Abstract
Type 2 diabetes mellitus (T2DM) has been related to alterations of oxidative metabolism in insulin-responsive tissues. Overt T2DM can present with acquired or inherited reductions of mitochondrial oxidative phosphorylation capacity, submaximal ADP-stimulated oxidative phosphorylation and plasticity of mitochondria and/or lower mitochondrial content in skeletal muscle cells and potentially also in hepatocytes. Acquired insulin resistance is associated with reduced insulin-stimulated mitochondrial activity as the result of blunted mitochondrial plasticity. Hereditary insulin resistance is frequently associated with reduced mitochondrial activity at rest, probably due to diminished mitochondrial content. Lifestyle and pharmacological interventions can enhance the capacity for oxidative phosphorylation and mitochondrial content and improve insulin resistance in some (pre)diabetic cases. Various mitochondrial features can be abnormal but are not necessarily responsible for all forms of insulin resistance. Nevertheless, mitochondrial abnormalities might accelerate progression of insulin resistance and subsequent organ dysfunction via increased production of reactive oxygen species. This Review discusses the association between mitochondrial function and insulin sensitivity in various tissues, such as skeletal muscle, liver and heart, with a main focus on studies in humans, and addresses the effects of therapeutic strategies that affect mitochondrial function and insulin sensitivity.
Key Points
-
Overt type 2 diabetes mellitus is associated with reduced oxidative phosphorylation capacity, submaximal ADP-stimulated oxidative phosphorylation and mitochondrial plasticity in insulin-responsive tissues
-
Acquired insulin resistance is associated with reduced insulin-stimulated mitochondrial plasticity that results in the inability of the organism to switch from fatty acid to glucose oxidation in skeletal muscle
-
Hereditary insulin resistance can be linked to reduced resting mitochondrial activity at least partly due to a decreased mitochondrial content
-
Lifestyle and pharmacological interventions can enhance oxidative phosphorylation capacity and mitochondrial content, and in most cases improve insulin resistance in (pre)diabetic states
-
Reduced oxidative phosphorylation capacity is unlikely to be the general cause of all forms of insulin resistance but might accelerate its progression and subsequent organ dysfunction via increased production of reactive oxygen species
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Roden, M. Non-invasive studies of glycogen metabolism in human skeletal muscle using nuclear magnetic resonance spectroscopy. Curr. Opin. Clin. Nutr. Metab. Care 4, 261–266 (2001).
Simoneau, J. A. & Kelley, D. E. Altered glycolytic and oxidative capacities of skeletal muscle contribute to insulin resistance in NIDDM. J. Appl. Physiol. 83, 166–171 (1997).
Ukropcova, B. et al. Family history of diabetes links impaired substrate switching and reduced mitochondrial content in skeletal muscle. Diabetes 56, 720–727 (2007).
Szendroedi, J. & Roden, M. Mitochondrial fitness and insulin sensitivity in humans. Diabetologia 51, 2155–2167 (2008).
Sivitz, W. I. & Yorek, M. A. Mitochondrial dysfunction in diabetes: from molecular mechanisms to functional significance and therapeutic opportunities. Antioxid. Redox. Signal 12, 537–577 (2010).
Lowell, B. B. & Shulman, G. I. Mitochondrial dysfunction and type 2 diabetes. Science 307, 384–387 (2005).
Sreekumar, R. & Nair, K. S. Skeletal muscle mitochondrial dysfunction & diabetes. Indian J. Med. Res. 125, 399–410 (2007).
Lanza, I. R. & Nair, K. S. Mitochondrial metabolic function assessed in vivo and in vitro. Curr. Opin. Clin. Nutr. Metab. Care 13, 511–517 (2010).
Kemp, G. J. The interpretation of abnormal 31P magnetic resonance saturation transfer measurements of Pi/ATP exchange in insulin-resistant skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 294, E640–E642 (2008).
Kraegen, E. W., Cooney, G. J. & Turner, N. Muscle insulin resistance: a case of fat overconsumption, not mitochondrial dysfunction. Proc. Natl Acad. Sci. USA 105, 7627–7628 (2008).
Holloszy, J. O. Skeletal muscle “mitochondrial deficiency” does not mediate insulin resistance. Am. J. Clin. Nutr. 89, 463S–466S (2009).
Schiff, M. et al. Mitochondria and diabetes mellitus: untangling a conflictive relationship? J. Inherit. Metab. Dis. 32, 684–698 (2009).
Erion, D. M. & Shulman, G. I. Diacylglycerol-mediated insulin resistance. Nat. Med. 16, 400–402 (2010).
