Abstract
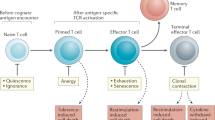
The proliferation of a few antigen-reactive lymphocytes into a large population of effector cells is a fundamental property of adaptive immunity. The cell division that fuels this process is driven by signals from antigen, co-stimulatory molecules and growth factor receptors, and is controlled by the cyclin-dependent kinase (CDK) cascade. In this Opinion article, we discuss how the CDK cascade provides one potential link between cell division and differentiation through the phosphorylation of immunologically relevant transcription factors, and how components of this pathway might ultimately participate in the decision between tolerance and immunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Butz, E. A. & Bevan, M. J. Massive expansion of antigen-specific CD8+ T cells during an acute virus infection. Immunity 8, 167–175 (1998).
Sourdive, D. J. et al. Conserved T cell receptor repertoire in primary and memory CD8 T cell responses to an acute viral infection. J. Exp. Med. 188, 71–82 (1998).
Jenkins, M. K. & Moon, J. J. The role of naive T cell precursor frequency and recruitment in dictating immune response magnitude. J. Immunol. 188, 4135–4140 (2012).
Lyons, A. B. & Parish, C. R. Determination of lymphocyte division by flow cytometry. J. Immunol. Methods 171, 131–137 (1994).
Wells, A., Gudmundsdottir, H. & Turka, L. Following the fate of individual T cells throughout activation and clonal expansion. Signals from T cell receptor and CD28 differentially regulate the induction and duration of a proliferative response. J. Clin. Invest. 100, 3173–3183 (1997).
Kurts, C., Kosaka, H., Carbone, F. R., Miller, J. F. & Heath, W. R. Class I-restricted cross-presentation of exogenous self-antigens leads to deletion of autoreactive CD8+ T cells. J. Exp. Med. 186, 239–245 (1997).
Gudmundsdottir, H., Wells, A. & Turka, L. Dynamics and requirements of T cell clonal expansion in vivo at the single-cell level: effector function is linked to proliferative capacity. J. Immunol. 162, 5212–5223 (1999).
Lim, S. & Kaldis, P. Cdks, cyclins and CKIs: roles beyond cell cycle regulation. Development 140, 3079–3093 (2013).
van den Heuvel, S. & Harlow, E. Distinct roles for cyclin-dependent kinases in cell cycle control. Science 262, 2050–2054 (1993).
Polyak, K. et al. Cloning of p27Kip1, a cyclin-dependent kinase inhibitor and a potential mediator of extracellular antimitogenic signals. Cell 78, 59–66 (1994).
Diril, M. K. et al. Cyclin-dependent kinase 1 (Cdk1) is essential for cell division and suppression of DNA re-replication but not for liver regeneration. Proc. Natl Acad. Sci. USA 109, 3826–3831 (2012).
Santamaría, D. et al. Cdk1 is sufficient to drive the mammalian cell cycle. Nature 448, 811–815 (2007).
Ortega, S. et al. Cyclin-dependent kinase 2 is essential for meiosis but not for mitotic cell division in mice. Nature Genet. 35, 25–31 (2003).
Berthet, C., Aleem, E., Coppola, V., Tessarollo, L. & Kaldis, P. Cdk2 knockout mice are viable. Curr. Biol. 13, 1775–1785 (2003).
Tsutsui, T. et al. Targeted disruption of CDK4 delays cell cycle entry with enhanced p27Kip1 activity. Mol. Cell. Biol. 19, 7011–7019 (1999).
Rane, S. G. et al. Loss of Cdk4 expression causes insulin-deficient diabetes and Cdk4 activation results in β-islet cell hyperplasia. Nature Genet. 22, 44–52 (1999).
Malumbres, M. et al. Mammalian cells cycle without the D-type cyclin-dependent kinases Cdk4 and Cdk6. Cell 118, 493–504 (2004).
Sicinska, E. et al. Requirement for cyclin D3 in lymphocyte development and T cell leukemias. Cancer Cell 4, 451–461 (2003).
Hu, M. G. et al. A requirement for cyclin-dependent kinase 6 in thymocyte development and tumorigenesis. Cancer Res. 69, 810–818 (2009).
Hu, M. G. et al. CDK6 kinase activity is required for thymocyte development. Blood 117, 6120–6131 (2011).
