Key Points
-
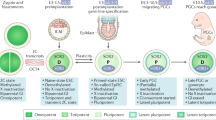
Testicular germ cell tumours (TGCTs) are the most frequent solid cancer in young adult Caucasian men; they have shown an increased incidence over the past four decades and are putatively derived from primordial germ cells (PGCs) or gonocytes.
-
Genome-wide association studies (GWAS) have revealed loci that suggest predisposition genes that are central to normal PGC biology (KIT ligand (KITLG; also known as SCF), sprouty 4 (SPRY4), BCL-2-antagonist/killer 1 (BAK1), telomerase reverse transcriptase (TERT), activating transcription factor 7 interacting protein (ATF7IP) and doublesex and mab-3-related transcription factor 1 (DMRT1)).
-
KITLG, SPRY4 and BAK1 are involved in KIT signalling, which is crucial to PGC migration and survival and is an important pathway that is frequently activated in TGCTs that occur in older men.
-
TERT and ATF7IP maintain telomere length and are reactivated in a range of tumour types.
-
DMRT1 is responsible for male sex determination. Experimental loss leads to TGCTs in mouse models and genomic gain and overexpression is seen in the spermatocytic seminoma subtype of TGCTs.
-
Spermatocytic seminomas exhibit overexpression and mutations of fibroblast growth factor 3 (FGFR3) and HRAS. These mutations confer a selective advantage to the spermatogonial stem cells within the testes and accrue over time, linking ageing to cancer predisposition and congenital syndromes in the offspring of affected gametes.
-
This improved understanding of TGCT predisposition and biology will lead to further refinements in the clinical management of this disease.
Abstract
Testicular germ cell tumours (TGCTs) of adults and adolescents are putatively derived from primordial germ cells or gonocytes. Recently reported genome-wide association studies implicate six gene loci that predispose to TGCT development. Remarkably, the functions of proteins encoded by genes within these regions bridge our understanding between the pathways involved in primordial germ cell physiology, male germ cell development and the molecular pathology of TGCTs. Furthermore, this improved understanding of the mechanisms underlying TGCT development and dissemination has clinical relevance for the management of patients with these tumours.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Oosterhuis, J. W. & Looijenga, L. H. Testicular germ-cell tumours in a broader perspective. Nature Rev. Cancer 5, 210–222 (2005).
Horwich, A., Shipley, J. & Huddart, R. Testicular germ-cell cancer. Lancet 367, 754–765 (2006).
Atkin, N. B. & Baker, M. C. Specific chromosome change, i(12p), in testicular tumours? Lancet 2, 1349 (1982).
Atkin, N. B. & Baker, M. C. i(12p): specific chromosomal marker in seminoma and malignant teratoma of the testis? Cancer Genet. Cytogenet. 10, 199–204 (1983).
Sandberg, A. A., Meloni, A. M. & Suijkerbuijk, R. F. Reviews of chromosome studies in urological tumors. III. Cytogenetics and genes in testicular tumors. J. Urol. 155, 1531–1556 (1996).
Henegariu, O., Vance, G. H., Heiber, D., Pera, M. & Heerema, N. A. Triple-color FISH analysis of 12p amplification in testicular germ-cell tumors using 12p band-specific painting probes. J. Mol. Med. 76, 648–655 (1998).
Rodriguez, S. et al. Expression profile of genes from 12p in testicular germ cell tumors of adolescents and adults associated with i(12p) and amplification at 12p11.2-p12.1. Oncogene 22, 1880–1891 (2003).
Roelofs, H. et al. Restricted 12p amplification and RAS mutation in human germ cell tumors of the adult testis. Am. J. Pathol. 157, 1155–1166 (2000).
Summersgill, B. et al. Molecular cytogenetic analysis of adult testicular germ cell tumours and identification of regions of consensus copy number change. Br. J. Cancer 77, 305–313 (1998).
Zafarana, G. et al. 12p-amplicon structure analysis in testicular germ cell tumors of adolescents and adults by array CGH. Oncogene 22, 7695–7701 (2003).
Clark, A. T. et al. Human STELLAR, NANOG, and GDF3 genes are expressed in pluripotent cells and map to chromosome 12p13, a hotspot for teratocarcinoma. Stem Cells 22, 169–179 (2004).
Korkola, J. E. et al. Down-regulation of stem cell genes, including those in a 200-kb gene cluster at 12p13.31, is associated with in vivo differentiation of human male germ cell tumors. Cancer Res. 66, 820–827 (2006).
Giuliano, C. J. et al. Retinoic acid represses a cassette of candidate pluripotency chromosome 12p genes during induced loss of human embryonal carcinoma tumorigenicity. Biochim. Biophys. Acta 1731, 48–56 (2005).
Skakkebaek, N. E. Possible carcinoma-in-situ of the testis. Lancet 2, 516–517 (1972).
Jacobsen, G. K. & Henriques, U. V. A fetal testis with intratubular germ cell neoplasia (ITGCN). Mod. Pathol. 5, 547–549 (1992).
Satge, D., Jacobsen, G. K., Cessot, F., Raffi, F. & Vekemans, M. A fetus with Down syndrome and intratubular germ cell neoplasia. Pediatr. Pathol. Lab. Med. 16, 107–112 (1996).
