Abstract
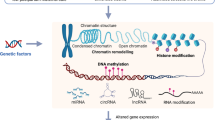
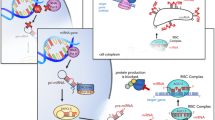
IBS is a common and debilitating disorder. The pathophysiology of IBS is poorly understood and is currently viewed as a biopsychosocial disorder with symptoms mediated via the brain–gut axis. Epidemiological studies of IBS point to risk factors such as familial clustering, sexual abuse and other forms of childhood trauma, low birth weight and gastrointestinal infection. Epigenetics focuses on the complex and dynamic interaction between the DNA sequence, DNA modifications and environmental factors, all of which combine to produce the phenotype. Studies in animal models of early stress and in humans who have experienced childhood trauma or abuse suggest that these events can lead to long-lasting epigenetic changes in the glucocorticoid receptor gene brought about by hypermethylation of a key regulatory component. Animal studies also indicate that the microbiota has a pivotal role in programming the core stress system, the hypothalamic–pituitary–adrenal axis and the immune system through epigenetic mechanisms. In this Perspectives, an epigenetic model of IBS is presented that incorporates many of the current findings regarding IBS, including proinflammatory markers, neuroendocrine alterations and links with both psychosocial stress and stress related to infection. We conclude that applying epigenetic methodology to this common and disabling disorder may help unravel its complex pathophysiology and lead to more effective treatments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hungin, A. P., Whorwell, P. J., Tack, J. & Mearin, F. The prevalence, patterns and impact of irritable bowel syndrome: an international survey of 40,000 subjects. Aliment. Pharmacol. Ther. 17, 643–650 (2003).
Quigley, E. M. Current concepts of the irritable bowel syndrome. Scand. J. Gastroenterol. Suppl. 1–8 (2003).
Lydiard, R. B. Irritable bowel syndrome, anxiety, and depression: what are the links? J. Clin. Psychiatry 62 (Suppl. 8), 38–45 (2001).
Lee, S. et al. Irritable bowel syndrome is strongly associated with generalized anxiety disorder: a community study. Aliment. Pharmacol. Ther. 30, 643–651 (2009).
Drossman, D. A. Presidential address: Gastrointestinal illness and the biopsychosocial model. Psychosom. Med. 60, 258–267 (1998).
Clarke, G., Quigley, E. M., Cryan, J. F. & Dinan, T. G. Irritable bowel syndrome: towards biomarker identification. Trends Mol. Med. 15, 478–489 (2009).
Grundmann, O. & Yoon, S. L. Irritable bowel syndrome: Epidemiology, diagnosis and treatment: An update for health-care practitioners. J. Gastroenterol. Hepatol. 25, 691–699 (2010).
Whorwell, P. J., McCallum, M., Creed, F. H. & Roberts, C. T. Non-colonic features of irritable bowel syndrome. Gut 27, 37–40 (1986).
Locke, G. R. 3rd et al. Familial association in adults with functional gastrointestinal disorders. Mayo Clin. Proc. 75, 907–912 (2000).
Kalantar, J. S. et al. Familial aggregation of irritable bowel syndrome: a prospective study. Gut 52, 1703–1707 (2003).
Saito, Y. A., Petersen, G. M., Locke, G. R. 3rd & Talley, N. J. The genetics of irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 3, 1057–1065 (2005).
Kanazawa, M. et al. Patients and nonconsulters with irritable bowel syndrome reporting a parental history of bowel problems have more impaired psychological distress. Dig. Dis. Sci. 49, 1046–1053 (2004).
Galton, F. Inquiries into Human Faculty and its Development (AMS Press, New York, 1973).
Morris-Yates, A. et al. Evidence of a genetic contribution to functional bowel disorder. Am. J. Gastroenterol. 93, 1311–1317 (1998).
Levy, R. L. et al. Irritable bowel syndrome in twins: heredity and social learning both contribute to etiology. Gastroenterology 121, 799–804 (2001).
Gottesman, I. I. & Shields, J. A. Critical review of recent adoption, twin and family studies of schizophrenia: Behavioural genetics perspectives. Schizophr. Bull. 2, 360–401 (1976).
Mohammed, I. et al. Genetic influences in irritable bowel syndrome: a twin study. Am. J. Gastroenterol. 100, 1340–1344 (2005).
Bengtson, M. B., Ronning, T., Vatn, M. H. & Harris, J. R. Irritable bowel syndrome in twins: genes and environment. Gut 55, 1754–1759 (2006).
Hotoleanu, C., Popp, R., Trifa, A. P., Nedelcu, L. & Dumitrascu, D. L. Genetic determinants of irritable bowel syndrome. World J. Gastroenterol. 14, 6636–6640 (2008).