Pagel-Langenickel, I., Bao, J., Pang, L. & Sack, M. N. The role of mitochondria in the pathophysiology of skeletal muscle insulin resistance. Endocr. Rev. 31, 25–51 (2010).
Chance, B. et al. Control of oxidative metabolism and oxygen delivery in human skeletal muscle: a steady-state analysis of the work/energy cost transfer function. Proc. Natl Acad. Sci. USA 82, 8384–8388 (1985).
Kacerovsky-Bielesz, G. et al. Short-term exercise training does not stimulate skeletal muscle ATP synthesis in relatives of humans with type 2 diabetes. Diabetes 58, 1333–1341 (2009).
Szendroedi, J. et al. Impaired mitochondrial function and insulin resistance of skeletal muscle in mitochondrial diabetes. Diabetes Care 32, 677–679 (2009).
Kemp, G. J. et al. Quantitative analysis by 31P magnetic resonance spectroscopy of abnormal mitochondrial oxidation in skeletal muscle during recovery from exercise. NMR Biomed. 6, 302–310 (1993).
Chance, B., Im, J., Nioka, S. & Kushmerick, M. Skeletal muscle energetics with PNMR: personal views and historic perspectives. NMR Biomed. 19, 904–926 (2006).
Prompers, J. J. et al. Dynamic MRS and MRI of skeletal muscle function and biomechanics. NMR Biomed. 19, 927–953 (2006).
Gnaiger, E. Capacity of oxidative phosphorylation in human skeletal muscle: new perspectives of mitochondrial physiology. Int. J. Biochem. Cell Biol. 41, 1837–1845 (2009).
Kemp, G. J., Meyerspeer, M. & Moser, E. Absolute quantification of phosphorus metabolite concentrations in human muscle in vivo by 31P MRS: a quantitative review. NMR Biomed. 20, 555–565 (2007).
Paganini, A. T., Foley, J. M. & Meyer, R. A. Linear dependence of muscle phosphocreatine kinetics on oxidative capacity. Am. J. Physiol. 272, C501–C510 (1997).
di Prampero, P. E. Factors limiting maximal performance in humans. Eur. J. Appl. Physiol. 90, 420–429 (2003).
Rossignol, R., Letellier, T., Malgat, M., Rocher, C. & Mazat, J. P. Tissue variation in the control of oxidative phosphorylation: implication for mitochondrial diseases. Biochem. J. 347 (Pt 1), 45–53 (2000).
Endo, M. & Iino, M. Measurement of Ca2+ release in skinned fibers from skeletal muscle. Methods Enzymol. 157, 12–26 (1988).
Chance, B. & Conrad, H. Acid-linked functions of intermediates in oxidative phosphorylation. II. Experimental studies of the effect of pH upon respiratory, phosphorylative and transfer activities of liver and heart mitochondria. J. Biol. Chem. 234, 1568–1570 (1959).
Brand, M. D. The efficiency and plasticity of mitochondrial energy transduction. Biochem. Soc. Trans. 33, 897–904 (2005).
Phielix, E. et al. Lower intrinsic ADP-stimulated mitochondrial respiration underlies in vivo mitochondrial dysfunction in muscle of male type 2 diabetic patients. Diabetes 57, 2943–2949 (2008).
Messer, J. I., Jackman, M. R. & Willis, W. T. Pyruvate and citric acid cycle carbon requirements in isolated skeletal muscle mitochondria. Am. J. Physiol. Cell Physiol. 286, C565–C572 (2004).
Pagel-Langenickel, I. et al. A discordance in rosiglitazone mediated insulin sensitization and skeletal muscle mitochondrial content/activity in type 2 diabetes mellitus. Am. J. Physiol. Heart Circ. Physiol. 293, H2659–H2666 (2007).
Hoppeler, H. & Fluck, M. Plasticity of skeletal muscle mitochondria: structure and function. Med. Sci. Sports Exerc. 35, 95–104 (2003).
Foretz, M. et al. Metformin inhibits hepatic gluconeogenesis in mice independently of the LKB1/AMPK pathway via a decrease in hepatic energy state. J. Clin. Invest. 120, 2355–2369 (2010).
Morino, K. et al. Reduced mitochondrial density and increased IRS-1 serine phosphorylation in muscle of insulin-resistant offspring of type 2 diabetic parents. J. Clin. Invest. 115, 3587–3593 (2005).
Patti, M. E. et al. Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: Potential role of PGC1 and NRF1. Proc. Natl Acad. Sci. USA 100, 8466–8471 (2003).
Yadava, N. & Nicholls, D. G. Spare respiratory capacity rather than oxidative stress regulates glutamate excitotoxicity after partial respiratory inhibition of mitochondrial complex I with rotenone. J. Neurosci. 27, 7310–7317 (2007).