Rowell, E. & Wells, A. The role of cyclin-dependent kinases in T-cell development, proliferation, and function. Crit. Rev. Immunol. 26, 189–212 (2006).
Sharpless, N. E. et al. Loss of p16Ink4a with retention of p19Arf predisposes mice to tumorigenesis. Nature 413, 86–91 (2001).
Berthet, C. et al. Combined loss of Cdk2 and Cdk4 results in embryonic lethality and Rb hypophosphorylation. Dev. Cell 10, 563–573 (2006).
Barrière, C. et al. Mice thrive without Cdk4 and Cdk2. Mol. Oncol. 1, 72–83 (2007).
Hasbold, J., Lyons, A. B., Kehry, M. R. & Hodgkin, P. D. Cell division number regulates IgG1 and IgE switching of B cells following stimulation by CD40 ligand and IL-4. Eur. J. Immunol. 28, 1040–1051 (1998).
Gett, A. & Hodgkin, P. Cell division regulates the T cell cytokine repertoire, revealing a mechanism underlying immune class regulation. Proc. Natl Acad. Sci. USA 95, 9488–9493 (1998).
Gudmundsdottir, H. & Turka, L. A closer look at homeostatic proliferation of CD4+ T cells: costimulatory requirements and role in memory formation. J. Immunol. 167, 3699–3707 (2001).
Bird, J. et al. Helper T cell differentiation is controlled by the cell cycle. Immunity 9, 229–237 (1998).
Wells, A., Walsh, M., Sankaran, D. & Turka, L. T cell effector function and anergy avoidance are quantitatively linked to cell division. J. Immunol. 165, 2432–2443 (2000).
Schwartz, R. T cell anergy. Annu. Rev. Immunol. 21, 305–334 (2003).
Powell, J., Lerner, C. & Schwartz, R. Inhibition of cell cycle progression by rapamycin induces T cell clonal anergy even in the presence of costimulation. J. Immunol. 162, 2775–2784 (1999).
Vanasek, T., Khoruts, A., Zell, T. & Mueller, D. Antagonistic roles for CTLA-4 and the mammalian target of rapamycin in the regulation of clonal anergy: enhanced cell cycle progression promotes recall antigen responsiveness. J. Immunol. 167, 5636–5644 (2001).
Jackson, S., DeLoose, A. & Gilbert, K. Induction of anergy in Th1 cells associated with increased levels of cyclin-dependent kinase inhibitors p21Cip1 and p27Kip1. J. Immunol. 166, 952–958 (2001).
Wells, A. Cyclin-dependent kinases: molecular switches controlling anergy and potential therapeutic targets for tolerance. Semin. Immunol. 19, 173–179 (2007).
Kanno, Y., Vahedi, G., Hirahara, K., Singleton, K. & O'Shea, J. J. Transcriptional and epigenetic control of T helper cell specification: molecular mechanisms underlying commitment and plasticity. Annu. Rev. Immunol. 30, 707–731 (2012).
Wells, A. D. New insights into the molecular basis of T cell anergy: anergy factors, avoidance sensors, and epigenetic imprinting. J. Immunol. 182, 7331–7341 (2009).
Chang, J. T. et al. Asymmetric proteasome segregation as a mechanism for unequal partitioning of the transcription factor T-bet during T lymphocyte division. Immunity 34, 492–504 (2011).
Chang, J. T. et al. Asymmetric T lymphocyte division in the initiation of adaptive immune responses. Science 315, 1687–1691 (2007).
Ciocca, M. L., Barnett, B. E., Burkhardt, J. K., Chang, J. T. & Reiner, S. L. Cutting edge: Asymmetric memory T cell division in response to rechallenge. J. Immunol. 188, 4145–4148 (2012).
Grimmler, M. et al. Cdk-inhibitory activity and stability of p27Kip1 are directly regulated by oncogenic tyrosine kinases. Cell 128, 269–280 (2007).
Shin, I. et al. PKB/Akt mediates cell-cycle progression by phosphorylation of p27Kip1 at threonine 157 and modulation of its cellular localization. Nature Med. 8, 1145–1152 (2002).
Liang, J. et al. PKB/Akt phosphorylates p27, impairs nuclear import of p27 and opposes p27-mediated G1 arrest. Nature Med. 8, 1153–1160 (2002).