Yeo, A. et al. Association between a functional polymorphism in the serotonin transporter gene and diarrhoea predominant irritable bowel syndrome in women. Gut 53, 1452–1458 (2004).
Kim, H. J., Camilleri, M. & Carlson, P. J. Association of distinct alpha(2) adrenoceptor and serotonin transporter polymorphisms with constipation and somatic symptoms in functional gastrointestinal disorders. Gut 53, 829–837 (2004).
Cremonini, F., Camilleri, M. & McKinzie, S. Effect of CCK-1 antagonist, dexloxiglumide, in female patients with irritable bowel syndrome: a pharmacodynamic and pharmacogenomic study. Am. J. Gastroenterol. 100, 652–663 (2005).
Ohman, L. & Simren, M. Pathogenesis of IBS: role of inflammation, immunity and neuroimmune interactions. Nat. Rev. Gastroenterol. Hepatol. 7, 163–173 (2010).
Camilleri, M. Genetics and irritable bowel syndrome: from genomics to intermediate phenotype and pharmacogenetics. Dig. Dis. Sci. 54, 2318–2324 (2009).
Haque, F. N., Gottesman, I. I. & Wong, A. H. Not really identical: epigenetic differences in monozygotic twins and implications for twin studies in psychiatry. Am. J. Med. Genet. 151C, 136–141 (2009).
Tang, W. Y. & Ho, S. M. Epigenetic reprogramming and imprinting in origins of disease. Rev. Endocr. Metab. Disord. 8, 173–182 (2007).
Mehler, M. F. Epigenetics and the nervous system. Ann. Neurol. 64, 602–617 (2008).
Mehler, M. F. Epigenetic principles and mechanisms underlying nervous system functions in health and disease. Prog. Neurobiol. 86, 305–341 (2008).
Mehler, M. F. & Mattick, J. S. Noncoding RNAs and RNA editing in brain development, functional diversification, and neurological disease. Physiol. Rev. 87, 799–823 (2007).
Farh, K. K. et al. The widespread impact of mammalian MicroRNAs on mRNA repression and evolution. Science 310, 1817–1821 (2005).
Lee, R. C., Feinbaum, R. L. & Ambros, V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75, 843–854 (1993).
Hobert, O. Gene regulation by transcription factors and microRNAs. Science 319, 1785–1786 (2008).
Valencia-Sanchez, M. A., Liu, J., Hannon, G. J. & Parker, R. Control of translation and mRNA degradation by miRNAs and siRNAs. Genes Dev. 20, 515–524 (2006).
Dinan, T. G. MicroRNAs as a target for novel antipsychotics: a systematic review of an emerging field. Int. J. Neuropsychopharmacol. 13, 395–404 (2010).
Heijmans, B. T. et al. Persistent epigenetic differences associated with prenatal exposure to famine in humans. Proc. Natl Acad. Sci. USA 105, 17046–17049 (2008).
Oh, G. & Petronis, A. Environmental studies of schizophrenia through the prism of epigenetics. Schizophr. Bull. 34, 1122–1129 (2008).
Baccarelli, A. & Bollati, V. Epigenetics and environmental chemicals. Curr. Opin. Pediatr. 21, 243–251 (2009).
Pembrey, M. et al. Sex specific, male-line transgenerational responses in humans. Eur. J. Hum. Genet. 14, 159–166 (2005).
Kaati, G., Bygren, L. O., Pembrey, M. & Sjostrom, M. Transgenerational response to nutrition, early life circumstances and longevity. Eur. J. Hum. Genet. 15, 784–790 (2007).
Stermer, E., Ber, H. & Levy, N. Chronic functional gastrointestinal symptoms in Holocaust survivors. Am. J. Gastroenterol. 86, 417–422 (2008).
Yehuda, R., Bell, A., Bierer, L. M. & Schmeidler, J. Maternal, not paternal, PTSD is related to increased risk for PTSD in offspring of Holocaust survivors. J. Psychiatr. Res. 42, 1104–1111 (2008).
Yehuda, R. & Bierer, L. M. The relevance of epigenetics to PTSD: implications for the DSM-V. J. Trauma Stress doi:10.1002/jts.20448.
Klooker, T. K. et al. Exposure to severe wartime conditions in early life is associated with an increased risk of irritable bowel syndrome: a population-based cohort study. Am. J. Gastroenterol. 104, 2250–2256 (2009).
Delvaux, M., Denis, P. & Allemand, H. Sexual abuse is more frequently reported by IBS patients than by patients with organic digestive diseases or controls. Results of a multicentre inquiry. French Club of Digestive Motility. Eur. J. Gastroenterol. Hepatol. 9, 345–352 (1997).
Walker, E. A., Katon, W. J., Roy-Byrne, P. P., Jemelka, R. P. & Russo, J. Histories of sexual victimization in patients with irritable bowel syndrome or inflammatory bowel disease. Am. J. Psychiatry 150, 1502–1506 (1993).
