Key Points
-
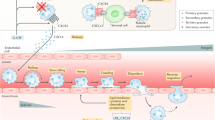
Neutrophils are primarily produced in the bone marrow through a process that is highly dependent on granulocyte colony-stimulating factor (G-CSF). However, changes that are associated with chronic inflammation, such as psychosocial stress, hyperlipidaemia and hyperglycaemia, can stimulate extramedullary neutrophil production and increase neutrophil numbers in the blood.
-
The function of circulating neutrophils is modified by the presence of risk factors for chronic inflammation. Ageing of the host impairs neutrophil migration, whereas metabolic changes can prime neutrophils for activation.
-
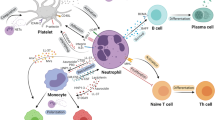
The interaction of neutrophils and platelets along the luminal aspect of sites of inflammation is essential for fine-tuning neutrophil recruitment. Neutrophil secretory products, partly in conjunction with platelet-derived proteins, deliver molecular cues to stimulate secondary monocyte recruitment.
-
Neutrophils and macrophages form an intricate partnership at sites of damage. During the inflammation phase, neutrophils enhance macrophage activities that are aimed at eliminating the initiating stimulus. During the resolution phase, dying neutrophils convey an important signal that reprogrammes macrophages to promote healing.
-
Neutrophils have recently been shown to be important in chronic inflammatory diseases, including neurodegenerative diseases and atherosclerosis. Relevant mechanisms include their interplay with the monocyte lineage — for example, the enhancement of monocyte recruitment and the activation of macrophages — and their direct pro-inflammatory effects, such as release of reactive oxygen species and neutrophil extracellular traps (NETs), and neutrophil-derived proteolytic activity.
-
Neutrophil-instructed chronic inflammation can be targeted by inhibiting NET release or by breaking down NETs, by translating recent findings about neutrophil recruitment during chronic inflammation and by promoting the clearance of apoptotic neutrophils. In addition, neutrophil-derived microparticles that elicit anti-inflammatory responses represent an innovative strategy to reduce inflammation.
Abstract
Traditionally, neutrophils have been acknowledged to be the first immune cells that are recruited to an inflamed tissue and have mainly been considered in the context of acute inflammation. By contrast, their importance during chronic inflammation has been studied in less depth. This Review aims to summarize our current understanding of the roles of neutrophils in chronic inflammation, with a focus on how they communicate with other immune and non-immune cells within tissues. We also scrutinize the roles of neutrophils in wound healing and the resolution of inflammation, and finally, we outline emerging therapeutic strategies that target neutrophils.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mayadas, T. N., Cullere, X. & Lowell, C. A. The multifaceted functions of neutrophils. Annu. Rev. Pathol. 9, 181–218 (2014).
de Oliveira, S., Rosowskim, E. E. & Huttenlocher, A. Neutrophil migration in infection and wound repair: going forward in reverse. Nat. Rev. Immunol. 16, 378–391 (2016).
Coffelt, S. B., Wellenstein, M. D. & de Visser, K. E. Neutrophils in cancer: neutral no more. Nat. Rev. Cancer 16, 431–446 (2016).
Dancey, J. T., Deubelbeiss, K. A., Harker, L. A. & Finch, C. A. Neutrophil kinetics in man. J. Clin. Invest. 58, 705–715 (1976).
Semerad, C. L., Liu, F., Gregory, A. D., Stumpf, K. & Link, D. C. G-CSF is an essential regulator of neutrophil trafficking from the bone marrow to the blood. Immunity 17, 413–423 (2002).
Casanova-Acebes, M. et al. Rhythmic modulation of the hematopoietic niche through neutrophil clearance. Cell 153, 1025–1035 (2013).
Köhler, A. et al. G-CSF-mediated thrombopoietin release triggers neutrophil motility and mobilization from bone marrow via induction of Cxcr2 ligands. Blood 117, 4349–4357 (2011).
Bajrami, B. et al. G-CSF maintains controlled neutrophil mobilization during acute inflammation by negatively regulating CXCR2 signaling. J. Exp. Med. 213, 1999–2018 (2016).
Stark, M. A. et al. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity 22, 285–294 (2005).
von Vietinghoff, S. & Ley, K. Homeostatic regulation of blood neutrophil counts. J. Immunol. 181, 5183–5188 (2008).