Koves, T. R. et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 7, 45–56 (2008).
Jucker, B. M. et al. Assessment of mitochondrial energy coupling in vivo by 13C/31P NMR. Proc. Natl Acad. Sci. USA 97, 6880–6884 (2000).
Chomentowski, P., Coen, P. M., Radiková, Z., Goodpaster, B. H. & Toledo, F. G. Skeletal muscle mitochondria in insulin resistance: differences in intermyofibrillar versus subsarcolemmal subpopulations and relationship to metabolic flexibility. J. Clin. Endocrinol. Metab. 96, 494–503 (2011).
Schrauwen-Hinderling, V. B. et al. Impaired in vivo mitochondrial function but similar intramyocellular lipid content in patients with type 2 diabetes mellitus and BMI-matched control subjects. Diabetologia 50, 113–120 (2007).
Kelley, D. E., He, J., Menshikova, E. V. & Ritov, V. B. Dysfunction of mitochondria in human skeletal muscle in type 2 diabetes. Diabetes 51, 2944–2950 (2002).
Boushel, R. et al. Patients with type 2 diabetes have normal mitochondrial function in skeletal muscle. Diabetologia 50, 790–796 (2007).
Ritov, V. B. et al. Deficiency of subsarcolemmal mitochondria in obesity and type 2 diabetes. Diabetes 54, 8–14 (2005).
Mogensen, M. et al. Mitochondrial respiration is decreased in skeletal muscle of patients with type 2 diabetes. Diabetes 56, 1592–1599 (2007).
Stump, C. S., Short, K. R., Bigelow, M. L., Schimke, J. M. & Nair, K. S. Effect of insulin on human skeletal muscle mitochondrial ATP production, protein synthesis, and mRNA transcripts. Proc. Natl Acad. Sci. USA 100, 7996–8001 (2003).
Szendroedi, J. et al. Muscle mitochondrial ATP synthesis and glucose transport/phosphorylation in type 2 diabetes. PLoS Med. 4, e154 (2007).
Ritov, V. B. et al. Deficiency of electron transport chain in human skeletal muscle mitochondria in type 2 diabetes mellitus and obesity. Am. J. Physiol. Endocrinol. Metab. 298, E49–E58 (2010).
Hwang, H. et al. Proteomics analysis of human skeletal muscle reveals novel abnormalities in obesity and type 2 diabetes. Diabetes 59, 33–42 (2010).
Heilbronn, L. K., Gan, S. K., Turner, N., Campbell, L. V. & Chisholm, D. J. Markers of mitochondrial biogenesis and metabolism are lower in overweight and obese insulin-resistant subjects. J. Clin. Endocrinol. Metab. 92, 1467–1473 (2007).
Toledo, F. G. et al. Mitochondrial capacity in skeletal muscle is not stimulated by weight loss despite increases in insulin action and decreases in intramyocellular lipid content. Diabetes 57, 987–994 (2008).
Mootha, V. K. et al. PGC 1α responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet. 34, 267–273 (2003).
Karlsson, H. K., Ahlsen, M., Zierath, J. R., Wallberg-Henriksson, H. & Koistinen, H. A. Insulin signaling and glucose transport in skeletal muscle from first-degree relatives of type 2 diabetic patients. Diabetes 55, 1283–1288 (2006).
Lin, J., Handschin, C. & Spiegelman, B. M. Metabolic control through the PGC-1 family of transcription coactivators. Cell Metab. 1, 361–370 (2005).
Petersen, K. F. et al. Mitochondrial dysfunction in the elderly: possible role in insulin resistance. Science 300, 1140–1142 (2003).
Petersen, K. F., Dufour, S., Befroy, D., Garcia, R. & Shulman, G. I. Impaired mitochondrial activity in the insulin-resistant offspring of patients with type 2 diabetes. N. Engl. J. Med. 350, 664–671 (2004).
Szendroedi, J. et al. Reduced basal ATP synthetic flux of skeletal muscle in patients with previous acromegaly. PLoS ONE 3, e3958 (2008).
Schmid, A. I. et al. Comparison of measuring energy metabolism by different 31P MRS techniques in resting, ischemic and exercising muscle. Magn. Reson. Med. (in press).
Petersen, K. F., Dufour, S. & Shulman, G. I. Decreased insulin-stimulated ATP synthesis and phosphate transport in muscle of insulin-resistant offspring of type 2 diabetic parents. PLoS Med. 2, e233 (2005).
Karakelides, H., Irving, B. A., Short, K. R., O'Brien, P. & Nair, K. S. Age, obesity, and sex effects on insulin sensitivity and skeletal muscle mitochondrial function. Diabetes 59, 89–97 (2009).