Jacobsen, G. K., Henriksen, O. B. & von der Maase, H. Carcinoma in situ of testicular tissue adjacent to malignant germ-cell tumors: a study of 105 cases. Cancer 47, 2660–2662 (1981).
Linke, J., Loy, V. & Dieckmann, K. P. Prevalence of testicular intraepithelial neoplasia in healthy males. J. Urol. 173, 1577–1579 (2005).
Oosterhuis, J. W., Stoop, H., Honecker, F. & Looijenga, L. H. Why human extragonadal germ cell tumours occur in the midline of the body: old concepts, new perspectives. Int. J. Androl. 30, 256–264 (2007).
Fossa, S. D. Long-term sequelae after cancer therapy-survivorship after treatment for testicular cancer. Acta Oncol. 43, 134–141 (2004).
Huddart, R. A. et al. Cardiovascular disease as a long-term complication of treatment for testicular cancer. J. Clin. Oncol. 21, 1513–1523 (2003).
Meinardi, M. T. et al. Cardiovascular morbidity in long-term survivors of metastatic testicular cancer. J. Clin. Oncol. 18, 1725–1732 (2000).
van den Belt-Dusebout, A. W. et al. Long-term risk of cardiovascular disease in 5-year survivors of testicular cancer. J. Clin. Oncol. 24, 467–475 (2006).
Travis, L. B. et al. Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. J. Natl Cancer Inst. 97, 1354–1365 (2005).
van den Belt-Dusebout, A. W. et al. Treatment-specific risks of second malignancies and cardiovascular disease in 5-year survivors of testicular cancer. J. Clin. Oncol. 25, 4370–4378 (2007).
Moul, J. W. et al. Testicular cancer in blacks. A multicenter experience. Cancer 73, 388–393 (1994).
Forman, D. et al. Familial testicular cancer: a report of the UK family register, estimation of risk and an HLA class 1 sib-pair analysis. Br. J. Cancer 65, 255–262 (1992).
Heimdal, K. et al. Familial testicular cancer in Norway and southern Sweden. Br. J. Cancer 73, 964–969 (1996).
Hemminki, K., Li, X. & Czene, K. Familial risk of cancer: data for clinical counseling and cancer genetics. Int. J. Cancer 108, 109–114 (2004).
Czene, K., Lichtenstein, P. & Hemminki, K. Environmental and heritable causes of cancer among 9.6 million individuals in the Swedish Family-Cancer Database. Int. J. Cancer 99, 260–266 (2002).
Fletcher, O. & Houlston, R. S. Architecture of inherited susceptibility to common cancer. Nature Rev. Cancer 10, 353–361 (2010).
Crockford, G. P. et al. Genome-wide linkage screen for testicular germ cell tumour susceptibility loci. Hum. Mol. Genet. 15, 443–451 (2006).
Rajpert-de Meyts, E. & Hoei-Hansen, C. E. From gonocytes to testicular cancer: the role of impaired gonadal development. Ann. N. Y. Acad. Sci. 1120, 168–180 (2007).
Ekbom, A., Richiardi, L., Akre, O., Montgomery, S. M. & Sparen, P. Age at immigration and duration of stay in relation to risk for testicular cancer among Finnish immigrants in Sweden. J. Natl Cancer Inst. 95, 1238–1240 (2003).
Montgomery, S. M., Granath, F., Ehlin, A., Sparen, P. & Ekbom, A. Germ-cell testicular cancer in offspring of Finnish immigrants to Sweden. Cancer Epidemiol. Biomarkers Prev. 14, 280–282 (2005).
Strohsnitter, W. C. et al. Cancer risk in men exposed in utero to diethylstilbestrol. J. Natl Cancer Inst. 93, 545–551 (2001).
Forman, D. et al. Aetiology of testicular cancer: association with congenital abnormalities, age at puberty, infertility, and exercise. United Kingdom Testicular Cancer Study Group. BMJ 308, 1393–1399 (1994).
Dieckmann, K. P. & Pichlmeier, U. Clinical epidemiology of testicular germ cell tumors. World J. Urol. 22, 2–14 (2004).
Baker, J. A., Buck, G. M., Vena, J. E. & Moysich, K. B. Fertility patterns prior to testicular cancer diagnosis. Cancer Causes Control 16, 295–299 (2005).
Doria-Rose, V. P., Biggs, M. L. & Weiss, N. S. Subfertility and the risk of testicular germ cell tumors (United States). Cancer Causes Control 16, 651–656 (2005).
Hotaling, J. M. & Walsh, T. J. Male infertility: a risk factor for testicular cancer. Nature Rev. Urol. 6, 550–556 (2009).
Walsh, T. J., Croughan, M. S., Schembri, M., Chan, J. M. & Turek, P. J. Increased risk of testicular germ cell cancer among infertile men. Arch. Intern. Med. 169, 351–356 (2009).
Skakkebaek, N. E., Rajpert-De Meyts, E. & Main, K. M. Testicular dysgenesis syndrome: an increasingly common developmental disorder with environmental aspects. Hum. Reprod. 16, 972–978 (2001).
Akre, O. & Richiardi, L. Does a testicular dysgenesis syndrome exist? Hum. Reprod. 24, 2053–2060 (2009).
Schnack, T. H., Poulsen, G., Myrup, C., Wohlfahrt, J. & Melbye, M. Familial coaggregation of cryptorchidism, hypospadias, and testicular germ cell cancer: a nationwide cohort study. J. Natl Cancer Inst. 102, 187–192 (2010).
