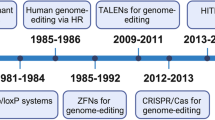
Abstract
Engineered nucleases enable the targeted alteration of nearly any gene in a wide range of cell types and organisms. The newly-developed transcription activator-like effector nucleases (TALENs) comprise a nonspecific DNA-cleaving nuclease fused to a DNA-binding domain that can be easily engineered so that TALENs can target essentially any sequence. The capability to quickly and efficiently alter genes using TALENs promises to have profound impacts on biological research and to yield potential therapeutic strategies for genetic diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Baker, M. Gene-editing nucleases. Nature Methods 9, 23–26 (2012).
Urnov, F. D., Rebar, E. J., Holmes, M. C., Zhang, H. S. & Gregory, P. D. Genome editing with engineered zinc finger nucleases. Nature Rev. Genet. 11, 636–646 (2010).
Perez, E. E. et al. Establishment of HIV-1 resistance in CD4+ T cells by genome editing using zinc-finger nucleases. Nature Biotech. 26, 808–816 (2008).
Boch, J. & Bonas, U. Xanthomonas AvrBs3 family-type III effectors: discovery and function. Annu. Rev. Phytopathol. 48, 419–436 (2010).
Li, T. et al. Modularly assembled designer TAL effector nucleases for targeted gene knockout and gene replacement in eukaryotes. Nucleic Acids Res. 39, 6315–6325 (2011).
Liu, J. et al. Efficient and specific modifications of the Drosophila genome by means of an easy TALEN strategy. J. Genet. Genom. 39, 209–215 (2012).
Wood, A. J. et al. Targeted genome editing across species using ZFNs and TALENs. Science 333, 307 (2011).
Watanabe, T. et al. Non-transgenic genome modifications in a hemimetabolous insect using zinc-finger and TAL effector nucleases. Nature Commun. 3, 1017 (2012).
Sander, J. D. et al. Targeted gene disruption in somatic zebrafish cells using engineered TALENs. Nature Biotech. 29, 697–698 (2011).
Huang, P. et al. Heritable gene targeting in zebrafish using customized TALENs. Nature Biotech. 29, 699–700 (2011).
Bedell, V. M. et al. In vivo genome editing using a high-efficiency TALEN system. Nature 23 Sep 2012 (doi:10.1038/nature11537).
Lei, Y. et al. Efficient targeted gene disruption in Xenopus embryos using engineered transcription activator-like effector nucleases (TALENs). Proc. Natl Acad. Sci. USA 109, 17484–17489 (2012).
Tesson, L. et al. Knockout rats generated by embryo microinjection of TALENs. Nature Biotech. 29, 695–696 (2011).
Carlson, D. F. et al. Efficient TALEN-mediated gene knockout in livestock. Proc. Natl Acad. Sci. USA 109, 17382–17387 (2012).
Cermak, T. et al. Efficient design and assembly of custom TALEN and other TAL effector-based constructs for DNA targeting. Nucleic Acids Res. 39, e82 (2011).
Li, T., Liu, B., Spalding, M. H., Weeks, D. P. & Yang, B. High-efficiency TALEN-based gene editing produces disease-resistant rice. Nature Biotech. 30, 390–392 (2012).
Ma, S. et al. Highly efficient and specific genome editing in silkworm using custom TALENs. PLoS ONE 7, e45035 (2012).
Reyon, D. et al. FLASH assembly of TALENs for high-throughput genome editing. Nature Biotech. 30, 460–465 (2012).
Miller, J. C. et al. A TALE nuclease architecture for efficient genome editing. Nature Biotech. 29, 143–148 (2011).
Hockemeyer, D. et al. Genetic engineering of human pluripotent cells using TALE nucleases. Nature Biotech. 29, 731–734 (2011).
Boch, J. et al. Breaking the code of DNA binding specificity of TAL-type III effectors. Science 326, 1509–1512 (2009).
Moscou, M. J. & Bogdanove, A. J. A simple cipher governs DNA recognition by TAL effectors. Science 326, 1501 (2009).