Alevizopoulos, K., Catarin, B., Vlach, J. & Amati, B. A novel function of adenovirus E1A is required to overcome growth arrest by the CDK2 inhibitor p27Kip1. EMBO J. 17, 5987–5997 (1998).
Montagnoli, A. et al. Ubiquitination of p27 is regulated by Cdk-dependent phosphorylation and trimeric complex formation. Genes Dev. 13, 1181–1189 (1999).
Appleman, L. J., Berezovskaya, A., Grass, I. & Boussiotis, V. A. CD28 costimulation mediates T cell expansion via IL-2-independent and IL-2-dependent regulation of cell cycle progression. J. Immunol. 164, 144–151 (2000).
Boussiotis, V. et al. p27kip1 functions as an anergy factor inhibiting interleukin 2 transcription and clonal expansion of alloreactive human and mouse helper T lymphocytes. Nature Med. 6, 290–297 (2000).
Rowell, E., Wang, L., Hancock, W. & Wells, A. The cyclin-dependent kinase inhibitor p27kip1 is required for transplantation tolerance induced by costimulatory blockade. J. Immunol. 177, 5169–5176 (2006).
Rowell, E., Walsh, M. & Wells, A. Opposing roles for the cyclin-dependent kinase inhibitor p27kip1 in the control of CD4+ T cell proliferation and effector function. J. Immunol. 174, 3359–3368 (2005).
Li, L., Iwamoto, Y., Berezovskaya, A. & Boussiotis, V. A pathway regulated by cell cycle inhibitor p27Kip1 and checkpoint inhibitor Smad3 is involved in the induction of T cell tolerance. Nature Immunol. 7, 1157–1165 (2006).
Besson, A., Gurian-West, M., Schmidt, A., Hall, A. & Roberts, J. M. p27Kip1 modulates cell migration through the regulation of RhoA activation. Genes Dev. 18, 862–876 (2004).
Chunder, N., Wang, L., Chen, C., Hancock, W. W. & Wells, A. D. Cyclin-dependent kinase 2 controls peripheral immune tolerance. J. Immunol. 189, 5659–5666 (2012).
Dhavan, R. & Tsai, L. H. A decade of CDK5. Nature Rev. Mol. Cell. Biol. 2, 749–759 (2001).
Ohshima, T. et al. Targeted disruption of the cyclin-dependent kinase 5 gene results in abnormal corticogenesis, neuronal pathology and perinatal death. Proc. Natl Acad. Sci. USA 93, 11173–11178 (1996).
Pareek, T. K. et al. Cyclin-dependent kinase 5 activity is required for T cell activation and induction of experimental autoimmune encephalomyelitis. J. Exp. Med. 207, 2507–2519 (2010).
Berthet, C. et al. Hematopoiesis and thymic apoptosis are not affected by the loss of Cdk2. Mol. Cell. Biol. 27, 5079–5089 (2007).
Schwartz, R. H. A cell culture model for T lymphocyte clonal anergy. Science 248, 1349–1356 (1990).
Jatzek, A. et al. p27Kip1 negatively regulates the magnitude and persistence of CD4 T cell memory. J. Immunol. 189, 5119–5128 (2012).
Singh, A. et al. Regulation of memory CD8 T-cell differentiation by cyclin-dependent kinase inhibitor p27Kip1. Mol. Cell. Biol. 30, 5145–5159 (2010).
Oestreich, K. J., Mohn, S. E. & Weinmann, A. S. Molecular mechanisms that control the expression and activity of Bcl-6 in TH1 cells to regulate flexibility with a TFH-like gene profile. Nature Immunol. 13, 405–411 (2012).
Arias, C. F. et al. p21CIP1/WAF1 controls proliferation of activated/memory T cells and affects homeostasis and memory T cell responses. J. Immunol. 178, 2296–2306 (2007).
Jatzek, A., Marie Tejera, M., Plisch, E. H., Fero, M. L. & Suresh, M. T-cell intrinsic and extrinsic mechanisms of p27Kip1 in the regulation of CD8 T-cell memory. Immunol. Cell Biol. 91, 120–129 (2013).