Ohinata, Y. et al. A signaling principle for the specification of the germ cell lineage in mice. Cell 137, 571–584 (2009).
Saitou, M., Barton, S. C. & Surani, M. A. A molecular programme for the specification of germ cell fate in mice. Nature 418, 293–300 (2002).
Nakamura, T. et al. PGC7/Stella protects against DNA demethylation in early embryogenesis. Nature Cell Biol. 9, 64–71 (2007).
Farini, D., La Sala, G., Tedesco, M. & De Felici, M. Chemoattractant action and molecular signaling pathways of Kit ligand on mouse primordial germ cells. Dev. Biol. 306, 572–583 (2007).
Besmer, P. et al. The kit-ligand (steel factor) and its receptor c-kit/W: pleiotropic roles in gametogenesis and melanogenesis. Dev. Suppl. 125–137 (1993).
De Felici, M. et al. Experimental approaches to the study of primordial germ cell lineage and proliferation. Hum. Reprod. Update 10, 197–206 (2004).
De Miguel, M. P., Cheng, L., Holland, E. C., Federspiel, M. J. & Donovan, P. J. Dissection of the c-Kit signaling pathway in mouse primordial germ cells by retroviral-mediated gene transfer. Proc. Natl Acad. Sci. USA 99, 10458–10463 (2002). Experimental evidence for the importance of KIT and AKT in primordial germ cell physiology.
Runyan, C. et al. Steel factor controls midline cell death of primordial germ cells and is essential for their normal proliferation and migration. Development 133, 4861–4869 (2006).
Doitsidou, M. et al. Guidance of primordial germ cell migration by the chemokine SDF-1. Cell 111, 647–659 (2002). Details the importance of CXCL12 guiding CXCR4-expressing primordial germ cells towards the gonadal ridges.
Knaut, H. & Schier, A. F. Clearing the path for germ cells. Cell 132, 337–339 (2008).
Molyneaux, K. A. et al. The chemokine SDF1/CXCL12 and its receptor CXCR4 regulate mouse germ cell migration and survival. Development 130, 4279–4286 (2003).
Boldajipour, B. et al. Control of chemokine-guided cell migration by ligand sequestration. Cell 132, 463–473 (2008). Details how CXCR7 binds CXCL12 in non-target cells, maintaining correct CXCL12 gradients for the accurate guidance of primordial germ cells.
Boldajipour, B. & Raz, E. What is left behind-quality control in germ cell migration. Sci. STKE 2007, pe16 (2007).
Allegrucci, C., Thurston, A., Lucas, E. & Young, L. Epigenetics and the germline. Reproduction 129, 137–149 (2005).
Molyneaux, K. & Wylie, C. Primordial germ cell migration. Int. J. Dev. Biol. 48, 537–544 (2004).
Maldonado-Saldivia, J. et al. Dppa2 and Dppa4 are closely linked SAP motif genes restricted to pluripotent cells and the germ line. Stem Cells 25, 19–28 (2007).
Western, P. Foetal germ cells: striking the balance between pluripotency and differentiation. Int. J. Dev. Biol. 53, 393–409 (2009).
Yamaguchi, S., Kimura, H., Tada, M., Nakatsuji, N. & Tada, T. Nanog expression in mouse germ cell development. Gene Expr. Patterns 5, 639–646 (2005).
Wawersik, M. et al. Somatic control of germline sexual development is mediated by the JAK/STAT pathway. Nature 436, 563–567 (2005).
Sariola, H. & Saarma, M. Novel functions and signalling pathways for GDNF. J. Cell Sci. 116, 3855–3862 (2003).
Ohta, H., Tohda, A. & Nishimune, Y. Proliferation and differentiation of spermatogonial stem cells in the w/wv mutant mouse testis. Biol. Reprod. 69, 1815–1821 (2003).
Dadoune, J. P. New insights into male gametogenesis: what about the spermatogonial stem cell niche? Folia Histochem. Cytobiol. 45, 141–147 (2007).
Chen, C. et al. ERM is required for transcriptional control of the spermatogonial stem cell niche. Nature 436, 1030–1034 (2005).
Buaas, F. W. et al. Plzf is required in adult male germ cells for stem cell self-renewal. Nature Genet. 36, 647–652 (2004).
Costoya, J. A. et al. Essential role of Plzf in maintenance of spermatogonial stem cells. Nature Genet. 36, 653–659 (2004).
Labbaye, C. et al. A three-step pathway comprising PLZF/miR-146a/CXCR4 controls megakaryopoiesis. Nature Cell Biol. 10, 788–801 (2008).
Payne, C. J. et al. Sin3a is required by sertoli cells to establish a niche for undifferentiated spermatogonia, germ cell tumors, and spermatid elongation. Stem Cells 28, 1424–1434 (2010).
Korkola, J. E. et al. Gene expression-based classification of nonseminomatous male germ cell tumors. Oncogene 24, 5101–5107 (2005).
Port, M. et al. Gene expression profiling in seminoma and nonseminoma. J. Clin. Oncol. 23, 58–69 (2005).
Skotheim, R. I. et al. New insights into testicular germ cell tumorigenesis from gene expression profiling. Cancer Res. 62, 2359–2364 (2002).
Sperger, J. M. et al. Gene expression patterns in human embryonic stem cells and human pluripotent germ cell tumors. Proc. Natl Acad. Sci. USA 100, 13350–13355 (2003).