Morbitzer, R., Romer, P., Boch, J. & Lahaye, T. Regulation of selected genome loci using de novo-engineered transcription activator-like effector (TALE)-type transcription factors. Proc. Natl Acad. Sci. USA 107, 21617–21622 (2010).
Christian, M. et al. Targeting DNA double-strand breaks with TAL effector nucleases. Genetics 186, 757–761 (2010).
Li, T. et al. TAL nucleases (TALNs): hybrid proteins composed of TAL effectors and FokI DNA-cleavage domain. Nucleic Acids Res. 39, 359–372 (2011).
Mak, A. N., Bradley, P., Cernadas, R. A., Bogdanove, A. J. & Stoddard, B. L. The crystal structure of TAL effector PthXo1 bound to its DNA target. Science 335, 716–719 (2012).
Deng, D. et al. Structural basis for sequence-specific recognition of DNA by TAL effectors. Science 335, 720–723 (2012).
Streubel, J., Blucher, C., Landgraf, A. & Boch, J. TAL effector RVD specificities and efficiencies. Nature Biotech. 30, 593–595 (2012).
Cong, L., Zhou, R., Kuo, Y. C., Cunniff, M. & Zhang, F. Comprehensive interrogation of natural TALE DNA-binding modules and transcriptional repressor domains. Nature Commun. 3, 968 (2012).
Handel, E. M. & Cathomen, T. Zinc-finger nuclease based genome surgery: it's all about specificity. Curr. Gene Ther. 11, 28–37 (2011).
Moehle, E. A. et al. Targeted gene addition into a specified location in the human genome using designed zinc finger nucleases. Proc. Natl Acad. Sci. USA 104, 3055–3060 (2007).
Chen, F. et al. High-frequency genome editing using ssDNA oligonucleotides with zinc-finger nucleases. Nature Methods 8, 753–755 (2011).
Bibikova, M., Golic, M., Golic, K. G. & Carroll, D. Targeted chromosomal cleavage and mutagenesis in Drosophila using zinc-finger nucleases. Genetics 161, 1169–1175 (2002).
Bibikova, M., Beumer, K., Trautman, J. K. & Carroll, D. Enhancing gene targeting with designed zinc finger nucleases. Science 300, 764 (2003).
Morton, J., Davis, M. W., Jorgensen, E. M. & Carroll, D. Induction and repair of zinc-finger nuclease-targeted double-strand breaks in Caenorhabditis elegans somatic cells. Proc. Natl Acad. Sci. USA 103, 16370–16375 (2006).
Doyon, Y. et al. Heritable targeted gene disruption in zebrafish using designed zinc-finger nucleases. Nature Biotech. 26, 702–708 (2008).
Meng, X., Noyes, M. B., Zhu, L. J., Lawson, N. D. & Wolfe, S. A. Targeted gene inactivation in zebrafish using engineered zinc-finger nucleases. Nature Biotech. 26, 695–701 (2008).
Yano, A. et al. An immune-related gene evolved into the master sex-determining gene in rainbow trout, Oncorhynchus mykiss. Curr. Biol. 22, 1423–1428 (2012).
Dong, Z. et al. Heritable targeted inactivation of myostatin gene in yellow catfish (Pelteobagrus fulvidraco) using engineered zinc finger nucleases. PLoS ONE 6, e28897 (2011).
Ochiai, H. et al. Targeted mutagenesis in the sea urchin embryo using zinc-finger nucleases. Genes Cells 15, 875–885 (2010).
Young, J. J. et al. Efficient targeted gene disruption in the soma and germ line of the frog Xenopus tropicalis using engineered zinc-finger nucleases. Proc. Natl Acad. Sci. USA 108, 7052–7057 (2011).
Hauschild, J. et al. Efficient generation of a biallelic knockout in pigs using zinc-finger nucleases. Proc. Natl Acad. Sci. USA 108, 12013–12017 (2011).
Yu, S. et al. Highly efficient modification of β-lactoglobulin (BLG) gene via zinc-finger nucleases in cattle. Cell Res. 21, 1638–1640 (2011).
Flisikowska, T. et al. Efficient immunoglobulin gene disruption and targeted replacement in rabbit using zinc finger nucleases. PLoS ONE 6, e21045 (2011).