Lefort, N. et al. Increased reactive oxygen species production and lower abundance of complex I subunits and carnitine palmitoyltransferase 1B protein despite normal mitochondrial respiration in insulin-resistant human skeletal muscle. Diabetes 59, 2444–2452 (2010).
Pospisilik, J. A. et al. Targeted deletion of AIF decreases mitochondrial oxidative phosphorylation and protects from obesity and diabetes. Cell 131, 476–491 (2007).
Nair, K. S. et al. Asian Indians have enhanced skeletal muscle mitochondrial capacity to produce ATP in association with severe insulin resistance. Diabetes 57, 1166–1175 (2008).
Reaven, G. M., Hollenbeck, C., Jeng, C. Y., Wu, M. S. & Chen, Y. D. Measurement of plasma glucose, free fatty acid, lactate, and insulin for 24 h in patients with NIDDM. Diabetes 37, 1020–1024 (1988).
Krssák, M. & Roden, M. The role of lipid accumulation in liver and muscle for insulin resistance and type 2 diabetes mellitus in humans. Rev. Endocr. Metab. Disord. 5, 127–134 (2004).
Roden, M. et al. Mechanism of free fatty acid-induced insulin resistance in humans. J. Clin. Invest. 97, 2859–2865 (1996).
Dresner, A. et al. Effects of free fatty acids on glucose transport and IRS 1 associated phosphatidylinositol 3-kinase activity. J. Clin. Invest. 103, 253–259 (1999).
Holland, W. L. et al. Lipid mediators of insulin resistance. Nutr. Rev. 65, S39–46 (2007).
Samuel, V. T. et al. Mechanism of hepatic insulin resistance in non-alcoholic fatty liver disease. J. Biol. Chem. 279, 32345–32353 (2004).
Høeg, L. D. et al. Lipid-induced insulin resistance affects women less than men and is not accompanied by inflammation or impaired proximal insulin signaling. Diabetes 60, 64–73 (2011).
Brehm, A. et al. Acute elevation of plasma lipids does not affect ATP synthesis in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 299, E33–E38 (2010).
Chavez, A. O. et al. Effect of short-term free fatty acids elevation on mitochondrial function in skeletal muscle of healthy individuals. J. Clin. Endocrinol. Metab. 95, 422–429 (2010).
Richardson, D. K. et al. Lipid infusion decreases the expression of nuclear encoded mitochondrial genes and increases the expression of extracellular matrix genes in human skeletal muscle. J. Biol. Chem. 280, 10290–10297 (2005).
Soeters, M. R. et al. Muscle acylcarnitines during short-term fasting in lean healthy men. Clin. Sci. (Lond.) 116, 585–592 (2009).
Turner, N. et al. Excess lipid availability increases mitochondrial fatty acid oxidative capacity in muscle: evidence against a role for reduced fatty acid oxidation in lipid-induced insulin resistance in rodents. Diabetes 56, 2085–2092 (2007).
Holloway, G. P., Gurd, B. J., Snook, L. A., Lally, J. & Bonen, A. Compensatory increases in nuclear PGC1α protein are primarily associated with subsarcolemmal mitochondrial adaptations in ZDF rats. Diabetes 59, 819–828 (2010).
Hancock, C. R. et al. High-fat diets cause insulin resistance despite an increase in muscle mitochondria. Proc. Natl Acad. Sci. USA 105, 7815–7820 (2008).
Iossa, S. et al. Effect of high-fat feeding on metabolic efficiency and mitochondrial oxidative capacity in adult rats. Br. J. Nutr. 90, 953–960 (2003).
Holloway, G. P. et al. Skeletal muscle mitochondrial FAT/CD36 content and palmitate oxidation are not decreased in obese women. Am. J. Physiol. Endocrinol. Metab. 292, E1782–E1789 (2007).
Bandyopadhyay, G. K., Yu, J. G., Ofrecio, J. & Olefsky, J. M. Increased malonyl-CoA levels in muscle from obese and type 2 diabetic subjects lead to decreased fatty acid oxidation and increased lipogenesis; thiazolidinedione treatment reverses these defects. Diabetes 55, 2277–2285 (2006).
Holloway, G. P., Bonen, A. & Spriet, L. L. Regulation of skeletal muscle mitochondrial fatty acid metabolism in lean and obese individuals. Am. J. Clin. Nutr. 89, 455S–462S (2009).
Noland, R. C. et al. Carnitine insufficiency caused by aging and overnutrition compromises mitochondrial performance and metabolic control. J. Biol. Chem. 284, 22840–22852 (2009).
