clin. Invest. 109, 1223–1229 (2002).
Evans, C. H. et al. Gene transfer to human joints: progress toward a gene therapy of arthritis. Proc. Natl Acad. Sci. USA 102, 8698–8703 (2005).
Bendele, A. et al. Efficacy of sustained blood levels of interleukin-1 receptor antagonist in animal models of arthritis: comparison of efficacy in animal models with human clinical data. Arthritis Rheum. 42, 498–506 (1999).
Makarov, S. S. et al. Suppression of experimental arthritis by gene transfer of interleukin 1 receptor antagonist cDNA. Proc. Natl Acad. Sci. USA 93, 402–406 (1996).
Bandara, G. et al. Intraarticular expression of biologically active interleukin 1-receptor-antagonist protein by ex vivo gene transfer. Proc. Natl Acad. Sci. USA 90, 10764–10768 (1993).
Wehling, P. et al. Clinical responses to gene therapy in joints of two subjects with rheumatoid arthritis. Hum. Gene Ther. 20, 97–101 (2009).
Kohn, D. B., Sadelain, M. & Glorioso, J. C. Occurrence of leukaemia following gene therapy of X-linked SCID. Nat. Rev. Cancer 3, 477–488 (2003).
Miles, B. J. et al. Prostate-specific antigen response and systemic T cell activation after in situ gene therapy in prostate cancer patients failing radiotherapy. Hum. Gene Ther. 12, 1955–1967 (2001).
Raper, S. E. et al. Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenoviral gene transfer. Mol. Genet. Metab. 80, 148–158 (2003).
Mease, P. J. et al. Local delivery of a recombinant adenoassociated vector containing a tumor necrosis factor alpha antagonist gene in inflammatory arthritis: a phase 1 dose-escalation safety and tolerability study. Ann. Rheum. Dis. 68, 1247–1254 (2009).
Frank, K. M. et al. Investigation of the cause of death in a gene-therapy trial. N. Engl. J. Med. 361, 161–169 (2009).
Gibbons, L. J. & Hyrich, K. L. Biologic therapy for rheumatoid arthritis: clinical efficacy and predictors of response. BioDrugs 23, 111–124 (2009).
Tomita, N., Morishita, R., Tomita, T. & Ogihara, T. Potential therapeutic applications of decoy oligonucleotides. Curr. Opin. Mol. Ther. 4, 166–170 (2002).
Safety study of TissueGene-C in degenerative joint disease of the knee (TGC-03-01). ClinicalTrials.gov identifier: NCT00599248, [online], (2010).
Pelletier, J. P. et al. In vivo suppression of early experimental osteoarthritis by interleukin-1 receptor antagonist using gene therapy. Arthritis Rheum. 40, 1012–1019 (1997).
Zhang, X., Mao, Z. & Yu, C. Suppression of early experimental osteoarthritis by gene transfer of interleukin-1 receptor antagonist and interleukin-10. J. Orthop. Res. 22, 742–750 (2004).
Frisbie, D. D., Ghivizzani, S. C., Robbins, P. D., Evans, C. H. & McIlwraith, C. W. Treatment of experimental equine osteoarthritis by in vivo delivery of the equine interleukin-1 receptor antagonist gene. Gene Ther. 9, 12–20 (2002).
Sun, J. et al. Intraarticular factor IX protein or gene replacement protects against development of hemophilic synovitis in the absence of circulating factor IX. Blood 112, 4532–4541 (2008).
Manno, C. S. et al. Successful transduction of liver in hemophilia by AAV-Factor IX and limitations imposed by the host immune response. Nat. Med. 12, 342–347 (2006).
Tyler, W. K., Vidal, A. F., Williams, R. J. & Healey, J. H. Pigmented villonodular synovitis. J. Am. Acad. Orthop. Surg. 14, 376–385 (2006).
Byers, S., Rothe, M., Lalic, J., Koldej, R. & Anson, D. S. Lentiviral-mediated correction of MPS VI cells and gene transfer to joint tissues. Mol. Genet. Metab. 97, 102–108 (2009).
Evans, C. H. Arthritis gene therapy at an inflection point. Future Rheumatol. 3, 207–210 (2008).
[No authors listed] Gene therapy deserves a fresh chance. Nature 461, 1173 (2009).
Acknowledgements
The authors' work in this area has been funded by NIH grants R01 AR43623, R21 AR049606, R01 AR048566, R01 AR057422 and R01 AR051085, and by Orthogen.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to researching data for the article, providing a substantial contribution to discussion of content, writing the article and review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
C. H. Evans and P. D. Robbins are consultants for, and have received honoraria from, TissueGene. C. H. Evans is on the supervisory board of and owns stock in Orthogen. P. D. Robbins and S. C. Ghivizzani are founders of Molecular Orthopaedics.
Rights and permissions
About this article
Cite this article
Evans, C., Ghivizzani, S. & Robbins, P. Getting arthritis gene therapy into the clinic. Nat Rev Rheumatol 7, 244–249 (2011). https://doi.org/10.1038/nrrheum.2010.193
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2010.193
This article is cited by
-
Treatment of osteoarthritis using a helper-dependent adenoviral vector retargeted to chondrocytes
Molecular Therapy - Methods & Clinical Development (2016)
-
Current research on pharmacologic and regenerative therapies for osteoarthritis
Bone Research (2016)
-
scAAVIL-1ra dosing trial in a large animal model and validation of long-term expression with repeat administration for osteoarthritis therapy
Gene Therapy (2015)
-
Gene therapy approaches to regenerating the musculoskeletal system
Nature Reviews Rheumatology (2015)