Hong, C. et al. Coordinate regulation of neutrophil homeostasis by liver X receptors in mice. J. Clin. Invest. 122, 337–347 (2012).
Westerterp, M. et al. Regulation of hematopoietic stem and progenitor cell mobilization by cholesterol efflux pathways. Cell Stem Cell 11, 195–206 (2012).
Devi, S. et al. Neutrophil mobilization via plerixafor-mediated CXCR4 inhibition arises from lung demargination and blockade of neutrophil homing to the bone marrow. J. Exp. Med. 210, 2321–2336 (2013).
Granick, J. L. et al. Staphylococcus aureus recognition by hematopoietic stem and progenitor cells via TLR2/MyD88/PGE2 stimulates granulopoiesis in wounds. Blood 122, 1770–1778 (2013).
Swirski, F. K. et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 325, 612–616 (2009).
Manz, M. G. & Boettcher, S. Emergency granulopoiesis. Nat. Rev. Immunol. 14, 302–314 (2014).
Boettcher, S. et al. Endothelial cells translate pathogen signals into G-CSF-driven emergency granulopoiesis. Blood 124, 1393–1403 (2014).
Nagareddy, P. R. et al. Hyperglycemia promotes myelopoiesis and impairs the resolution of atherosclerosis. Cell Metab. 17, 695–708 (2013).
Drechsler, M., Megens, R. T., van Zandvoort, M., Weber, C. & Soehnlein, O. Hyperlipidemia-triggered neutrophilia promotes early atherosclerosis. Circulation 122, 1837–1845 (2010).
Yvan-Charvet, L. et al. ATP-binding cassette transporters and HDL suppress hematopoietic stem cell proliferation. Science 328, 1689–1693 (2010).
Heidt, T. et al. Chronic variable stress activates hematopoietic stem cells. Nat. Med. 20, 754–758 (2014). In this study, the authors link psychosocial stress to enhanced myeloid cell production and the development of chronic arterial inflammation.
Tseng, C. W. & Liu, G. Y. Expanding roles of neutrophils in aging hosts. Curr. Opin. Immunol. 29, 43–48 (2014).
Sapey, E. et al. Phosphoinositide 3-kinase inhibition restores neutrophil accuracy in the elderly: toward targeted treatments for immunosenescence. Blood 123, 239–248 (2014).
Qian, F. et al. Reduced bioenergetics and Toll-like receptor 1 function in human polymorphonuclear leukocytes in aging. Aging (Albany NY) 6, 131–139 (2014).
Wong, S. L. et al. Diabetes primes neutrophils to undergo NETosis, which impairs wound healing. Nat. Med. 21, 815–819 (2015).
Araujo, F. B., Barbosa, D. S., Hsin, C. Y., Maranhão, R. C. & Abdalla, D. S. Evaluation of oxidative stress in patients with hyperlipidemia. Atherosclerosis 117, 61–71 (1995).
Mazor, R. et al. Primed polymorphonuclear leukocytes constitute a possible link between inflammation and oxidative stress in hyperlipidemic patients. Atherosclerosis 197, 937–943 (2008).
Rossaint, J. & Zarbock, A. Platelets in leucocyte recruitment and function. Cardiovasc. Res. 107, 386–395 (2015).
Sreeramkumar, V. et al. Neutrophils scan for activated platelets to initiate inflammation. Science 346, 1234–1238 (2014).
Abdulnour, R. E. et al. Maresin 1 biosynthesis during platelet–neutrophil interactions is organ-protective. Proc. Natl Acad. Sci. USA 111, 16526–16531 (2014).
Rossaint, J. et al. Synchronized integrin engagement and chemokine activation is crucial in neutrophil extracellular trap-mediated sterile inflammation. Blood 123, 2573–2584 (2014).
Megens, R. T. et al. Presence of luminal neutrophil extracellular traps in atherosclerosis. Thromb. Haemost. 107, 597–598 (2012).
Quillard, T. et al. TLR2 and neutrophils potentiate endothelial stress, apoptosis and detachment: implications for superficial erosion. Eur. Heart J. 36, 1394–1404 (2015).
Grommes, J. et al. Disruption of platelet-derived chemokine heteromers prevents neutrophil extravasation in acute lung injury. Am. J. Respir. Crit. Care Med. 185, 628–636 (2012).
Hartwig, H. et al. Platelet-derived PF4 reduces neutrophil apoptosis following arterial occlusion. Thromb. Haemost. 111, 562–564 (2014).