Kim, M. V., Ouyang, W., Liao, W., Zhang, M. Q. & Li, M. O. The transcription factor Foxo1 controls central-memory CD8+ T cell responses to infection. Immunity 39, 286–297 (2013).
Tejera, M. M., Kim, E. H., Sullivan, J. A., Plisch, E. H. & Suresh, M. FoxO1 controls effector-to-memory transition and maintenance of functional CD8 T cell memory. J. Immunol. 191, 187–199 (2013).
Brunet, A. et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96, 857–868 (1999).
Yuan, Z. et al. Activation of FOXO1 by Cdk1 in cycling cells and postmitotic neurons. Science 319, 1665–1668 (2008).
Hedrick, S. M., Hess Michelini, R., Doedens, A. L., Goldrath, A. W. & Stone, E. L. FOXO transcription factors throughout T cell biology. Nature Rev. Immunol. 12, 649–661 (2012).
Lawson, B. R. et al. Deficiency of the cyclin kinase inhibitor p21(WAF-1/CIP-1) promotes apoptosis of activated/memory T cells and inhibits spontaneous systemic autoimmunity. J. Exp. Med. 199, 547–557 (2004).
Balomenos, D. et al. The cell cycle inhibitor p21 controls T-cell proliferation and sex-linked lupus development. Nature Med. 6, 171–176 (2000).
Santiago-Raber, M. L. et al. Role of cyclin kinase inhibitor p21 in systemic autoimmunity. J. Immunol. 167, 4067–4074 (2001).
Selma Dagtas, A. & Gilbert, K. M. p21Cip1 up-regulated during histone deacetylase inhibitor-induced CD4+ T-cell anergy selectively associates with mitogen-activated protein kinases. Immunology 129, 589–599 (2010).
Iglesias, M. et al. p27Kip1 inhibits systemic autoimmunity through the control of Treg cell activity and differentiation. Arthritis Rheum. 65, 343–354 (2013).
Larsen, C. P. et al. Long-term acceptance of skin and cardiac allografts after blocking CD40 and CD28 pathways. Nature 381, 434–438 (1996).
Sakaguchi, S., Wing, K., Onishi, Y., Prieto-Martin, P. & Yamaguchi, T. Regulatory T cells: how do they suppress immune responses? Int. Immunol. 21, 1105–1111 (2009).
Killebrew, J. R. et al. A self-reactive TCR drives the development of Foxp3+ regulatory T cells that prevent autoimmune disease. J. Immunol. 187, 861–869 (2011).
Morawski, P. A., Mehra, P., Chen, C., Bhatti, T. & Wells, A. D. Foxp3 protein stability is regulated by cyclin-dependent kinase 2. J. Biol. Chem. 288, 24494–24502 (2013).
Maddika, S. et al. Akt-mediated phosphorylation of CDK2 regulates its dual role in cell cycle progression and apoptosis. J. Cell. Sci. 121, 979–988 (2008).
Patterson, S. J. et al. Cutting edge: PHLPP regulates the development, function, and molecular signaling pathways of regulatory T cells. J. Immunol. 186, 5533–5537 (2011).
König, S. et al. First insight into the kinome of human regulatory T cells. PLoS ONE 7, e40896 (2012).
Li, L. et al. CD4+CD25+ regulatory T-cell lines from human cord blood have functional and molecular properties of T-cell anergy. Blood 106, 3068–3073 (2005).
Oberg, H.-H. et al. Differential but direct abolishment of human regulatory T cell suppressive capacity by various TLR2 ligands. J. Immunol. 184, 4733–4740 (2010).
Chen, W. et al. Conversion of peripheral CD4+CD25− naive T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. J. Exp. Med. 198, 1875–1886 (2003).
Krystof, V. & Uldrijan, S. Cyclin-dependent kinase inhibitors as anticancer drugs. Curr. Drug Targets 11, 291–302 (2010).
Gallorini, M., Cataldi, A. & di Giacomo, V. Cyclin-dependent kinase modulators and cancer therapy. BioDrugs 26, 377–391 (2012).
Yoshida, H. et al. CDK inhibitors suppress Th17 and promote iTreg differentiation, and ameliorate experimental autoimmune encephalomyelitis in mice. Biochem. Biophys. Res. Commun. 435, 378–384 (2013).