Takasu, Y. et al. Targeted mutagenesis in the silkworm Bombyx mori using zinc finger nuclease mRNA injection. Insect Biochem. Mol. Biol. 40, 759–765 (2010).
Merlin, C., Beaver, L. E., Taylor, O. R., Wolfe, S. A. & Reppert, S. M. Efficient targeted mutagenesis in the monarch butterfly using zinc finger nucleases. Genome Res. 25 Sep 2012 (doi:10.1101/gr.145599.112).
Meyer, M., de Angelis, M. H., Wurst, W. & Kuhn, R. Gene targeting by homologous recombination in mouse zygotes mediated by zinc-finger nucleases. Proc. Natl Acad. Sci. USA 107, 15022–15026 (2010).
Carbery, I. D. et al. Targeted genome modification in mice using zinc-finger nucleases. Genetics 186, 451–459 (2010).
Cui, X. et al. Targeted integration in rat and mouse embryos with zinc-finger nucleases. Nature Biotech. 29, 64–67 (2011).
Geurts, A. M. et al. Knockout rats via embryo microinjection of zinc-finger nucleases. Science 325, 433 (2009).
Curtin, S. J. et al. Targeted mutagenesis of duplicated genes in soybean with zinc-finger nucleases. Plant Physiol. 156, 466–473 (2011).
Sander, J. D. et al. Selection-free zinc-finger-nuclease engineering by context-dependent assembly (CoDA). Nature Methods 8, 67–69 (2011).
Lloyd, A., Plaisier, C. L., Carroll, D. & Drews, G. N. Targeted mutagenesis using zinc-finger nucleases in Arabidopsis. Proc. Natl Acad. Sci. USA 102, 2232–2237 (2005).
Zhang, F. et al. High frequency targeted mutagenesis in Arabidopsis thaliana using zinc finger nucleases. Proc. Natl Acad. Sci. USA 107, 12028–12033 (2010).
Shukla, V. K. et al. Precise genome modification in the crop species Zea mays using zinc-finger nucleases. Nature 459, 437–441 (2009).
Townsend, J. A. et al. High-frequency modification of plant genes using engineered zinc-finger nucleases. Nature 459, 442–445 (2009).
Marton, I. et al. Nontransgenic genome modification in plant cells. Plant Physiol. 154, 1079–1087 (2010).
Santiago, Y. et al. Targeted gene knockout in mammalian cells by using engineered zinc-finger nucleases. Proc. Natl Acad. Sci. USA 105, 5809–5814 (2008).
Urnov, F. D. et al. Highly efficient endogenous human gene correction using designed zinc-finger nucleases. Nature 435, 646–651 (2005).
Porteus, M. H. & Baltimore, D. Chimeric nucleases stimulate gene targeting in human cells. Science 300, 763 (2003).
Zou, J. et al. Gene targeting of a disease-related gene in human induced pluripotent stem and embryonic stem cells. Cell Stem Cell 5, 97–110 (2009).
Hockemeyer, D. et al. Efficient targeting of expressed and silent genes in human ESCs and iPSCs using zinc-finger nucleases. Nature Biotech. 27, 851–857 (2009).
Lombardo, A. et al. Gene editing in human stem cells using zinc finger nucleases and integrase-defective lentiviral vector delivery. Nature Biotech. 25, 1298–1306 (2007).
Lee, H. J., Kim, E. & Kim, J. S. Targeted chromosomal deletions in human cells using zinc finger nucleases. Genome Res. 20, 81–89 (2010).
Brunet, E. et al. Chromosomal translocations induced at specified loci in human stem cells. Proc. Natl Acad. Sci. USA 106, 10620–10625 (2009).
Simsek, D. et al. DNA ligase III promotes alternative nonhomologous end-joining during chromosomal translocation formation. PLoS Genet. 7, e1002080 (2011).
Lee, H. J., Kweon, J., Kim, E., Kim, S. & Kim, J. S. Targeted chromosomal duplications and inversions in the human genome using zinc finger nucleases. Genome Res. 22, 539–548 (2012).