Mokart, D. et al. Monocyte deactivation in neutropenic acute respiratory distress syndrome patients treated with granulocyte colony-stimulating factor. Crit. Care. 12, R17 (2008).
Gallin, J. I. et al. Human neutrophil-specific granule deficiency: a model to assess the role of neutrophil-specific granules in the evolution of the inflammatory response. Blood 59, 1317–1329 (1982).
Shiohara, M. et al. Phenotypic and functional alterations of peripheral blood monocytes in neutrophil-specific granule deficiency. J. Leukoc. Biol. 75, 190–197 (2004).
Soehnlein, O., Lindbom, L. & Weber, C. Mechanisms underlying neutrophil-mediated monocyte recruitment. Blood 114, 4613–4623 (2009).
Ortega-Gomez, A. et al. Cathepsin G controls arterial but not venular myeloid cell recruitment. Circulation 134, 1176–1188 (2016).
Alard, J. E. et al. Recruitment of classical monocytes can be inhibited by disturbing heteromers of neutrophil HNP1 and platelet CCL5. Sci. Transl Med. 7, 317ra196 (2015). References 40 and 41 provide novel mechanisms by which platelets and neutrophils work together to recruit inflammatory monocytes, and they establish therapeutic strategies for the inhibition of monocyte recruitment.
Wantha, S. et al. Neutrophil-derived cathelicidin promotes adhesion of classical monocytes. Circ. Res. 112, 792–801 (2013).
Lim, K. et al. Neutrophil trails guide influenza-specific CD8+ T cells in the airways. Science 349, aaa4352 (2015).
Soehnlein, O. et al. Neutrophil primary granule proteins HBP and HNP1-3 boost bacterial phagocytosis by human and murine macrophages. J. Clin. Invest. 118, 3491–3502 (2008).
Venereau, E. et al. Mutually exclusive redox forms of HMGB1 promote cell recruitment or proinflammatory cytokine release. J. Exp. Med. 209, 1519–1528 (2012).
Vogl, T. et al. Mrp8 and Mrp14 are endogenous activators of Toll-like receptor 4, promoting lethal, endotoxin-induced shock. Nat. Med. 13, 1042–1049 (2007).
Warnatsch, A., Ioannou, M., Wang, Q. & Papayannopoulos, V. Inflammation. Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science 349, 316–320 (2015).
Maianski, N. A., Mul, F. P., van Buul, J. D., Roos, D. & Kuijpers, T. W. Granulocyte colony-stimulating factor inhibits the mitochondria-dependent activation of caspase-3 in neutrophils. Blood 99, 672–679 (2002).
van den Berg, J. M., Weyer, S., Weening, J. J., Roos, D. & Kuijpers, T. W. Divergent effects of tumor necrosis factor α on apoptosis of human neutrophils. J. Leukoc. Biol. 69, 467–473 (2001).
Ariel, A. et al. Apoptotic neutrophils and T cells sequester chemokines during immune response resolution through modulation of CCR5 expression. Nat. Immunol. 7, 1209–1216 (2006).
Schauer, C. et al. Aggregated neutrophil extracellular traps limit inflammation by degrading cytokines and chemokines. Nat. Med. 20, 511–517 (2014).
Drechsler, M. et al. Annexin A1 counteracts chemokine-induced arterial myeloid cell recruitment. Circ. Res. 116, 827–835 (2015).
Scannell, M. et al. Annexin-1 and peptide derivatives are released by apoptotic cells and stimulate phagocytosis of apoptotic neutrophils by macrophages. J. Immunol. 178, 4595–4605 (2007).
Poon, I. K., Lucas, C. D., Rossi, A. G. & Ravichandran, K. S. Apoptotic cell clearance: basic biology and therapeutic potential. Nat. Rev. Immunol. 14, 166–180 (2014).
A-Gonzalez, N. et al. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity 31, 245–258 (2009).
Mukundan, L. et al. PPAR-δ senses and orchestrates clearance of apoptotic cells to promote tolerance. Nat. Med. 15, 1266–1272 (2009).
Schrijvers, D. M., De Meyer, G. R., Kockx, M. M., Herman, A. G. & Martinet, W. Phagocytosis of apoptotic cells by macrophages is impaired in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 25, 1256–1261 (2005).
Morimoto, K., Janssen, W. J. & Terada, M. Defective efferocytosis by alveolar macrophages in IPF patients. Respir. Med. 106, 1800–1803 (2012).