Almstrup, K. et al. Genome-wide gene expression profiling of testicular carcinoma in situ progression into overt tumours. Br. J. Cancer 92, 1934–1941 (2005).
Summersgill, B., Osin, P., Lu, Y. J., Huddart, R. & Shipley, J. Chromosomal imbalances associated with carcinoma in situ and associated testicular germ cell tumours of adolescents and adults. Br. J. Cancer 85, 213–220 (2001).
Draper, J. S. et al. Recurrent gain of chromosomes 17q and 12 in cultured human embryonic stem cells. Nature Biotech. 22, 53–54 (2004). Intriguing data reinforcing the importance of chromosome 12 components in undifferentiated lineages.
de Jong, J. et al. Diagnostic value of OCT3/4 for pre-invasive and invasive testicular germ cell tumours. J. Pathol. 206, 242–249 (2005).
Subramaniam, K. & Seydoux, G. Dedifferentiation of primary spermatocytes into germ cell tumors in, C. elegans lacking the pumilio-like protein PUF-8. Curr. Biol. 13, 134–139 (2003).
Guan, K. et al. Pluripotency of spermatogonial stem cells from adult mouse testis. Nature 440, 1199–1203 (2006).
Nathanson, K. L. et al. The Y deletion gr/gr and susceptibility to testicular germ cell tumor. Am. J. Hum. Genet. 77, 1034–1043 (2005).
Kanetsky, P. A. et al. Common variation in KITLG and at 5q31.3 predisposes to testicular germ cell cancer. Nature Genet. 41, 811–815 (2009).
Rapley, E. A. et al. A genome-wide association study of testicular germ cell tumor. Nature Genet. 41, 807–810 (2009).
Turnbull, C. et al. Variants near DMRT1, TERT and ATF7IP are associated with testicular germ cell cancer. Nature Genet. 42, 604–607 (2009). This reference and references 84 and 85 report the major GWAS of TGCTs, identifying predisposition genes that reinforce links between primordial germ cell biology and the malignant counterparts.
Izquierdo, M. A. et al. Differential expression of the c-kit proto-oncogene in germ cell tumours. J. Pathol. 177, 253–258 (1995).
Kemmer, K. et al. KIT mutations are common in testicular seminomas. Am. J. Pathol. 164, 305–313 (2004).
Looijenga, L. H. et al. Stem cell factor receptor (c-KIT) codon 816 mutations predict development of bilateral testicular germ-cell tumors. Cancer Res. 63, 7674–7678 (2003).
McIntyre, A. et al. Amplification and overexpression of the KIT gene is associated with progression in the seminoma subtype of testicular germ cell tumors of adolescents and adults. Cancer Res. 65, 8085–8089 (2005). Evidence of the importance of activation of KIT signalling in TGCTs.
Rapley, E. A. et al. Somatic mutations of KIT in familial testicular germ cell tumours. Br. J. Cancer 90, 2397–2401 (2004).
Willmore-Payne, C., Layfield, L. J. & Holden, J. A. c-KIT mutation analysis for diagnosis of gastrointestinal stromal tumors in fine needle aspiration specimens. Cancer 105, 165–170 (2005).
Goddard, N. C. et al. KIT and RAS signalling pathways in testicular germ cell tumours: new data and a review of the literature. Int. J. Androl. 30, 337–349 (2007).
Roskoski, R. Jr. Signaling by Kit protein-tyrosine kinase-the stem cell factor receptor. Biochem. Biophys. Res. Commun. 337, 1–13 (2005).
Heaney, J. D., Lam, M. Y., Michelson, M. V. & Nadeau, J. H. Loss of the transmembrane but not the soluble kit ligand isoform increases testicular germ cell tumor susceptibility in mice. Cancer Res. 68, 5193–5197 (2008).
Sasaki, A. et al. Mammalian Sprouty4 suppresses Ras-independent ERK activation by binding to Raf1. Cell Cycle 2, 281–282 (2003).
Frolov, A. et al. Response markers and the molecular mechanisms of action of Gleevec in gastrointestinal stromal tumors. Mol. Cancer Ther. 2, 699–709 (2003).
Tsumura, Y., Toshima, J., Leeksma, O. C., Ohashi, K. & Mizuno, K. Sprouty-4 negatively regulates cell spreading by inhibiting the kinase activity of testicular protein kinase. Biochem. J. 387, 627–637 (2005).
Yan, W., Samson, M., Jegou, B. & Toppari, J. Bcl-w forms complexes with Bax and Bak, and elevated ratios of Bax/Bcl-w and Bak/Bcl-w correspond to spermatogonial and spermatocyte apoptosis in the testis. Mol. Endocrinol. 14, 682–699 (2000).
McIntyre, A. et al. Activating mutations and/or expression levels of tyrosine kinase receptors GRB7, RAS, and BRAF in testicular germ cell tumors. Neoplasia 7, 1047–1052 (2005).
Olie, R. A. et al. N- and KRAS mutations in primary testicular germ cell tumors: incidence and possible biological implications. Genes Chromosom. Cancer 12, 110–116 (1995).
McIntyre, A. et al. Defining minimum genomic regions of imbalance involved in testicular germ cell tumors of adolescents and adults through genome wide microarray analysis of cDNA clones. Oncogene 23, 9142–9147 (2004).