Mihalik, S. J. et al. Increased levels of plasma acylcarnitines in obesity and type 2 diabetes and identification of a marker of glucolipotoxicity. Obesity (Silver Spring) 18, 1695–1700 (2010).
Newgard, C. B. et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 9, 311–326 (2009).
Adams, S. H. et al. Plasma acylcarnitine profiles suggest incomplete long-chain fatty acid beta-oxidation and altered tricarboxylic acid cycle activity in type 2 diabetic African-American women. J. Nutr. 139, 1073–1081 (2009).
Brehm, A. et al. Increased lipid availability impairs insulin-stimulated ATP synthesis in human skeletal muscle. Diabetes 55, 136–140 (2006).
Lim, E. L., Hollingsworth, K. G., Thelwall, P. E. & Taylor, R. Measuring the acute effect of insulin infusion on ATP turnover rate in human skeletal muscle using phosphorus-31 magnetic resonance saturation transfer spectroscopy. NMR Biomed. 23, 952–957 (2010).
Kacerovsky, M. et al. Impaired insulin stimulation of muscular ATP production in patients with type 1 diabetes. J. Intern. Med. 269, 189–199 (2011).
Cline, G. W. et al. Impaired glucose transport as a cause of decreased insulin-stimulated muscle glycogen synthesis in type 2 diabetes. N. Engl. J. Med. 341, 240–246 (1999).
Ortenblad, N. et al. Reduced insulin-mediated citrate synthase activity in cultured skeletal muscle cells from patients with type 2 diabetes: evidence for an intrinsic oxidative enzyme defect. Biochim. Biophys. Acta 1741, 206–214 (2005).
Krssák, M. et al. Alterations in postprandial hepatic glycogen metabolism in type 2 diabetes. Diabetes 53, 3048–3056 (2004).
Kotronen, A., Westerbacka, J., Bergholm, R., Pietiläinen, K. H. & Yki-Järvinen, H. Liver fat in the metabolic syndrome. J. Clin. Endocrinol. Metab. 92, 3490–3497 (2007).
Holland, W. L. et al. Inhibition of ceramide synthesis ameliorates glucocorticoid-, saturated fat, and obesity-induced insulin resistance. Cell Metab. 5, 167–179 (2007).
Mantena, S. K. et al. High fat diet induces dysregulation of hepatic oxygen gradients and mitochondrial function in vivo. Biochem. J. 417, 183–193 (2008).
Sanyal, A. J. et al. Nonalcoholic steatohepatitis: association of insulin resistance and mitochondrial abnormalities. Gastroenterology 120, 1183–1192 (2001).
Caldwell, S. H. et al. Mitochondrial abnormalities in non-alcoholic steatohepatitis. J. Hepatol. 31, 430–434 (1999).
Kawahara, H., Fukura, M., Tsuchishima, M. & Takase, S. Mutation of mitochondrial DNA in livers from patients with alcoholic hepatitis and nonalcoholic steatohepatitis. Alcohol Clin. Exp. Res. 31, S54–S60 (2007).
Sevastianova, K. et al. Nonalcoholic fatty liver disease: detection of elevated nicotinamide adenine dinucleotide phosphate with in vivo 3.0-T 31P MR spectroscopy with proton decoupling. Radiology 256, 466–473 (2010).
Miele, L. et al. Hepatic mitochondrial beta-oxidation in patients with nonalcoholic steatohepatitis assessed by 13C-octanoate breath test. Am. J. Gastroenterol. 98, 2335–2336 (2003).
Cortez-Pinto, H. et al. Alterations in liver ATP homeostasis in human nonalcoholic steatohepatitis: a pilot study. JAMA 282, 1659–1664 (1999).
Prikoszovich, T. et al. Body and liver fat mass rather than muscle mitochondrial function determine glucose metabolism in women with a history of gestational diabetes mellitus. Diabetes Care 34, 430–436 (2011).
Szendroedi, J. et al. Abnormal hepatic energy homeostasis in type 2 diabetes. Hepatology 50, 1079–1086 (2009).
Schmid, A. I. et al. Liver ATP synthesis is lower and relates to insulin sensitivity in patients with type 2 diabetes mellitus. Diabetes Care 34, 448–453 (2011).
Eckel, J., Wirdeier, A., Herberg, L. & Reinauer, H. Insulin resistance in the heart: studies on isolated cardiocytes of genetically obese Zucker rats. Endocrinology 116, 1529–1534 (1985).
Razeghi, P., Young, M. E., Cockrill, T. C., Frazier, O. H. & Taegtmeyer, H. Downregulation of myocardial myocyte enhancer factor 2C and myocyte enhancer factor 2C-regulated gene expression in diabetic patients with nonischemic heart failure. Circulation 106, 407–411 (2002).