Thorp, E. et al. Shedding of the Mer tyrosine kinase receptor is mediated by ADAM17 protein through a pathway involving reactive oxygen species, protein kinase Cδ, and p38 mitogen-activated protein kinase (MAPK). J. Biol. Chem. 286, 33335–33344 (2011).
Sather, S. et al. A soluble form of the Mer receptor tyrosine kinase inhibits macrophage clearance of apoptotic cells and platelet aggregation. Blood 109, 1026–1033 (2007).
Driscoll, W. S., Vaisar, T., Tang, J., Wilson, C. L. & Raines, E. W. Macrophage ADAM17 deficiency augments CD36-dependent apoptotic cell uptake and the linked anti-inflammatory phenotype. Circ. Res. 113, 52–61 (2013).
Friggeri, A. et al. HMGB1 inhibits macrophage activity in efferocytosis through binding to the αvβ3-integrin. Am. J. Physiol. Cell Physiol. 299, C1267–C1276 (2010).
Hampton, H. R., Bailey, J., Tomura, M., Brink, R. & Chtanova, T. Microbe-dependent lymphatic migration of neutrophils modulates lymphocyte proliferation in lymph nodes. Nat. Commun. 6, 7139 (2015).
Yang, C. W. & Unanue, E. R. Neutrophils control the magnitude and spread of the immune response in a thromboxane A2-mediated process. J. Exp. Med. 210, 375–387 (2013).
Gorlino, C. V. et al. Neutrophils exhibit differential requirements for homing molecules in their lymphatic and blood trafficking into draining lymph nodes. J. Immunol. 193, 1966–1974 (2014).
Hampton, H. R. & Chtanova, T. The lymph node neutrophil. Semin. Immunol. 28, 129–136 (2016).
Scapini, P. & Cassatella, M. A. Social networking of human neutrophils within the immune system. Blood 124, 710–719 (2014).
Hoenderdos, K. & Condliffe, A. The neutrophil in chronic obstructive pulmonary disease. Am. J. Respir. Cell. Mol. Biol. 48, 531–539 (2014).
Wright, H. L., Moots, R. J. & Edwards, S. W. The multifactorial role of neutrophils in rheumatoid arthritis. Nat. Rev. Rheumatol. 10, 593–601 (2014).
Lackey, D. E. & Olefsky, J. M. Regulation of metabolism by the innate immune system. Nat. Rev. Endocrinol. 12, 15–28 (2016).
Elgazar-Carmon, V., Rudich, A., Hadad, N. & Levy, R. Neutrophils transiently infiltrate intraabdominal fat early in the course of high-fat feeding. J. Lipid Res. 49, 1894–1903 (2008).
Chavey, C. et al. CXC ligand 5 is an adipose-tissue derived factor that links obesity to insulin resistance. Cell Metab. 9, 339–349 (2009).
Spite, M. et al. Deficiency of the leukotriene B4 receptor, BLT-1, protects against systemic insulin resistance in diet-induced obesity. J. Immunol. 187, 1942–1949 (2011).
Braster, Q. et al. Cathelicidin regulates myeloid cell accumulation in adipose tissue and promotes insulin resistance during obesity. Thromb. Haemost. 115, 1237–1239 (2016).
Wang, Q. et al. Myeloperoxidase deletion prevents high-fat diet-induced obesity and insulin resistance. Diabetes 63, 4172–4185 (2014).
Talukdar, S. et al. Neutrophils mediate insulin resistance in mice fed a high-fat diet through secreted elastase. Nat. Med. 18, 1407–1412 (2012).
Mansuy-Aubert, V. et al. Imbalance between neutrophil elastase and its inhibitor α1-antitrypsin in obesity alters insulin sensitivity, inflammation, and energy expenditure. Cell Metab. 17, 534–548 (2013).
Heppner, F. L., Ransohoff, R. M. & Becher, B. Immune attack: the role of inflammation in Alzheimer disease. Nat. Rev. Neurosci. 16, 358–372 (2015).
Baik, S. H. et al. Migration of neutrophils targeting amyloid plaques in Alzheimer's disease mouse model. Neurobiol. Aging 35, 1286–1292 (2014).