Gilbert, D. C. et al. Clinical and biological significance of CXCL12 and CXCR4 expression in adult testes and germ cell tumours of adults and adolescents. J. Pathol. 217, 94–102 (2009).
Mosselman, S. et al. Aberrant platelet-derived growth factor α-receptor transcript as a diagnostic marker for early human germ cell tumors of the adult testis. Proc. Natl Acad. Sci. USA 93, 2884–2888 (1996).
Mandoky, L., Geczi, L., Bodrogi, I., Toth, J. & Bak, M. Expression of HER-2/neu in testicular tumors. Anticancer Res. 23, 3447–3451 (2003).
Gilbert, D. C. et al. Minimum regions of genomic imbalance in stage I testicular embryonal carcinoma and association of 22q loss with relapse. Genes Chromosom. Cancer 50, 186–195 (2011).
Campbell, P. M. & Der, C. J. Oncogenic Ras and its role in tumor cell invasion and metastasis. Semin. Cancer Biol. 14, 105–114 (2004).
Cox, A. D. & Der, C. J. The dark side of Ras: regulation of apoptosis. Oncogene 22, 8999–9006 (2003).
Downward, J. Targeting RAS signalling pathways in cancer therapy. Nature Rev. Cancer 3, 11–22 (2003).
Marte, B. M. & Downward, J. PKB/Akt: connecting phosphoinositide 3-kinase to cell survival and beyond. Trends Biochem. Sci. 22, 355–358 (1997).
Moe-Behrens, G. H. et al. Akt/PTEN signaling mediates estrogen-dependent proliferation of primordial germ cells in vitro. Mol. Endocrinol. 17, 2630–2638 (2003).
Di Vizio, D. et al. Loss of the tumor suppressor gene PTEN marks the transition from intratubular germ cell neoplasias (ITGCN) to invasive germ cell tumors. Oncogene 24, 1882–1894 (2005).
Teng, D. H. et al. MMAC1/PTEN mutations in primary tumor specimens and tumor cell lines. Cancer Res. 57, 5221–5225 (1997).
Kimura, T. et al. Conditional loss of PTEN leads to testicular teratoma and enhances embryonic germ cell production. Development 130, 1691–1700 (2003).
Sommerer, F. et al. Mutations of BRAF and RAS are rare events in germ cell tumours. Int. J. Cancer 113, 329–335 (2005).
Honecker, F. et al. Microsatellite instability, mismatch repair deficiency, and BRAF mutation in treatment-resistant germ cell tumors. J. Clin. Oncol. 27, 2129–2136 (2009).
Kortesidis, A. et al. Stromal-derived factor-1 promotes the growth, survival, and development of human bone marrow stromal stem cells. Blood 105, 3793–3801 (2005).
Dorsam, R. T. & Gutkind, J. S. G-protein-coupled receptors and cancer. Nature Rev. Cancer 7, 79–94 (2007).
Cabioglu, N. et al. CCR7 and CXCR4 as novel biomarkers predicting axillary lymph node metastasis in T1 breast cancer. Clin. Cancer Res. 11, 5686–5693 (2005).
Kodama, J., Hasengaowa, Seki, N., Kusumoto, T. & Hiramatsu, Y. Expression of the CXCR4 and CCR7 chemokine receptors in human endometrial cancer. Eur. J. Gynaecol. Oncol. 28, 370–375 (2007).
Muller, A. et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 410, 50–56 (2001).
Su, L. et al. Differential expression of CXCR4 is associated with the metastatic potential of human non-small cell lung cancer cells. Clin. Cancer Res. 11, 8273–8280 (2005).
Barbero, S. et al. Stromal cell-derived factor 1α stimulates human glioblastoma cell growth through the activation of both extracellular signal-regulated kinases 1/2 and Akt. Cancer Res. 63, 1969–1974 (2003).
Kijima, T. et al. Regulation of cellular proliferation, cytoskeletal function, and signal transduction through CXCR4 and c-Kit in small cell lung cancer cells. Cancer Res. 62, 6304–6311 (2002).
Yoon, Y. et al. CXC chemokine receptor-4 antagonist blocks both growth of primary tumor and metastasis of head and neck cancer in xenograft mouse models. Cancer Res. 67, 7518–7524 (2007).
Chinni, S. R. et al. CXCL12/CXCR4 signaling activates Akt-1 and MMP-9 expression in prostate cancer cells: the role of bone microenvironment-associated CXCL12. Prostate 66, 32–48 (2006).
Ehtesham, M., Winston, J. A., Kabos, P. & Thompson, R. C. CXCR4 expression mediates glioma cell invasiveness. Oncogene 25, 2801–2806 (2006).
Fernandis, A. Z., Prasad, A., Band, H., Klosel, R. & Ganju, R. K. Regulation of CXCR4-mediated chemotaxis and chemoinvasion of breast cancer cells. Oncogene 23, 157–167 (2004).
Wang, J. et al. Diverse signaling pathways through the SDF-1/CXCR4 chemokine axis in prostate cancer cell lines leads to altered patterns of cytokine secretion and angiogenesis. Cell. Signal. 17, 1578–1592 (2005).
Ara, T. Impaired colonization of the gonads by primordial germ cells in mice lacking a chemokine, stromal cell-derived factor-1 (SDF-1). Proc. Natl Acad. Sci. USA 100, 5319–5323 (2003).