Sharma, S. et al. Intramyocardial lipid accumulation in the failing human heart resembles the lipotoxic rat heart. FASEB J. 18, 1692–1700 (2004).
McGavock, J. M. et al. Cardiac steatosis in diabetes mellitus: a 1H-magnetic resonance spectroscopy study. Circulation 116, 1170–1175 (2007).
Rijzewijk, L. J. et al. Myocardial steatosis is an independent predictor of diastolic dysfunction in type 2 diabetes mellitus. J. Am. Coll. Cardiol. 52, 1793–1799 (2008).
Rijzewijk, L. J. et al. Altered myocardial substrate metabolism and decreased diastolic function in nonischemic human diabetic cardiomyopathy: studies with cardiac positron emission tomography and magnetic resonance imaging. J. Am. Coll. Cardiol. 54, 1524–1532 (2009).
Diamant, M. et al. Diastolic dysfunction is associated with altered myocardial metabolism in asymptomatic normotensive patients with well-controlled type 2 diabetes mellitus. J. Am. Coll. Cardiol. 42, 328–335 (2003).
Metzler, B. et al. Decreased high-energy phosphate ratios in the myocardium of men with diabetes mellitus type I. J. Cardiovasc. Magn. Reson. 4, 493–502 (2002).
Scheuermann-Freestone, M. et al. Abnormal cardiac and skeletal muscle energy metabolism in patients with type 2 diabetes. Circulation 107, 3040–3046 (2003).
Perseghin, G. et al. Abnormal left ventricular energy metabolism in obese men with preserved systolic and diastolic functions is associated with insulin resistance. Diabetes Care 30, 1520–1526 (2007).
Perseghin, G. et al. Increased mediastinal fat and impaired left ventricular energy metabolism in young men with newly found fatty liver. Hepatology 47, 51–58 (2008).
Anderson, E. J. et al. Substrate-specific derangements in mitochondrial metabolism and redox balance in the atrium of the type 2 diabetic human heart. J. Am. Coll. Cardiol. 54, 1891–1898 (2009).
Parra, V. et al. Changes in mitochondrial dynamics during ceramide-induced cardiomyocyte early apoptosis. Cardiovasc. Res. 77, 387–397 (2008).
Schwartz, M. W., Figlewicz, D. P., Baskin, D. G., Woods, S. C. & Porte, D. Jr. Insulin in the brain: a hormonal regulator of energy balance. Endocr. Rev. 13, 387–414 (1992).
Koch, L. et al. Central insulin action regulates peripheral glucose and fat metabolism in mice. J. Clin. Invest. 118, 2132–2147 (2008).
Schmoller, A. et al. Evidence for a relationship between body mass and energy metabolism in the human brain. J. Cereb. Blood Flow Metab. 30, 1403–1410 (2010).
Bischof, M. G. et al. Brain energy metabolism during hypoglycaemia in healthy and type 1 diabetic subjects. Diabetologia 47, 648–651 (2004).
Bischof, M. G. et al. Cerebral glutamate metabolism during hypoglycaemia in healthy and type 1 diabetic humans. Eur. J. Clin. Invest. 36, 164–169 (2006).
Rosen, E. D. & Spiegelman, B. M. Adipocytes as regulators of energy balance and glucose homeostasis. Nature 444, 847–853 (2006).
Bogacka, I., Xie, H., Bray, G. A. & Smith, S. R. Pioglitazone induces mitochondrial biogenesis in human subcutaneous adipose tissue in vivo. Diabetes 54, 1392–1399 (2005).
Mattson, M. P. Perspective: Does brown fat protect against diseases of aging? Ageing Res. Rev. 9, 69–76 (2010).
Lidell, M. E. & Enerbäck, S. Brown adipose tissue—a new role in humans? Nat. Rev. Endocrinol. 6, 319–325 (2010).
Ouellet, V. et al. Outdoor temperature, age, sex, body mass index, and diabetic status determine the prevalence, mass, and glucose-uptake activity of 18F FDG detected BAT in humans. J. Clin. Endocrinol. Metab. 96, 192–199 (2011).
Bartelt, A. et al. Brown adipose tissue activity controls triglyceride clearance. Nat. Med. 17, 200–205 (2011).
Holloszy, J. O. Biochemical adaptations in muscle. Effects of exercise on mitochondrial oxygen uptake and respiratory enzyme activity in skeletal muscle. J. Biol. Chem. 242, 2278–2282 (1967).
Rabøl, R. et al. Reduced skeletal muscle mitochondrial respiration and improved glucose metabolism in nondiabetic obese women during a very low calorie dietary intervention leading to rapid weight loss. Metabolism 58, 1145–1152 (2009).