Zenaro, E. et al. Neutrophils promote Alzheimer's disease-like pathology and cognitive decline via LFA-1 integrin. Nat. Med. 21, 880–886 (2015). This study reveals the importance of neutrophils in a mouse model of Alzheimer disease and establishes the importance of neutrophil integrin α L (also known as LFA1), thus suggesting a therapeutic angle.
Gautam, N. et al. Heparin-binding protein (HBP/CAP37): a missing link in neutrophil-evoked alteration of vascular permeability. Nat. Med. 7, 1123–1127 (2001).
Allen, C. et al. Neutrophil cerebrovascular transmigration triggers rapid neurotoxicity through release of proteases associated with decondensed DNA. J. Immunol. 189, 381–392 (2012).
Siffrin, V. et al. In vivo imaging of partially reversible Th17 cell-induced neuronal dysfunction in the course of encephalomyelitis. Immunity 33, 424–436 (2010).
Ionita, M. G. et al. High neutrophil numbers in human carotid atherosclerotic plaques are associated with characteristics of rupture-prone lesions. Arterioscler. Thromb. Vasc. Biol. 30, 1842–1848 (2010).
Chèvre, R. et al. High-resolution imaging of intravascular atherogenic inflammation in live mice. Circ. Res. 114, 770–779 (2014).
Eriksson, E. E. Intravital microscopy on atherosclerosis in apolipoprotein e-deficient mice establishes microvessels as major entry pathways for leukocytes to advanced lesions. Circulation 124, 2129–2138 (2011).
Friedman, G. D., Klatsky, A. L. & Siegelaub, A. B. The leukocyte count as a predictor of myocardial infarction. N. Engl. J. Med. 290, 1275–1278 (1974).
Zernecke, A. et al. Protective role of CXC receptor 4/CXC ligand 12 unveils the importance of neutrophils in atherosclerosis. Circ. Res. 102, 209–217 (2008).
Döring, Y. et al. Lack of neutrophil-derived CRAMP reduces atherosclerosis in mice. Circ. Res. 110, 1052–1056 (2012).
Döring, Y. et al. Auto-antigenic protein-DNA complexes stimulate plasmacytoid dendritic cells to promote atherosclerosis. Circulation 125, 1673–1683 (2012).
Lande, R. et al. Neutrophils activate plasmacytoid dendritic cells by releasing self-DNA-peptide complexes in systemic lupus erythematosus. Sci. Transl Med. 3, 73ra19 (2011).
Diana, J. et al. Crosstalk between neutrophils, B-1a cells and plasmacytoid dendritic cells initiates autoimmune diabetes. Nat. Med. 19, 65–73 (2013).
Soehnlein, O., Ortega- Gómez, A., Döring, Y. & Weber, C. Neutrophil-macrophage interplay in atherosclerosis: protease-mediated cytokine processing versus NET release. Thromb. Haemost. 114, 866–867 (2015).
Montecucco, F. et al. The activation of the cannabinoid receptor type 2 reduces neutrophilic protease-mediated vulnerability in atherosclerotic plaques. Eur. Heart J. 33, 846–856 (2012).
Dovi, J. V., He, L. K. & DiPietro, L. A. Accelerated wound closure in neutrophil-depleted mice. J. Leukoc. Biol. 73, 448–455 (2003).
Stirling, D. P., Liu, S., Kubes, P. & Yong, V. W. Depletion of Ly6G/Gr-1 leukocytes after spinal cord injury in mice alters wound healing and worsens neurological outcome. J. Neurosci. 29, 753–764 (2009).
Kurimoto, T. et al. Neutrophils express oncomodulin and promote optic nerve regeneration. J. Neurosci. 33, 14816–14824 (2013).
Nishio, N., Okawa, Y., Sakurai, H. & Isobe, K. Neutrophil depletion delays wound repair in aged mice. Age (Dordr.) 30, 11–19 (2008).
Paris, A. J. et al. Neutrophils promote alveolar epithelial regeneration by enhancing type II pneumocyte proliferation in a model of acid-induced acute lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 311, L1062–L1075 (2016).
Neely, C. J. et al. Flagellin treatment prevents increased susceptibility to systemic bacterial infection after injury by inhibiting anti-inflammatory IL-10+ IL-12− neutrophil polarization. PLoS ONE 9, e85623 (2014).
Tsuda, Y. et al. Three different neutrophil subsets exhibited in mice with different susceptibilities to infection by methicillin-resistant Staphylococcus aureus. Immunity 21, 215–226 (2004).