Mussolino, C. et al. A novel TALE nuclease scaffold enables high genome editing activity in combination with low toxicity. Nucleic Acids Res. 39, 9283–9293 (2011).
Sanjana, N. E. et al. A transcription activator-like effector toolbox for genome engineering. Nature Protoc. 7, 171–192 (2012).
Sun, N., Liang, J., Abil, Z. & Zhao, H. Optimized TAL effector nucleases (TALENs) for use in treatment of sickle cell disease. Mol. Biosyst. 8, 1255–1263 (2012).
Kim, H., Um, E., Cho, S. R., Jung, C. & Kim, J. S. Surrogate reporters for enrichment of cells with nuclease-induced mutations. Nature Methods 8, 941–943 (2011).
Sebastiano, V. et al. In situ genetic correction of the sickle cell anemia mutation in human induced pluripotent stem cells using engineered zinc finger nucleases. Stem Cells 29, 1717–1726 (2011).
Yusa, K. et al. Targeted gene correction of α1-antitrypsin deficiency in induced pluripotent stem cells. Nature 478, 391–394 (2011).
Soldner, F. et al. Generation of isogenic pluripotent stem cells differing exclusively at two early onset Parkinson point mutations. Cell 146, 318–331 (2011).
Cade, L. et al. Highly efficient generation of heritable zebrafish gene mutations using homo- and heterodimeric TALENs. Nucleic Acids Res. 140, 8001–8010 (2012).
Doyon, Y. et al. Enhancing zinc-finger-nuclease activity with improved obligate heterodimeric architectures. Nature Methods 8, 74–79 (2011).
Moore, F. E. et al. Improved somatic mutagenesis in zebrafish using transcription activator-like effector nucleases (TALENs). PLoS ONE 7, e37877 (2012).
Ramirez, C. L. et al. Engineered zinc finger nickases induce homology-directed repair with reduced mutagenic effects. Nucleic Acids Res. 40, 5560–5568 (2012).
Wang, J. et al. Targeted gene addition to a predetermined site in the human genome using a ZFN-based nicking enzyme. Genome Res. 22, 1316–1326 (2012).
Kim, E. et al. Precision genome engineering with programmable DNA-nicking enzymes. Genome Res. 22, 1327–1333 (2012).
Gaj, T., Guo, J., Kato, Y., Sirk, S. J. & Barbas, C. F. Targeted gene knockout by direct delivery of zinc-finger nuclease proteins. Nature Methods 9, 805–807 (2012).
Mercer, A. C., Gaj, T., Fuller, R. P. & Barbas, C. F. Chimeric TALE recombinases with programmable DNA sequence specificity. Nucleic Acids Res. 26 Sep 2012 (doi:10.1093/nar/gks875).
Wolfe, S. A., Nekludova, L. & Pabo, C. O. DNA recognition by Cys2His2 zinc finger proteins. Annu. Rev. Biophys. Biomol. Struct. 29, 183–212 (2000).
Pabo, C. O., Peisach, E. & Grant, R. A. Design and selection of novel Cys2His2 zinc finger proteins. Annu. Rev. Biochem. 70, 313–340 (2001).
Greisman, H. A. & Pabo, C. O. A general strategy for selecting high-affinity zinc finger proteins for diverse DNA target sites. Science 275, 657–661 (1997).
Isalan, M., Choo, Y. & Klug, A. Synergy between adjacent zinc fingers in sequence-specific DNA recognition. Proc. Natl Acad. Sci. USA 94, 5617–5621 (1997).
Wolfe, S. A., Greisman, H. A., Ramm, E. I. & Pabo, C. O. Analysis of zinc fingers optimized via phage display: evaluating the utility of a recognition code. J. Mol. Biol. 285, 1917–1934 (1999).
Beerli, R. R. & Barbas, C. F. Engineering polydactyl zinc-finger transcription factors. Nature Biotech. 20, 135–141 (2002).
Ramirez, C. L. et al. Unexpected failure rates for modular assembly of engineered zinc fingers. Nature Methods 5, 374–375 (2008).