Simoneau, J. A., Veerkamp, J. H., Turcotte, L. P. & Kelley, D. E. Markers of capacity to utilize fatty acids in human skeletal muscle: relation to insulin resistance and obesity and effects of weight loss. FASEB J. 13, 2051–2060 (1999).
Toledo, F. G. et al. Effects of physical activity and weight loss on skeletal muscle mitochondria and relationship with glucose control in type 2 diabetes. Diabetes 56, 2142–2147 (2007).
Meex, R. C. et al. Restoration of muscle mitochondrial function and metabolic flexibility in type 2 diabetes by exercise training is paralleled by increased myocellular fat storage and improved insulin sensitivity. Diabetes 59, 572–579 (2010).
Bruce, C. R., Kriketos, A. D., Cooney, G. J. & Hawley, J. A. Disassociation of muscle triglyceride content and insulin sensitivity after exercise training in patients with type 2 diabetes. Diabetologia 47, 23–30 (2004).
Hansen, D. et al. Continuous low- to moderate-intensity exercise training is as effective as moderate- to high-intensity exercise training at lowering blood HbA1c in obese type 2 diabetes patients. Diabetologia 52, 1789–1797 (2009).
Phielix, E., Meex, R., Moonen-Kornips, E., Hesselink, M. K. & Schrauwen, P. Exercise training increases mitochondrial content and ex vivo mitochondrial function similarly in patients with type 2 diabetes and in control individuals. Diabetologia 53, 1714–1721 (2010).
Petersen, K. F. et al. Reversal of nonalcoholic hepatic steatosis, hepatic insulin resistance, and hyperglycemia by moderate weight reduction in patients with type 2 diabetes. Diabetes 54, 603–608 (2005).
Tainter, M. L., Cutting, W. C. & Stockton, A. B. Use of dinitrophenol in nutritional disorders: a critical survey of clinical results. Am. J. Public Health Nations Health 24, 1045–1053 (1934).
Cha, B. S. et al. Impaired fatty acid metabolism in type 2 diabetic skeletal muscle cells is reversed by PPARγ agonists. Am. J. Physiol. Endocrinol. Metab. 289, E151–E159 (2005).
Coletta, D. K. et al. Pioglitazone stimulates AMP-activated protein kinase signalling and increases the expression of genes involved in adiponectin signalling, mitochondrial function and fat oxidation in human skeletal muscle in vivo: a randomised trial. Diabetologia 52, 723–732 (2009).
Belfort, R. et al. A placebo-controlled trial of pioglitazone in subjects with nonalcoholic steatohepatitis. N. Engl. J. Med. 355, 2297–2307 (2006).
Sanyal, A. J. et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N. Engl. J. Med. 362, 1675–1685 (2010).
Zou, M. H. et al. Activation of the AMP-activated protein kinase by the anti-diabetic drug metformin in vivo. Role of mitochondrial reactive nitrogen species. J. Biol. Chem. 279, 43940–43951 (2004).
Kukidome, D. et al. Activation of AMP-activated protein kinase reduces hyperglycemia-induced mitochondrial reactive oxygen species production and promotes mitochondrial biogenesis in human umbilical vein endothelial cells. Diabetes 55, 120–127 (2006).
Mathieu-Costello, O. et al. Regulation of skeletal muscle morphology in type 2 diabetic subjects by troglitazone and metformin: relationship to glucose disposal. Metabolism 52, 540–546 (2003).
Bujanda, L. et al. Resveratrol inhibits nonalcoholic fatty liver disease in rats. BMC Gastroenterol. 8, 40 (2008).
Lagouge, M. et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell 127, 1109–1122 (2006).
Nielsen, J. et al. Increased subsarcolemmal lipids in type 2 diabetes: effect of training on localization of lipids, mitochondria, and glycogen in sedentary human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 298, E706–E713 (2010).
Sreekumar, R., Halvatsiotis, P., Schimke, J. C. & Nair, K. S. Gene expression profile in skeletal muscle of type 2 diabetes and the effect of insulin treatment. Diabetes 51, 1913–1920 (2002).
Detmer, S. A. & Chan, D. C. Functions and dysfunctions of mitochondrial dynamics. Nat. Rev. Mol. Cell Biol. 8, 870–879 (2007).
Conley, K. E., Jubrias, S. A. & Esselman, P. C. Oxidative capacity and ageing in human muscle. J. Physiol. 526 (Pt 1), 203–210 (2000).
Nair, S., V, P. C., Arnold, C. & Diehl, A. M. Hepatic ATP reserve and efficiency of replenishing: comparison between obese and nonobese normal individuals. Am. J. Gastroenterol. 98, 466–470 (2003).