Massena, S. et al. Identification and characterization of VEGF-A-responsive neutrophils expressing CD49d, VEGFR1, and CXCR4 in mice and humans. Blood 126, 2016–2026 (2015).
Chen, F. et al. Neutrophils prime a long-lived effector macrophage phenotype that mediates accelerated helminth expulsion. Nat. Immunol. 15, 938–946 (2014).
Soehnlein, O. & Lindbom, L. Phagocyte partnership during the onset and resolution of inflammation. Nat. Rev. Immunol. 10, 427–439 (2010).
Forbes, S. J. & Rosenthal, N. Preparing the ground for tissue regeneration: from mechanism to therapy. Nat. Med. 20, 857–869 (2014).
Benelli, R., Albini, A. & Noonan, D. Neutrophils and angiogenesis: potential initiators of the angiogenic cascade. Chem. Immunol. Allergy 83, 167–181 (2003).
Christoffersson, G. et al. VEGF-A recruits a proangiogenic MMP-9-delivering neutrophil subset that induces angiogenesis in transplanted hypoxic tissue. Blood 120, 4653–4662 (2012).
Gong, Y. & Koh, D. R. Neutrophils promote inflammatory angiogenesis via release of preformed VEGF in an in vivo corneal model. Cell Tissue Res. 339, 437–448 (2010).
Christoffersson, G. et al. Clinical and experimental pancreatic islet transplantation to striated muscle: establishment of a vascular system similar to that in native islets. Diabetes 59, 2569–2578 (2010).
Koczulla, R. et al. An angiogenic role for the human peptide antibiotic LL-37/hCAP-18. J. Clin. Invest. 111, 1665–1672 (2003).
Nozawa, H., Chiu, C. & Hanahan, D. Infiltrating neutrophils mediate the initial angiogenic switch in a mouse model of multistage carcinogenesis. Proc. Natl Acad. Sci. USA 103, 12493–12498 (2006).
Wilgus, T. A., Roy, S. & McDaniel, J. C. Neutrophils and wound repair: positive actions and negative reactions. Adv. Wound Care (New Rochelle) 2, 379–388 (2013).
Hristov, M. et al. Importance of CXC chemokine receptor 2 in the homing of human peripheral blood endothelial progenitor cells to sites of arterial injury. Circ. Res. 100, 590–597 (2007).
Soehnlein, O. et al. Neutrophil-derived cathelicidin protects from neointimal hyperplasia. Sci. Transl Med. 3, 103ra98 (2011).
Salvado, M. D., Di Gennaro, A., Lindbom, L., Agerberth, B. & Haeggström, J. Z. Cathelicidin LL-37 induces angiogenesis via PGE2–EP3 signaling in endothelial cells, in vivo inhibition by aspirin. Arterioscler. Thromb. Vasc. Biol. 33, 1965–1972 (2013).
Horckmans, M. et al. Neutrophils orchestrate post-myocardial infarction healing by polarizing macrophages towards a reparative phenotype. Eur. Heart J. 38, 187–197 (2016). On the basis of continued neutrophil depletion, this study establishes the beneficial effects of neutrophils during healing after myocardial infarction.
Schloss, M. J. et al. The time-of-day of myocardial infarction onset affects healing through oscillations in cardiac neutrophil recruitment. EMBO Mol. Med. 8, 937–948 (2016).
Zhang, R. et al. Dextran sulphate sodium increases splenic Gr1+CD11b+ cells which accelerate recovery from colitis following intravenous transplantation. Clin. Exp. Immunol. 164, 417–427 (2011).
Kühl, A. A. et al. Aggravation of different types of experimental colitis by depletion or adhesion blockade of neutrophils. Gastroenterology 133, 1882–1892 (2007).
Otte, J. M. et al. Effects of the cathelicidin LL-37 on intestinal epithelial barrier integrity. Regul. Pept. 156, 104–117 (2009).
Mangino, M. J., Brounts, L., Harms, B. & Heise, C. Lipoxin biosynthesis in inflammatory bowel disease. Prostaglandins Other Lipid Mediat. 79, 84–92 (2006).
Fiorucci, S. et al. A β-oxidation-resistant lipoxin A4 analog treats hapten-induced colitis by attenuating inflammation and immune dysfunction. Proc. Natl Acad. Sci. USA 101, 15736–15741 (2004).
Fournier, B. M. & Parkos, C. A. The role of neutrophils during intestinal inflammation. Mucosal Immunol. 5, 354–366 (2012).