Li, L., Wang, H., Kim, J. S., Pihan, G. & Boussiotis, V. The cyclin dependent kinase inhibitor (R)-roscovitine prevents alloreactive T cell clonal expansion and protects against acute GvHD. Cell Cycle 8, 1794–1802 (2009).
Zoja, C. et al. Cyclin-dependent kinase inhibition limits glomerulonephritis and extends lifespan of mice with systemic lupus. Arthritis Rheum. 56, 1629–1637 (2007).
Pezzotta, A. et al. Effect of seliciclib (CYC202, R-roscovitine) on lymphocyte alloreactivity and acute kidney allograft rejection in rat. Transplantation 85, 1476–1482 (2008).
Wesierska-Gadek, J. & Krystof, V. Selective cyclin-dependent kinase inhibitors discriminating between cell cycle and transcriptional kinases: future reality or utopia? Ann. NY Acad. Sci. 1171, 228–241 (2009).
Whittaker, S. R. et al. The cyclin-dependent kinase inhibitor seliciclib (R-roscovitine; CYC202) decreases the expression of mitotic control genes and prevents entry into mitosis. Cell Cycle 6, 3114–3131 (2007).
Jorda, R. et al. Pyrazolo[4,3-d]pyrimidine bioisostere of roscovitine: evaluation of a novel selective inhibitor of cyclin-dependent kinases with antiproliferative activity. J. Med. Chem. 54, 2980–2993 (2011).
Legraverend, M. & Grierson, D. S. The purines: potent and versatile small molecule inhibitors and modulators of key biological targets. Bioorg. Med. Chem. 14, 3987–4006 (2006).
Blagosklonny, M. & Pardee, A. The restriction point of the cell cycle. Cell Cycle 1, 103–110 (2002).
Kohli, R. M. & Zhang, Y. TET enzymes, TDG and the dynamics of DNA demethylation. Nature 502, 472–479 (2013).
Contreras, A. et al. The dynamic mobility of histone H1 is regulated by cyclin/CDK phosphorylation. Mol. Cell. Biol. 23, 8626–8636 (2003).
Chen, S. et al. Cyclin-dependent kinases regulate epigenetic gene silencing through phosphorylation of EZH2. Nature Cell Biol. 12, 1108–1114 (2010).
Lavoie, G. & St-Pierre, Y. Phosphorylation of human DNMT1: implication of cyclin-dependent kinases. Biochem. Biophys. Res. Commun. 409, 187–192 (2011).
Liakopoulos, D., Kusch, J., Grava, S., Vogel, J. & Barral, Y. Asymmetric loading of Kar9 onto spindle poles and microtubules ensures proper spindle alignment. Cell 112, 561–574 (2003).
Cheung, Z. H. & Ip, N. Y. Cdk5: a multifaceted kinase in neurodegenerative diseases. Trends Cell Biol. 22, 169–175 (2012).
Matsuura, I. et al. Cyclin-dependent kinases regulate the antiproliferative function of Smads. Nature 430, 226–231 (2004).
Vanden Bush, T. J. & Bishop, G. A. CDK-mediated regulation of cell functions via c-Jun phosphorylation and AP-1 activation. PLoS ONE 6, e19468 (2011).
Banchio, C., Schang, L. M. & Vance, D. E. Phosphorylation of Sp1 by cyclin-dependent kinase 2 modulates the role of Sp1 in CTP:phosphocholine cytidylyltransferase-α regulation during the S phase of the cell cycle. J. Biol. Chem. 279, 40220–40226 (2004).
Perkins, N. et al. Regulation of NF-κB by cyclin-dependent kinases associated with the p300 coactivator. Science 275, 523–527 (1997).
Chen, E. & Li, C. Association of Cdk2/cyclin E and NF-κB complexes at G1/S phase. Biochem. Biophys. Res. Commun. 249, 728–734 (1998).
Guo, H. & Friedman, A. D. Phosphorylation of RUNX1 by cyclin-dependent kinase reduces direct interaction with HDAC1 and HDAC3. J. Biol. Chem. 286, 208–215 (2011).
Levy, D. E. & Darnell, J. E. Stats: transcriptional control and biological impact. Nature Rev. Mol. Cell. Biol. 3, 651–662 (2002).
Bancerek, J. et al. CDK8 kinase phosphorylates transcription factor STAT1 to selectively regulate the interferon response. Immunity 38, 250–262 (2013).