Plotkin, J., Prockop, S. E., Lepique, A. & Petrie, H. T. Critical role for CXCR4 signaling in progenitor localization and T cell differentiation in the postnatal thymus. J. Immunol. 171, 4521–4527 (2003).
Janas, M. L. et al. Thymic development beyond β-selection requires phosphatidylinositol 3-kinase activation by CXCR4. J. Exp. Med. 207, 247–261 (2010).
Trampont, P. C. et al. CXCR4 acts as a costimulator during thymic β-selection. Nature Immunol. 11, 162–170 (2010).
Wendt, M. K., Cooper, A. N. & Dwinell, M. B. Epigenetic silencing of CXCL12 increases the metastatic potential of mammary carcinoma cells. Oncogene 27, 1461–1471 (2008).
Wendt, M. K. et al. Silencing of epithelial CXCL12 expression by DNA hypermethylation promotes colonic carcinoma metastasis. Oncogene 25, 4986–4997 (2006).
Dimberg, J., Hugander, A., Lofgren, S. & Wagsater, D. Polymorphism and circulating levels of the chemokine CXCL12 in colorectal cancer patients. Int. J. Mol. Med. 19, 11–15 (2007).
Hassan, S., Baccarelli, A., Salvucci, O. & Basik, M. Plasma stromal cell-derived factor-1: host derived marker predictive of distant metastasis in breast cancer. Clin. Cancer Res. 14, 446–454 (2008).
Drury, L. J., Wendt, M. K. & Dwinell, M. B. CXCL12 chemokine expression and secretion regulates colorectal carcinoma cell anoikis through bim-mediated intrinsic apoptosis. PLoS ONE 5, e1 2895.
Ferguson-Smith, M. The evolution of sex chromosomes and sex determination in vertebrates and the key role of DMRT1. Sex. Dev. 1, 2–11 (2007).
Smith, C. A., McClive, P. J., Western, P. S., Reed, K. J. & Sinclair, A. H. Conservation of a sex-determining gene. Nature 402, 601–602 (1999).
Veyrunes, F. et al. Bird-like sex chromosomes of platypus imply recent origin of mammal sex chromosomes. Genome Res. 18, 965–973 (2008).
Raymond, C. S. et al. Evidence for evolutionary conservation of sex-determining genes. Nature 391, 691–695 (1998).
Barbaro, M. et al. Characterization of deletions at 9p affecting the candidate regions for sex reversal and deletion 9p syndrome by MLPA. Eur. J. Hum. Genet. 17, 1439–1447 (2009).
Livadas, S. et al. Gonadoblastoma in a patient with del(9)(p22) and sex reversal: report of a case and review of the literature. Cancer Genet. Cytogenet. 143, 174–177 (2003).
Krentz, A. D. et al. The DM domain protein DMRT1 is a dose-sensitive regulator of fetal germ cell proliferation and pluripotency. Proc. Natl Acad. Sci. USA 106, 22323–22328 (2009). Expands the role of DMRT1 in normal germ cell biology, relevant as DMRT1 identified by GWAS as candidate predisposition gene for TGCTs.
Feldser, D. M., Hackett, J. A. & Greider, C. W. Telomere dysfunction and the initiation of genome instability. Nature Rev. Cancer 3, 623–627 (2003).
Hackett, J. A., Feldser, D. M. & Greider, C. W. Telomere dysfunction increases mutation rate and genomic instability. Cell 106, 275–286 (2001).
Shen, J. et al. Short telomere length and breast cancer risk: a study in sister sets. Cancer Res. 67, 5538–5544 (2007).
Widmann, T. A., Herrmann, M., Taha, N., Konig, J. & Pfreundschuh, M. Short telomeres in aggressive non-Hodgkin's lymphoma as a risk factor in lymphomagenesis. Exp. Hematol. 35, 939–946 (2007).
Wu, X. et al. Telomere dysfunction: a potential cancer predisposition factor. J. Natl Cancer Inst. 95, 1211–1218 (2003).
Fernandez-Garcia, I., Ortiz-de-Solorzano, C. & Montuenga, L. M. Telomeres and telomerase in lung cancer. J. Thorac. Oncol. 3, 1085–1088 (2008).
Kang, J. U., Koo, S. H., Kwon, K. C., Park, J. W. & Kim, J. M. Gain at chromosomal region 5p15.33, containing TERT, is the most frequent genetic event in early stages of non-small cell lung cancer. Cancer Genet. Cytogenet. 182, 1–11 (2008).
Zhang, A. et al. Genetic alterations in cervical carcinomas: frequent low-level amplifications of oncogenes are associated with human papillomavirus infection. Int. J. Cancer 101, 427–433 (2002).
Schrader, M. et al. The differentiation status of primary gonadal germ cell tumors correlates inversely with telomerase activity and the expression level of the gene encoding the catalytic subunit of telomerase. BMC Cancer 2, 32 (2002).
Schrader, M. et al. Quantification of human telomerase reverse transcriptase mRNA in testicular germ cell tumors by quantitative fluorescence real-time RT-PCR. Oncol. Rep. 9, 1097–1105 (2002).
Liu, L. et al. MCAF1/AM is involved in Sp1-mediated maintenance of cancer-associated telomerase activity. J. Biol. Chem. 284, 5165–5174 (2009).