Lood, C. et al. Neutrophil extracellular traps enriched in oxidized mitochondrial DNA are interferogenic and contribute to lupus-like disease. Nat. Med. 22, 146–153 (2016).
Knight, J. S. et al. Peptidylarginine deiminase inhibition reduces vascular damage and modulates innate immune responses in murine models of atherosclerosis. Circ. Res. 114, 947–956 (2014).
Wildhagen, K. C. et al. Nonanticoagulant heparin prevents histone-mediated cytotoxicity in vitro and improves survival in sepsis. Blood 123, 1098–1101 (2014).
Saffarzadeh, M. & Preissner, K. T. Fighting against the dark side of neutrophil extracellular traps in disease: manoeuvres for host protection. Curr. Opin. Hematol. 20, 3–9 (2013).
Barnado, A., Crofford, L. J. & Oates, J. C. At the bedside: neutrophil extracellular traps (NETs) as targets for biomarkers and therapies in autoimmune diseases. J. Leukoc. Biol. 99, 265–278 (2016).
Rossaint, J. & Zarbock, A. Tissue-specific neutrophil recruitment into the lung, liver, and kidney. J. Innate Immun. 5, 348–357 (2013).
Sager, H. B. et al. RNAi targeting multiple cell adhesion molecules reduces immune cell recruitment and vascular inflammation after myocardial infarction. Sci. Transl Med. 8, 342ra80 (2016).
Schmitt, M. M. et al. Endothelial junctional adhesion molecule-A guides monocytes into flow-dependent predilection sites of atherosclerosis. Circulation 129, 66–76 (2014).
Kempf, T. et al. GDF-15 is an inhibitor of leukocyte integrin activation required for survival after myocardial infarction in mice. Nat. Med. 17, 581–588 (2011).
Robertson, A. L. et al. A zebrafish compound screen reveals modulation of neutrophil reverse migration as an anti-inflammatory mechanism. Sci. Transl Med. 6, 225ra29 (2014).
Rossi, A. G. et al. Cyclin-dependent kinase inhibitors enhance the resolution of inflammation by promoting inflammatory cell apoptosis. Nat. Med. 12, 1056–1064 (2006).
Sydlik, U. et al. Recovery of neutrophil apoptosis by ectoine: a new strategy against lung inflammation. Eur. Respir. J. 41, 433–442 (2013).
Moon, C., Lee, Y. J., Park, H. J., Chong, Y. H. & Kang, J. L. N-Acetylcysteine inhibits RhoA and promotes apoptotic cell clearance during intense lung inflammation. Am. J. Respir. Crit. Care Med. 181, 374–387 (2010).
Vucic, E. et al. Regression of inflammation in atherosclerosis by the LXR agonist R211945: a noninvasive assessment and comparison with atorvastatin. JACC Cardiovasc. Imaging 5, 819–828 (2012).
Viola, J. R. et al. Resolving lipid mediators maresin 1 and resolvin D2 prevent athero-progression in mice. Circ. Res. 119, 1030–1038 (2016). This is the first study to show the therapeutic potential of pro-resolving lipid mediators during atheroprogression.
Börgeson, E. et al. Lipoxin A4 attenuates obesity-induced adipose inflammation and associated liver and kidney disease. Cell Metab. 22, 125–137 (2015).
Wang, Z., Li, J., Cho, J. & Malik, A. B. Prevention of vascular inflammation by nano-particle targeting of adherent neutrophils. Nat. Nanotechnol. 9, 204–210 (2014).
Dalli, J. et al. Heterogeneity in neutrophil microparticles reveals distinct proteome and functional properties. Mol. Cell. Proteomics 12, 2205–2219 (2013).
Gasser, O. & Schifferli, J. A. Activated polymorphonuclear neutrophils disseminate anti-inflammatory microparticles by ectocytosis. Blood 104, 2543–2548 (2004).
Dalli, J. & Serhan, C. N. Specific lipid mediator signatures of human phagocytes: micro-particles stimulate macrophage efferocytosis and pro-resolving mediators. Blood 120, e60–e72 (2012).
Eken, C. et al. Ectosomes released by polymorphonuclear neutrophils induce a MerTK-dependent anti-inflammatory pathway in macrophages. J. Biol. Chem. 285, 39914–39921 (2010).