Chance, B., Eleff, S., Leigh, J. S. Jr, Sokolow, D. & Sapega, A. Mitochondrial regulation of phosphocreatine/inorganic phosphate ratios in exercising human muscle: a gated 31P NMR study. Proc. Natl Acad. Sci. USA 78, 6714–6718 (1981).
Binzoni, T., Ferretti, G., Schenker, K. & Cerretelli, P. Phosphocreatine hydrolysis by 31P-NMR at the onset of constant-load exercise in humans. J. Appl. Physiol. 73, 1644–1649 (1992).
Vorgerd, M. et al. Mitochondrial impairment of human muscle in Friedreich ataxia in vivo. Neuromuscul. Disord. 10, 430–435 (2000).
Lodi, R. et al. Antioxidant treatment improves in vivo cardiac and skeletal muscle bioenergetics in patients with Friedreich's ataxia. Ann. Neurol. 49, 590–596 (2001).
Brown, T. R., Ugurbil, K. & Shulman, R. G. 31P nuclear magnetic resonance measurements of ATPase kinetics in aerobic Escherichia coli cells. Proc. Natl Acad. Sci. USA 74, 5551–5553 (1977).
Schmid, A. I. et al. Quantitative ATP synthesis in human liver measured by localized 31P spectroscopy using the magnetization transfer experiment. NMR Biomed. 21, 437–443 (2008).
Diepart, C. et al. Comparison of methods for measuring oxygen consumption in tumor cells in vitro. Anal. Biochem. 396, 250–256 (2010).
Beeson, C. C., Beeson, G. C. & Schnellmann, R. G. A high-throughput respirometric assay for mitochondrial biogenesis and toxicity. Anal. Biochem. 404, 75–81 (2010).
Author information
Authors and Affiliations
Contributions
All authors researched the data and contributed equally to writing the article. J. Szendroedi and M. Roden provided substantial contributions to discussions of the content, and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M. Roden declares an association with the following company: Takeda (consultant). The other authors declare no competing interests.
Supplementary information
Supplementary Figure 1
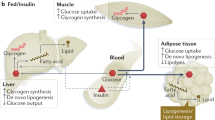
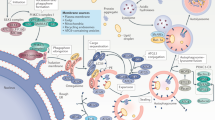
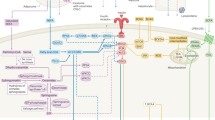
Cellular processes in insulin-responsive tissues. a. In the fasting state, glucose uptake into insulin-responsive tissues decreases and fuel selection switches from glucose to lipid oxidation (metabolic flexibility). Substrate availability and energy demand are low. b. The insulin-resistant state is characterized by the inability to adapt ATP synthetic flux rates (fATP) to substrate availability; lipid oxidation rates are, therefore, not increased adequately. Lipid metabolites (Acyl-CoA, DAG, ceramides) increase and TG accumulate. Mitochondrial plasticity, for example metabolic flexibility, is the limiting factor for in vivo ATP synthetic rates in insulin-resistant humans. c. During hyperinsulinemia, glucose uptake increases in insulin-responsive tissues, fuel selection switches from lipid to glucose oxidation (metabolic flexibility). In the insulin-stimulated state, energy demand is high and ATP synthetic flux rate is increased. d. In the insulin-resistant state, glucose transport and phosphorylation is reduced and lipid oxidation rates are not decreased adequately. DAG and ceramides induce insulin resistance. Abbreviations: DAG, diacylglycerol; FFA, free fatty acids; GLUT, glucose transporter; IR, insulin receptor; IRS, insulin receptor substrate; TCA, tricarboxylic acid; TG, triglycerides. (PPT 139 kb)
Rights and permissions
About this article
Cite this article
Szendroedi, J., Phielix, E. & Roden, M. The role of mitochondria in insulin resistance and type 2 diabetes mellitus. Nat Rev Endocrinol 8, 92–103 (2012). https://doi.org/10.1038/nrendo.2011.138
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2011.138
This article is cited by
-
Skeletal muscle TET3 promotes insulin resistance through destabilisation of PGC-1α
Diabetologia (2024)
-
The potential of therapeutic strategies targeting mitochondrial biogenesis for the treatment of insulin resistance and type 2 diabetes mellitus
Archives of Pharmacal Research (2024)
-
MRPS6 modulates glucose-stimulated insulin secretion in mouse islet cells through mitochondrial unfolded protein response
Scientific Reports (2023)
-
Mitochondria as a target for exercise-mitigated type 2 diabetes
Journal of Molecular Histology (2023)
-
Empagliflozin protects mice against diet-induced obesity, insulin resistance and hepatic steatosis
Diabetologia (2023)