Vastermark, K. et al. Polymorphic variation in the androgen receptor gene: association with risk of testicular germ cell cancer and metastatic disease. Eur. J. Cancer 47, 413–419 (2010).
Ferlin, A. et al. Male infertility and androgen receptor gene mutations: clinical features and identification of seven novel mutations. Clin. Endocrinol. 65, 606–610 (2006).
Suzuki, Y. et al. Screening for mutations of the androgen receptor gene in patients with isolated cryptorchidism. Fertil. Steril. 76, 834–836 (2001).
Suzuki, Y. et al. Mutation screening and CAG repeat length analysis of the androgen receptor gene in Klinefelter's syndrome patients with and without spermatogenesis. Hum. Reprod. 16, 1653–1656 (2001).
Wiener, J. S., Marcelli, M., Gonzales, E. T., Jr, Roth, D. R. & Lamb, D. J. Androgen receptor gene alterations are not associated with isolated cryptorchidism. J. Urol. 160, 863–865 (1998).
Aschim, E. L. et al. Linkage between cryptorchidism, hypospadias, and GGN repeat length in the androgen receptor gene. J. Clin. Endocrinol. Metab. 89, 5105–5109 (2004).
Ferlin, A. et al. Androgen receptor gene CAG and GGC repeat lengths in cryptorchidism. Eur. J. Endocrinol. 152, 419–425 (2005).
Radpour, R., Rezaee, M., Tavasoly, A., Solati, S. & Saleki, A. Association of long polyglycine tracts (GGN repeats) in exon 1 of the androgen receptor gene with cryptorchidism and penile hypospadias in Iranian patients. J. Androl. 28, 164–169 (2007).
Silva-Ramos, M. et al. The CAG repeat within the androgen receptor gene and its relationship to cryptorchidism. Int. Braz. J. Urol. 32, 330–335 (2006).
Chamberlain, N. L., Driver, E. D. & Miesfeld, R. L. The length and location of CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Res. 22, 3181–3186 (1994).
Choong, C. S., Kemppainen, J. A., Zhou, Z. X. & Wilson, E. M. Reduced androgen receptor gene expression with first exon CAG repeat expansion. Mol. Endocrinol. 10, 1527–1535 (1996).
Brooks, B. P. & Fischbeck, K. H. Spinal and bulbar muscular atrophy: a trinucleotide-repeat expansion neurodegenerative disease. Trends Neurosci. 18, 459–461 (1995).
Kazemi-Esfarjani, P., Trifiro, M. A. & Pinsky, L. Evidence for a repressive function of the long polyglutamine tract in the human androgen receptor: possible pathogenetic relevance for the (CAG)n-expanded neuronopathies. Hum. Mol. Genet. 4, 523–527 (1995).
Czeizel, A., Erodi, E. & Toth, J. Genetics of undescended testis. J. Urol. 126, 528–529 (1981).
Elert, A., Jahn, K., Heidenreich, A. & Hofmann, R. Population-based investigation of familial undescended testis and its association with other urogenital anomalies. J. Pediatr. Urol. 1, 403–407 (2005).
Toppari, J., Virtanen, H., Skakkebaek, N. E. & Main, K. M. Environmental effects on hormonal regulation of testicular descent. J. Steroid Biochem. Mol. Biol. 102, 184–186 (2006).
Rajpert-De Meyts, E. et al. The immunohistochemical expression pattern of Chk2, p53, p19INK4d, MAGE-A4 and other selected antigens provides new evidence for the premeiotic origin of spermatocytic seminoma. Histopathology 42, 217–226 (2003).
Goriely, A. et al. Activating mutations in FGFR3 and HRAS reveal a shared genetic origin for congenital disorders and testicular tumors. Nature Genet. 41, 1247–1252 (2009). Paternal age effect mutations in pathways central to normal germ cell biology and relevant to spermatocytic seminomas.
McIntyre, A. et al. Genomic copy number and expression patterns in testicular germ cell tumours. Br. J. Cancer 97, 1707–1712 (2007).
Sievers, S. et al. IGF2/H19 imprinting analysis of human germ cell tumors (GCTs) using the methylation-sensitive single-nucleotide primer extension method reflects the origin of GCTs in different stages of primordial germ cell development. Genes Chromosom. Cancer 44, 256–264 (2005).
Verdorfer, I., Rogatsch, H., Tzankov, A., Steiner, H. & Mikuz, G. Molecular cytogenetic analysis of human spermatocytic seminomas. J. Pathol. 204, 277–281 (2004).
Looijenga, L. H. et al. Genomic and expression profiling of human spermatocytic seminomas: primary spermatocyte as tumorigenic precursor and DMRT1 as candidate chromosome 9 gene. Cancer Res. 66, 290–302 (2006). Data implicating DMRT1 in tumorigenesis of spermatocytic seminomas, relating to implication from GWAS of a role as a TGCT predisposition gene.
Stoop, H. et al. Stem cell factor as a novel diagnostic marker for early malignant germ cells. J. Pathol. 216, 43–54 (2008).
Read, G. et al. Medical Research Council prospective study of surveillance for stage I testicular teratoma. Medical Research Council Testicular Tumors Working Party. J. Clin. Oncol. 10, 1762–1768 (1992).
Cullen, M. H. et al. Short-course adjuvant chemotherapy in high-risk stage I nonseminomatous germ cell tumors of the testis: a Medical Research Council report. J. Clin. Oncol. 14, 1106–1113 (1996).