Kerdiles, Y. M. et al. Foxo transcription factors control regulatory T cell development and function. Immunity 33, 890–904 (2010).
Harada, Y. et al. Transcription factors Foxo3a and Foxo1 couple the E3 ligase Cbl-b to the induction of Foxp3 expression in induced regulatory T cells. J. Exp. Med. 207, 1381–1391 (2010).
Tzivion, G., Dobson, M. & Ramakrishnan, G. FoxO transcription factors; Regulation by AKT and 14-3-3 proteins. Biochim. Biophys. Acta 1813, 1938–1945 (2011).
Huang, H., Regan, K. M., Lou, Z., Chen, J. & Tindall, D. J. CDK2-dependent phosphorylation of FOXO1 as an apoptotic response to DNA damage. Science 314, 294–297 (2006).
Medema, R. H., Kops, G. J., Bos, J. L. & Burgering, B. M. AFX-like Forkhead transcription factors mediate cell-cycle regulation by Ras and PKB through p27kip1. Nature 404, 782–787 (2000).
Acknowledgements
A.D.W. is a member of the Biesecker Pediatric Liver Disease Center at The Children's Hospital of Philadelphia, Pennsylvania, USA. Work relevant to this article by A.D.W. was supported by US National Institutes of Health grants AI054643 and AI070807, and work by P.A.M. was supported by a Goldie Simon Preceptorship Award sponsored by the Lupus Foundation of America Philadelphia Tri-State Chapter, Pennsylvania, USA, and the Pennsylvania Department of Health, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Anergy
-
The functional non-responsiveness of antigen-specific T cells.
- Autoimmune accelerator locus
-
(Yaa). A duplication of a 4 Mbp segment of the Y chromosome of inbred BXSB mice containing 19 genes, including Tlr7, that leads to the spontaneous development of systemic autoimmunity similar to systemic lupus erythematosus.
- Bioisosteres
-
In drug design, compounds that have highly similar properties and structures, but varied efficacy, activity or toxicity as a result of minor chemical substitutions.
- DN3 to DN4 transition
-
The transition during thymocyte maturation during which CD4−CD8− double-negative cells undergo T cell receptor-β (TCRβ) rearrangement and downregulate CD25 expression.
- G0 phase
-
(Resting phase). The period of the cell cycle during which the cell is quiescent that lasts until the initiation of cell division.
- G1 phase
-
(Gap1 phase). The phase of the cell cycle during which the cell grows and carries out large amounts of protein synthesis.
- Interphase
-
The period representing the majority of the cell cycle including G1, S and G2 phases, during which the cell increases in size and replicates its DNA.
- Interphase CDK
-
Cyclin-dependent kinases that are active during the interphase (G1, S and G2 phases) of the cell cycle, including CDK2, CDK4 and CDK6.
- Nuclear mediator complex
-
The complex of multiple nuclear proteins that is required to enhance RNA polymerase II-mediated gene transcription.
- RNA polymerase II
-
(RNA Pol II). A 550 kDa, 12-subunit eukaryotic enzyme that is required for the initiation of DNA transcription.
- S phase
-
(Synthesis phase). The phase of the cell cycle during which the genomic DNA is replicated.
Rights and permissions
About this article
Cite this article
Wells, A., Morawski, P. New roles for cyclin-dependent kinases in T cell biology: linking cell division and differentiation. Nat Rev Immunol 14, 261–270 (2014). https://doi.org/10.1038/nri3625
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3625
This article is cited by
-
Synthesis, docking, MD simulation, ADMET, drug likeness, and DFT studies of novel furo[2,3-b]indol-3a-ol as promising Cyclin-dependent kinase 2 inhibitors
Scientific Reports (2024)
-
Infiltrating T lymphocytes in the tumor microenvironment of small cell lung cancer: a state of knowledge review
Journal of Cancer Research and Clinical Oncology (2022)
-
9p21 loss confers a cold tumor immune microenvironment and primary resistance to immune checkpoint therapy
Nature Communications (2021)
-
Immunomodulation by anticancer cell cycle inhibitors
Nature Reviews Immunology (2020)
-
Recent advances in the clinical development of immune checkpoint blockade therapy
Cellular Oncology (2019)