O'Mahony, S. M. et al. Early life stress alters behavior, immunity, and microbiota in rats: implications for irritable bowel syndrome and psychiatric illnesses. Biol. Psychiatry 65, 263–267 (2009).
Weaver, S. A., Diorio, J. & Meaney, M. J. Maternal separation leads to persistent reductions in pain sensitivity in female rats. J. Pain 8, 962–969 (2007).
McGowan, P. O. et al. Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nat. Neurosci. 12, 342–348 (2009).
McGowan, P. O. et al. Promoter-wide hypermethylation of the ribosomal RNA gene promoter in the suicide brain. PLoS ONE 3, e2085 (2008).
Cameron, N. M. et al. Epigenetic programming of phenotypic variations in reproductive strategies in the rat through maternal care. J. Neuroendocrinol. 20, 795–801 (2008).
Meaney, M. J., Szyf, M. & Seckl, J. R. Epigenetic mechanisms of perinatal programming of hypothalamic-pituitary-adrenal function and health. Trends Mol. Med. 13, 269–277 (2007).
Murgatroyd, C. et al. Dynamic DNA methylation programs persistent adverse effects of early-life stress. Nat. Neurosci. 12, 1559–1566 (2009).
Talley, N. J. Serotoninergic neuroenteric modulators. Lancet 358, 2061–2068 (2001).
Sudo, N. et al. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J. Physiol. 558, 263–275 (2004).
Shanahan, F. Gut microbes: from bugs to drugs. Am. J. Gastroenterol. 105, 275–279 (2010).
O'Mahony, C. et al. Commensal-induced regulatory T cells mediate protection against pathogen-stimulated NFκB activation. PLoS Pathog. 4, e1000112 (2008).
McKernan, D. P. et al. Toll-like receptor mRNA expression is selectively increased in the colonic mucosa of two animal models relevant to irritable bowel syndrome. PLoS ONE 4, e8226 (2010).
Kallio, P. et al. Dietary carbohydrate modification induces alterations in gene expression in abdominal subcutaneous adipose tissue in persons with the metabolic syndrome: the FUNGENUT Study. Am. J. Clin. Nutr. 85, 1417–1427 (2007).
O'Mahony, L. et al. A randomized, placebo-controlled, double-blind comparison of the probiotic bacteria lactobacillus and bifidobacterium in irritable bowel syndrome (IBS): symptom responses and relationship to cytokine profiles. Gastroenterology 128, 541–551 (2005).
Kapeller, J. et al. First evidence for an association of a functional variant in the microRNA-510 target site of the serotonin receptor-type 3E gene with diarrhea predominant irritable bowel syndrome. Hum. Mol. Genet. 17, 2967–2977 (2008).
Zhou, Q., Souba, W. W., Croce, C. M. & Verne, G. N. MicroRNA-29a regulates intestinal membrane permeability in patients with irritable bowel syndrome. Gut 59, 775–784 (2010).
Fitzgerald, P. et al. Tryptophan degradation in irritable bowel syndrome: evidence of indoleamine 2, 3-dioxygenase activation in a male cohort. BMC Gastroenterol. 9, 6 (2009).
Fitzgerald, P. et al. Tryptophan catabolism in females with irritable bowel syndrome: relationship to interferon-gamma, severity of symptoms and psychiatric co-morbidity. Neurogastroenterol. Motil. 20, 1291–1297 (2008).
Dinan, T. G. et al. Hypothalamic-pituitary-gut axis dysregulation in irritable bowel syndrome: Plasma cytokines as a potential biomarker? Gastroenterology 130, 304–311 (2006).
Acknowledgements
The authors are supported in part by Science Foundation Ireland in the form of a center grant (Alimentary Pharmabiotic Center), by the Health Research Board of Ireland, and the Higher Education Authority of Ireland. They are also in receipt of research funding from the pharmaceutical company GSK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Dinan, T., Cryan, J., Shanahan, F. et al. IBS: an epigenetic perspective. Nat Rev Gastroenterol Hepatol 7, 465–471 (2010). https://doi.org/10.1038/nrgastro.2010.99
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2010.99
This article is cited by
-
The association between irritable bowel syndrome and osteoporosis: a systematic review and meta-analysis
Osteoporosis International (2020)
-
Metabolome and microbiome profiling of a stress-sensitive rat model of gut-brain axis dysfunction
Scientific Reports (2019)
-
Enteric nervous system development: what could possibly go wrong?
Nature Reviews Neuroscience (2018)
-
Lessons learned — resolving the enigma of genetic factors in IBS
Nature Reviews Gastroenterology & Hepatology (2016)
-
'Gut health': a new objective in medicine?
BMC Medicine (2011)