Gilbert, D. C. et al. Treating stage I nonseminomatous germ cell tumours with a single cycle of chemotherapy. BJU Int. 98, 67–69 (2006).
Westermann, D. H. et al. Long-term followup results of 1 cycle of adjuvant bleomycin, etoposide and cisplatin chemotherapy for high risk clinical stage I nonseminomatous germ cell tumors of the testis. J. Urol. 179, 163–166 (2008).
Einhorn, L. H. Treatment of testicular cancer: a new and improved model. J. Clin. Oncol. 8, 1777–1781 (1990).
Koberle, B., Masters, J. R., Hartley, J. A. & Wood, R. D. Defective repair of cisplatin-induced DNA damage caused by reduced XPA protein in testicular germ cell tumours. Curr. Biol. 9, 273–276 (1999).
Masters, J. R. & Koberle, B. Curing metastatic cancer: lessons from testicular germ-cell tumours. Nature Rev. Cancer 3, 517–525 (2003).
Feldman, D. R. et al. Phase II trial of sunitinib in patients with relapsed or refractory germ cell tumors. Invest. New Drugs 28, 523–528 (2010).
Noel, E. E. et al. The association of CCND1 overexpression and cisplatin resistance in testicular germ cell tumors and other cancers. Am. J. Pathol. 176, 2607–2615 (2010).
Korkola, J. E. et al. Identification and validation of a gene expression signature that predicts outcome in adult men with germ cell tumors. J. Clin. Oncol. 27, 5240–5247 (2009).
Mueller, T. et al. Loss of Oct-3/4 expression in embryonal carcinoma cells is associated with induction of cisplatin resistance. Tumour Biol. 27, 71–83 (2006).
Beyrouthy, M. J. et al. High DNA methyltransferase 3B expression mediates 5-aza-deoxycytidine hypersensitivity in testicular germ cell tumors. Cancer Res. 69, 9360–9366 (2009).
Koster, R. et al. Cytoplasmic p21 expression levels determine cisplatin resistance in human testicular cancer. J. Clin. Invest. 120, 3594–3605 (2010).
Spierings, D. C. et al. Low p21Waf1/Cip1 protein level sensitizes testicular germ cell tumor cells to Fas-mediated apoptosis. Oncogene 23, 4862–4872 (2004).
Potter, J. D. Morphogens, morphostats, microarchitecture and malignancy. Nature Rev. Cancer 7, 464–474 (2007). Review highlighting the importance of the microenvironment in tumorigenesis, giving a different perspective on how the predisposition genes for TGCTs might promote tumour formation.
Stevens, L. C. The development of transplantable teratocarcinomas from intratesticular grafts of pre- and postimplantation mouse embryos. Dev. Biol. 21, 364–382 (1970).
Pierce, G. B. Jr, Dixon, F. J. Jr & Verney, E. L. Teratocarcinogenic and tissue-forming potentials of the cell types comprising neoplastic embryoid bodies. Lab. Invest. 9, 583–602 (1960).
Pettersson, A., Richiardi, L., Nordenskjold, A., Kaijser, M. & Akre, O. Age at surgery for undescended testis and risk of testicular cancer. N. Engl. J. Med. 356, 1835–1841 (2007).
Acknowledgements
The authors are grateful for funding from the Bob Champion Cancer Trust and the Medical Research Council (Grant 88238). They also acknowledge NHS funding to the NIHR Biomedical Research Centre. The authors are indebted to A. McIntyre, other members of the research team and R. Huddart for their help and support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Genome-wide association studies
-
These studies (also known as whole-genome association studies) seek to identify associations between SNPs with specific traits across the entire genome by comparing cases and controls.
- Primordial germ cells
-
Cells of the germ line that migrate to the gonadal ridges during embryogenesis. Expression profiling suggests that they are similar physiologically to TGCTs and represent a putative cell of origin.
- Intratubular germ cell neoplasia unclassified
-
Precursor lesions for TGCTs.
- Hypospadias
-
Developmental abnormality of the male genitalia, in which the urethra opens at a variable point along the underside of the penis.
- Genomic imprinting
-
The epigenetic modification of DNA that delineates parental origin. It is established during germ cell development and is responsible for the differential expression of imprinted genes in which monoallelic (either paternal or maternal) expression patterns occur.
Rights and permissions
About this article
Cite this article
Gilbert, D., Rapley, E. & Shipley, J. Testicular germ cell tumours: predisposition genes and the male germ cell niche. Nat Rev Cancer 11, 278–288 (2011). https://doi.org/10.1038/nrc3021
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3021
This article is cited by
-
Spectrum of Germ Cell Tumor (GCT): 5 Years’ Experience in a Tertiary Care Center and Utility of OCT4 as a Diagnostic Adjunct
Indian Journal of Surgical Oncology (2022)
-
Causal inference multiple imputation investigation of the impact of cannabinoids and other substances on ethnic differentials in US testicular cancer incidence
BMC Pharmacology and Toxicology (2021)
-
Human germ cell tumours from a developmental perspective
Nature Reviews Cancer (2019)
-
Increasing incidence of testicular cancer in the United States and Europe between 1992 and 2009
World Journal of Urology (2015)
-
A miR-199a/miR-214 Self-Regulatory Network via PSMD10, TP53 and DNMT1 in Testicular Germ Cell Tumor
Scientific Reports (2014)