Maeder, M. L. et al. Rapid “open-source” engineering of customized zinc-finger nucleases for highly efficient gene modification. Mol. Cell 31, 294–301 (2008).
Kim, Y. G., Cha, J. & Chandrasegaran, S. Hybrid restriction enzymes: zinc finger fusions to Fok I cleavage domain. Proc. Natl Acad. Sci. USA 93, 1156–1160 (1996).
Blancafort, P., Segal, D. J. & Barbas, C. F. Designing transcription factor architectures for drug discovery. Mol. Pharmacol. 66, 1361–1371 (2004).
Zhang, F. et al. Efficient construction of sequence-specific TAL effectors for modulating mammalian transcription. Nature Biotech. 29, 149–153 (2011).
Geissler, R. et al. Transcriptional activators of human genes with programmable DNA-specificity. PLoS ONE 6, e19509 (2011).
Garg, A., Lohmueller, J. J., Silver, P. A. & Armel, T. Z. Engineering synthetic TAL effectors with orthogonal target sites. Nucleic Acids Res. 40, 7584–7595 (2012).
Tremblay, J. P., Chapdelaine, P., Coulombe, Z. & Rousseau, J. TALE proteins induced the expression of the frataxin gene. Hum. Gene Ther. 23, 883–890 (2012).
Wang, Z. et al. An integrated Chip for the high-throughput synthesis of transcription activator-like effectors. Angew. Chem. Int. Ed. Engl. 51, 8505–8508 (2012).
Bultmann, S. et al. Targeted transcriptional activation of silent oct4 pluripotency gene by combining designer TALEs and inhibition of epigenetic modifiers. Nucleic Acids Res. 40, 5368–5377 (2012).
Liu, P. Q. et al. Regulation of an endogenous locus using a panel of designed zinc finger proteins targeted to accessible chromatin regions. Activation of vascular endothelial growth factor A. J. Biol. Chem. 276, 11323–11334 (2001).
Doyle, E. L. et al. TAL effector-nucleotide targeter (TALE-NT) 2.0: tools for TAL effector design and target prediction. Nucleic Acids Res. 40, W117–W122 (2012).
Acknowledgements
J.K.J. acknowledges support from the US National Institutes of Health (NIH) (grants DP1 GM105378, R01 GM088040 and P50 HG005550) and The Jim and Ann Orr Massachusetts General Hospital Research Scholar Award. J.D.S. was supported by the NIH grant T32CA009216. The authors apologize to colleagues whose studies were not cited due to length constraints.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
J. Keith Joung has a financial interest in Transposagen Biopharmaceuticals. His interests were reviewed and are managed by Massachusetts General Hospital and Partners HealthCare in accordance with their conflict of interest policies. J. Keith Joung and Jeffry D. Sander are inventors on a patent application describing the FLASH assembly method.
Supplementary information
Supplementary Information S3 | Platforms for assembling DNA encoding TALE repeat arrays (PDF 189 kb)
Supplementary Information S4 | Methods for constructing DNA encoding TALE repeat arrays (PDF 119 kb)
41580_2013_BFnrm3486_MOESM395_ESM.pdf
Supplementary Information S5 | Characteristics and features of various platforms for assembling DNA encoding TALE repeat arrays (PDF 125 kb)
Related links
Related links
FURTHER INFORMATION
Rights and permissions
About this article
Cite this article
Joung, J., Sander, J. TALENs: a widely applicable technology for targeted genome editing. Nat Rev Mol Cell Biol 14, 49–55 (2013). https://doi.org/10.1038/nrm3486
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3486
This article is cited by
-
The regulation of plasma gelsolin by DNA methylation in ovarian cancer chemo-resistance
Journal of Ovarian Research (2024)
-
CRISPR/Cas-Mediated Genome Editing for Sugarcane Improvement
Sugar Tech (2024)
-
Crop bioengineering via gene editing: reshaping the future of agriculture
Plant Cell Reports (2024)
-
Identification of a genomic DNA sequence that quantitatively modulates KLF1 transcription factor expression in differentiating human hematopoietic cells
Scientific Reports (2023)
-
Cellular agriculture for milk bioactive production
Nature Reviews Bioengineering (2023)