Headland, S. E. et al. Neutrophil-derived microvesicles enter cartilage and protect the joint in inflammatory arthritis. Sci. Transl Med. 7, 315ra190 (2015).
Hasenberg, A. et al. Catchup: a mouse model for imaging-based tracking and modulation of neutrophil granulocytes. Nat. Methods 12, 445–452 (2015).
Bhattacharya, A. et al. Autophagy is required for neutrophil-mediated inflammation. Cell Rep. 12, 1731–1739 (2015).
Silvestre-Roig, C., Hidalgo, A. & Soehnlein, O. Neutrophil heterogeneity: implications for homeostasis and pathogenesis. Blood 127, 2173–2181 (2016).
Zhang, D. et al. Neutrophil ageing is regulated by the microbiome. Nature 525, 528–532 (2015).
Acknowledgements
The authors' research is supported by the Deutsche Forschungsgemeinschaft (grants SO876/6-1, SO876/11-1, SFB914-B08 and SFB1123-A01/A06/B05), the Nederlandse Organisatie voor Wetenschappelijk Onderzoek (VIDI project 91712303), the European Union (ITN Evolution) and the European Research Council (AdG°692511 PROVASC). A.H. is supported by the Ministry of Economy, Industry and Competitiveness (MINECO; grant SAF2015-65607-R). The Centro Nacional de Investigaciones Cardiovasculares (CNIC) is supported by the MINECO and the Pro CNIC Foundation, and is a Severo Ochoa Center of Excellence (MINECO award SEV-2015-0505).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Granulopoiesis
-
The production of mature granulocytes from their progenitors, which predominantly occurs in the bone marrow.
- Hypercholesterolaemia
-
A condition in which there are high levels of cholesterol in the plasma. Values below 200 mg per dl are desirable.
- Hyperglycaemia
-
A condition in which an excessive amount of glucose circulates in the blood plasma. The normal range is 80–120 mg per dl.
- Chemokinesis
-
An undirected, motile response of cells that is elicited by a chemical stimulus.
- Neutrophil priming
-
A state of enhanced neutrophil reactivity induced by molecules that by themselves do not activate neutrophils, but in conjunction with a pro-inflammatory molecule enhance the responsiveness to this pro-inflammatory molecule.
- Healing
-
Repair of a defect in the absence of the replacement of damaged tissue and achieved with minimal scar tissue.
- Specific granule deficiency
-
A rare congenital immunodeficiency that is mainly caused by mutations in CEBPE (which encodes CCAAT/enhancer-binding protein-ɛ) and leads to the lack of neutrophil granule proteins, such as human neutrophil peptides and human cationic antimicrobial protein 18 (the proform of LL-37).
- Efferocytosis
-
A term that is derived from the Latin word efferre (which means 'to take to the grave' or 'to bury') and describes a process by which dead cells are removed by professional phagocytes.
- Scarring
-
A feature of sites that have experienced tissue damage. Scar tissue remains at the end of the repair process and there is no substantial replacement of damaged tissue.
- Regeneration
-
The complete replacement of damaged tissue with new cells.
- Neointima formation
-
A process that often occurs as a consequence of arterial injury, and involves the proliferation of vascular smooth muscle cells and their migration to the tunica intima, which results in the thickening of arterial walls and a decreased arterial lumen diameter.
Rights and permissions
About this article
Cite this article
Soehnlein, O., Steffens, S., Hidalgo, A. et al. Neutrophils as protagonists and targets in chronic inflammation. Nat Rev Immunol 17, 248–261 (2017). https://doi.org/10.1038/nri.2017.10
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri.2017.10
This article is cited by
-
Association of immunologic findings of atheromatous plaques with subsequent cardiovascular events in patients with peripheral artery disease
Scientific Reports (2024)
-
Apoptosis in post-streptococcal glomerulonephritis and mechanisms for failed of inflammation resolution
Pediatric Nephrology (2024)
-
Hijacking CD177 for whole-body visualization of neutrophil dynamics
Nature Cardiovascular Research (2023)
-
Patients with schizophrenia and bipolar disorder display a similar global gene expression signature in whole blood that reflects elevated proportion of immature neutrophil cells with association to lipid changes
Translational Psychiatry (2023)
-
Biseugenol from Ocotea cymbarum (Lauraceae) attenuates inflammation, angiogenesis and collagen deposition of sponge-induced fibrovascular tissue in mice
Inflammopharmacology (2023)